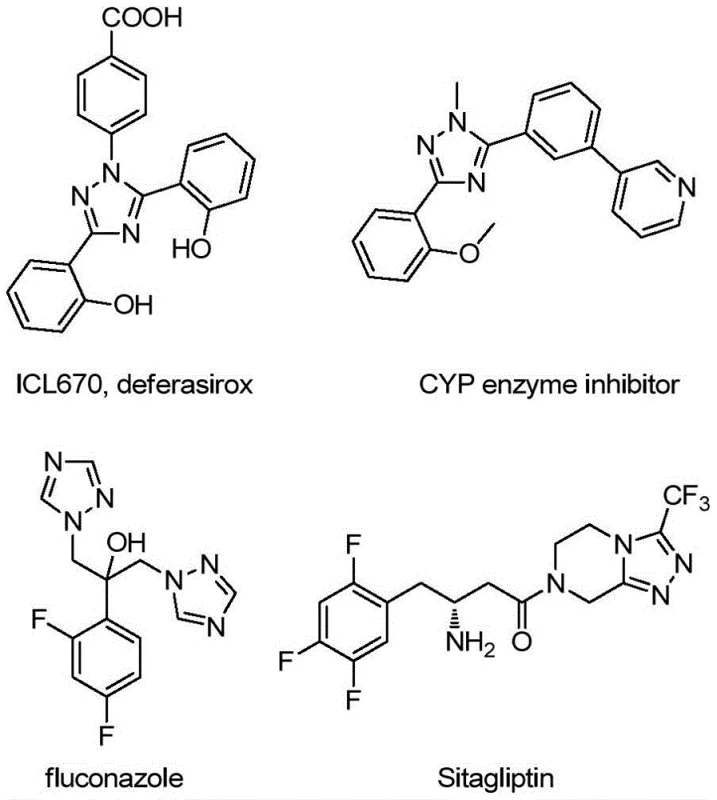
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Intermediates
The rapid evolution of heterocyclic chemistry has placed 1,2,4-triazole derivatives at the forefront of modern drug discovery and agrochemical development. As detailed in the groundbreaking patent CN113307790B, a novel preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds has been established, offering a robust alternative to traditional synthetic routes. These nitrogen-containing heterocycles are critical scaffolds found in numerous bioactive molecular frameworks, serving not only as key pharmacophores in active pharmaceutical ingredients (APIs) but also as versatile ligands in coordination chemistry for organic light-emitting diodes (OLEDs). The strategic incorporation of both quinoline and trifluoromethyl moieties into the triazole core significantly enhances the lipophilicity and metabolic stability of the resulting molecules, making them highly desirable candidates for next-generation therapeutics. This technical insight report analyzes the transformative potential of this metal-free oxidative cyclization strategy for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficient multi-step protocols that hinder large-scale manufacturing viability. Prior art methodologies typically rely on quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to achieve the final target structure. This traditional approach suffers from severe reaction conditions that often require stringent control over temperature and pressure, alongside the use of hazardous reagents that complicate waste management. Furthermore, the cumulative yield of such linear syntheses is notoriously poor, with reported total yields hovering around a mere 17%, rendering the process economically unfeasible for industrial applications. The reliance on multiple isolation and purification steps between each transformation not only increases solvent consumption but also extends the overall production lead time, creating bottlenecks for procurement teams seeking reliable sources of high-purity intermediates.
The Novel Approach
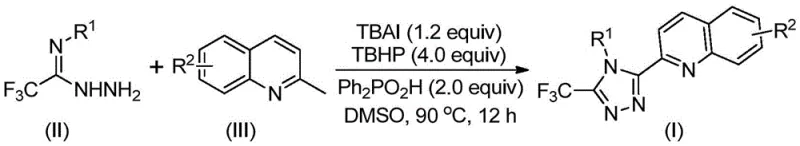
In stark contrast, the methodology disclosed in patent CN113307790B introduces a streamlined, one-pot oxidative cyclization strategy that fundamentally reshapes the economic landscape of triazole manufacturing. By utilizing cheap and readily available 2-methylquinoline and trifluoroacetimidoyl hydrazide as starting materials, this novel route bypasses the need for pre-functionalized carboxylic acid derivatives. The reaction is promoted by a synergistic catalytic system comprising tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), which facilitates the direct construction of the triazole ring under relatively mild thermal conditions. This approach eliminates the necessity for anhydrous or oxygen-free environments, allowing the reaction to proceed efficiently under ambient atmospheric conditions. The simplicity of the operation, combined with the ability to tolerate a wide range of functional groups on the substrate, significantly broadens the applicability of this method for generating diverse libraries of bioactive compounds without compromising on yield or purity.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core innovation of this synthesis lies in its elegant mechanistic pathway, which avoids the use of toxic transition metals while maintaining high catalytic efficiency. The reaction initiates with the oxidation of 2-methylquinoline to the corresponding 2-quinolinecarbaldehyde intermediate, driven by the TBAI and TBHP system. This aldehyde species subsequently undergoes a condensation reaction with the trifluoroacetimidoyl hydrazide to form a dehydrated hydrazone intermediate. Following this condensation, the system promotes an oxidative iodination event, which activates the methyl group for subsequent intramolecular electrophilic substitution. This cyclization step is crucial for closing the five-membered triazole ring, followed by a final aromatization process that yields the stable 3-quinolyl-5-trifluoromethyl substituted product. Alternatively, the reaction may proceed via a radical mechanism, where iodine radicals generated in situ abstract hydrogen atoms to propagate the oxidative cycle. This dual possibility ensures robust conversion rates across various substrate electronic environments.
From an impurity control perspective, the choice of diphenylphosphinic acid as an additive plays a pivotal role in stabilizing the reaction intermediates and suppressing side reactions. The use of DMSO as the preferred solvent ensures excellent solubility for all polar and non-polar components, facilitating homogeneous reaction kinetics that minimize the formation of oligomeric byproducts. The absence of heavy metal residues is a critical quality attribute for pharmaceutical intermediates, as it removes the need for complex and costly metal scavenging steps during downstream processing. This metal-free nature simplifies the regulatory filing process for API manufacturers, as the impurity profile is significantly cleaner compared to palladium or copper-catalyzed alternatives. The high selectivity of this oxidative cyclization ensures that the trifluoromethyl group remains intact throughout the process, preserving the desired physicochemical properties of the final molecule.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of scale-up, requiring standard laboratory equipment without specialized high-pressure reactors. The protocol involves simply mixing the stoichiometric amounts of tetrabutylammonium iodide, aqueous tert-butyl hydroperoxide, diphenylphosphinic acid, the hydrazide derivative, and 2-methylquinoline in dimethyl sulfoxide (DMSO). The mixture is then heated to a temperature range of 80-100°C and maintained for 8 to 14 hours to ensure complete conversion. Post-reaction processing is straightforward, involving filtration to remove insoluble salts, followed by adsorption onto silica gel and purification via standard column chromatography. This operational simplicity makes it an ideal candidate for technology transfer from R&D to pilot plant scales.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphinic acid, trifluoroacetimidoyl hydrazide, and 2-methylquinoline in DMSO solvent.
- Heat the reaction mixture to 80-100°C and stir for 8-14 hours to facilitate oxidative cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from a five-step linear synthesis to a single-pot oxidative cyclization represents a paradigm shift in cost structure and logistical reliability. The elimination of multiple intermediate isolation steps drastically reduces the consumption of solvents and silica gel, directly lowering the variable costs associated with manufacturing. Furthermore, the removal of expensive transition metal catalysts not only reduces raw material expenses but also mitigates the supply risk associated with fluctuating prices of precious metals like palladium or rhodium. The ability to run the reaction under ambient air conditions removes the capital expenditure requirement for gloveboxes or extensive nitrogen purging systems, making the technology accessible to a wider range of contract manufacturing organizations (CMOs).
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the atom economy and step reduction. By consolidating five synthetic transformations into a single operational unit, labor costs and energy consumption are significantly minimized. The use of commodity chemicals such as TBAI and TBHP, which are produced on a massive industrial scale, ensures long-term price stability and availability. Additionally, the avoidance of heavy metals eliminates the downstream costs associated with metal residue testing and specialized waste disposal, contributing to a leaner overall cost of goods sold (COGS) structure for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, commercially available starting materials that are not subject to the geopolitical constraints often seen with rare earth catalysts. The high tolerance for functional groups means that a single standardized protocol can be used to produce a wide variety of analogues, allowing manufacturers to respond rapidly to changing market demands without re-validating entirely new processes. The simplified workflow reduces the probability of batch failures due to operator error or equipment malfunction, ensuring consistent on-time delivery performance for downstream customers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with green chemistry principles. The reduction in solvent usage and the absence of toxic heavy metals lower the environmental footprint of the manufacturing process. The reaction conditions are mild enough to be safely scaled from gram-scale laboratory experiments to multi-kilogram pilot batches without encountering exothermic runaway risks. This inherent safety profile facilitates smoother regulatory approvals and reduces the insurance and compliance overheads associated with hazardous chemical processing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the scalability and substrate scope of this patented synthesis method. Understanding these details is crucial for R&D teams evaluating this route for their specific pipeline projects. The answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-making.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method utilizes an organic catalytic system involving tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), completely eliminating the need for toxic or costly heavy metal catalysts.
Q: What is the substrate tolerance for this reaction?
A: The method demonstrates excellent functional group tolerance, successfully accommodating various substituents on the aryl ring such as methyl, methoxy, halogens (F, Cl, Br), and nitro groups, with yields ranging from 51% to 97%.
Q: Is strict anhydrous or anaerobic condition required?
A: No, a significant advantage of this protocol is that it operates effectively under ambient atmospheric conditions without the need for rigorous anhydrous or oxygen-free environments, simplifying operational requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
As the pharmaceutical industry continues to demand more efficient and sustainable manufacturing pathways, NINGBO INNO PHARMCHEM stands ready to leverage this advanced oxidative cyclization technology for your project needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial supply is seamless. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole meets the exacting standards required for GMP API synthesis. Our commitment to quality assurance ensures that impurity profiles are tightly controlled, supporting your regulatory filings with comprehensive analytical data packages.
We invite you to engage with our technical procurement team to discuss how this metal-free synthesis can optimize your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this novel route for your intermediates. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique molecular requirements, ensuring a partnership built on technical excellence and commercial value.
