Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Pharma Applications
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Pharma Applications
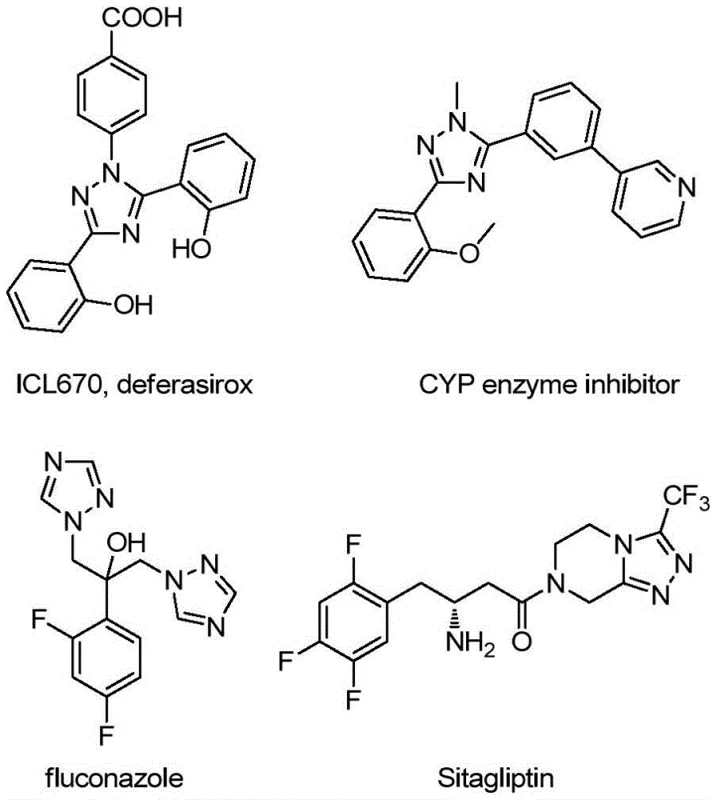
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways to construct complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is detailed in Chinese Patent CN113307790B, which discloses a highly efficient preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These nitrogen-containing heterocycles are not merely academic curiosities; they are foundational structures found in numerous bioactive molecular frameworks, ranging from potent antifungal agents to specialized ligands for organic light-emitting diodes (OLEDs). The ability to access these structures reliably is paramount for any organization aiming to be a reliable pharmaceutical intermediate supplier. The disclosed technology represents a paradigm shift from laborious multi-step syntheses to a streamlined, one-pot oxidative cyclization strategy that promises to redefine cost structures and supply chain reliability for high-purity pharmaceutical intermediates.
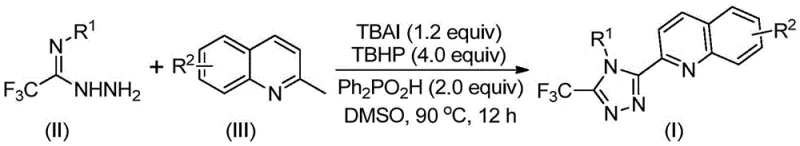
The strategic value of this patent lies in its direct address of long-standing bottlenecks in heterocyclic chemistry. Traditionally, the incorporation of a quinoline moiety onto a triazole ring has been fraught with synthetic challenges, often necessitating the use of expensive transition metal catalysts or proceeding through unstable intermediates that degrade process efficiency. By leveraging a metal-free catalytic system involving tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), this invention offers a pathway that is not only chemically elegant but also commercially viable. For R&D directors and procurement managers alike, understanding the nuances of this transformation is key to unlocking substantial cost savings and ensuring the continuity of supply for critical drug candidates that rely on this specific chemical architecture.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the methodology described in CN113307790B, the synthesis of quinolyl-substituted 1,2,4-triazoles was a formidable challenge that often deterred large-scale application. The background art highlights a traditional approach that utilizes quinoline-2-carboxylic acid as the primary starting material. This legacy route is plagued by inefficiency, requiring a tedious five-step reaction sequence to arrive at the target molecule. More critically, the overall yield of this conventional method is abysmal, reported at merely 17%, which renders it economically unfeasible for commercial manufacturing. Furthermore, the reaction conditions associated with these older methods are often severe, demanding strict anhydrous and anaerobic environments that increase operational complexity and safety risks. The reliance on such cumbersome protocols creates a fragile supply chain where minor deviations can lead to batch failures, making it difficult for manufacturers to guarantee the consistent delivery of high-purity OLED material or pharmaceutical precursors required by global clients.
The Novel Approach
In stark contrast to the limitations of the past, the novel approach disclosed in the patent utilizes readily available and inexpensive starting materials: 2-methylquinoline and trifluoroacetohydrazide. This new strategy employs a catalytic system driven by tetrabutylammonium iodide and tert-butyl peroxide to promote a direct oxidative cyclization reaction. This single-pot transformation bypasses the need for multiple isolation steps and eliminates the requirement for toxic heavy metal catalysts, which are often difficult to remove to the stringent ppm levels required in pharmaceutical applications. The reaction proceeds smoothly in common organic solvents like DMSO at moderate temperatures between 80°C and 100°C. This simplicity of operation, combined with the avoidance of water- and oxygen-free conditions, drastically lowers the barrier to entry for production. By simplifying the synthetic route, the method not only improves the overall yield significantly but also enhances the designability of the substrate, allowing for the introduction of diverse substituents at various positions on the aromatic rings.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core of this technological advancement lies in the intricate interplay between the iodide catalyst and the peroxide oxidant. The mechanism initiates with the oxidation of 2-methylquinoline by the TBAI/TBHP system to generate a reactive 2-quinoline carbaldehyde intermediate in situ. This aldehyde species then undergoes a condensation reaction with the trifluoroacetohydrazide to form a dehydrated hydrazone intermediate. Subsequently, the system facilitates an oxidative iodination followed by an intramolecular electrophilic substitution reaction. This cascade culminates in an aromatization step that constructs the stable 1,2,4-triazole ring fused with the quinoline system. The presence of diphenylphosphoric acid as an additive plays a crucial role in modulating the acidity and stabilizing the transition states, thereby driving the reaction towards completion with high selectivity. Understanding this mechanistic pathway is essential for process chemists aiming to optimize reaction parameters for commercial scale-up of complex pharmaceutical intermediates.
From an impurity control perspective, the metal-free nature of this catalytic cycle offers distinct advantages. Traditional transition-metal catalyzed couplings often leave behind trace amounts of palladium, copper, or nickel, which require expensive scavenging resins or complex recrystallization protocols to remove. In this iodide-promoted radical or electrophilic process, the primary byproducts are benign organic species and inorganic salts that are easily separated during the aqueous workup or silica gel filtration. The patent data indicates that the reaction tolerates a wide range of functional groups, including halogens, alkyls, and nitro groups, without significant side reactions. This robustness ensures that the impurity profile of the final product remains clean and predictable, facilitating faster regulatory approval processes for downstream drug substances. The ability to control the regioselectivity and minimize side products is a testament to the precision of this oxidative cyclization strategy.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and thermal conditions to maximize efficiency. The standard protocol involves charging a reactor with the requisite molar equivalents of tetrabutylammonium iodide, tert-butyl peroxide aqueous solution, diphenylphosphoric acid, trifluoroacetohydrazide, and 2-methylquinoline in a suitable organic solvent such as DMSO. The mixture is then heated to a temperature range of 80°C to 100°C and maintained under stirring for a duration of 8 to 14 hours. Upon completion, the reaction mixture is subjected to a straightforward workup procedure involving filtration and silica gel treatment, followed by column chromatography to isolate the pure product. For a detailed breakdown of the specific molar ratios and step-by-step operational parameters validated across multiple examples, please refer to the standardized synthesis guide below.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl peroxide (TBHP), diphenylphosphoric acid, trifluoroacetohydrazide, and 2-methylquinoline in DMSO solvent.
- Heat the reaction mixture to 80-100°C and stir for 8-14 hours to facilitate oxidative cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic benefits that extend beyond simple yield improvements. The elimination of precious metal catalysts removes a significant variable cost and mitigates the risk associated with the volatility of metal prices. Furthermore, the use of commodity chemicals like 2-methylquinoline and TBAI ensures that the raw material supply base is broad and resilient, reducing the likelihood of shortages that can disrupt production schedules. The simplified post-treatment process, which avoids complex extraction or metal scavenging steps, leads to a drastic reduction in processing time and solvent consumption. These factors collectively contribute to a more lean and agile manufacturing process that can respond quickly to market demands for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate impact of this technology is the substantial reduction in manufacturing costs driven by the exclusion of expensive transition metal catalysts. In traditional cross-coupling reactions, the cost of palladium or copper catalysts, along with the specialized ligands required to stabilize them, can constitute a significant portion of the bill of materials. By replacing these with inexpensive iodide salts and peroxides, the direct material cost is significantly lowered. Additionally, the high atom economy of the oxidative cyclization means less waste is generated per kilogram of product, which reduces waste disposal fees. The simplified purification process further lowers costs by reducing the consumption of silica gel and elution solvents, creating a compounding effect on overall profitability.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. 2-Methylquinoline and trifluoroacetic acid derivatives are bulk commodities produced by numerous chemical manufacturers globally, ensuring that there is no single point of failure in the supply network. Unlike specialized organometallic reagents that may have long lead times or limited suppliers, the reagents for this process can be sourced locally in most major chemical hubs. This availability allows for the maintenance of healthy inventory levels without tying up excessive capital. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in raw material quality, further stabilizing the supply of the final intermediate.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly smaller than that of conventional methods, aligning with modern green chemistry principles and increasingly strict environmental regulations. The absence of heavy metals simplifies the treatment of effluent streams, reducing the burden on wastewater treatment facilities and lowering compliance costs. The reaction's ability to proceed in polar aprotic solvents like DMSO, which can be recovered and recycled, enhances the sustainability profile of the operation. From a scalability standpoint, the exothermic nature of the oxidation is manageable, and the lack of sensitive catalysts makes the transfer from gram-scale to multi-ton production straightforward. This ease of scale-up ensures that the technology can meet the growing demands of the pharmaceutical industry without requiring massive capital investment in new specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These answers are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear picture of what partners can expect when integrating this chemistry into their operations. Understanding these details is crucial for making informed decisions about process adoption and vendor selection.
Q: What are the advantages of this metal-free synthesis over traditional methods?
A: Unlike traditional methods requiring 5 steps and harsh conditions with only 17% yield, this novel route uses cheap starting materials, operates under mild conditions without heavy metals, and achieves significantly higher yields suitable for scale-up.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states the reaction can be easily expanded to gram-scale and provides a foundation for future large-scale production due to simple operation and easy post-treatment.
Q: What solvents are compatible with this oxidative cyclization?
A: While DMF and dioxane can be used, DMSO is the preferred organic solvent as it effectively dissolves starting materials and promotes high conversion rates for the triazole formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route disclosed in CN113307790B for the production of advanced pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole derivative meets the highest industry standards. We are committed to leveraging this metal-free technology to deliver superior value to our global partners.
We invite R&D directors and procurement specialists to engage with our technical team to explore how this efficient synthesis can be tailored to your specific project needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this novel route. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your drug development timeline while optimizing your manufacturing budget.
