Advanced Catalytic Synthesis of Oxindole Compounds for Commercial Scale-up
Advanced Catalytic Synthesis of Oxindole Compounds for Commercial Scale-up
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for constructing privileged heterocyclic scaffolds, among which the oxindole core remains paramount due to its prevalence in bioactive molecules. Patent CN102875444A discloses a highly efficient synthetic methodology for oxidized indole compounds that addresses critical bottlenecks in traditional manufacturing. This technical insight report analyzes the proprietary process which leverages a copper-catalyzed intermolecular arylation strategy, offering a compelling alternative to legacy methods. By utilizing readily available aniline derivatives and beta-dicarbonyl compounds, the disclosed route achieves high chemical yields under mild conditions, positioning it as a viable solution for cost reduction in API manufacturing. The integration of L-Proline as a ligand alongside cuprous iodide not only enhances reaction kinetics but also simplifies the downstream purification landscape, a key consideration for any reliable pharmaceutical intermediate supplier aiming to deliver consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indole and oxindole framework has relied heavily on classical methodologies such as the Fischer indole synthesis or the Larock heteroannulation. While chemically valid, these traditional routes often suffer from significant operational drawbacks that hinder their application in modern, regulated supply chains. The Fischer synthesis, for instance, frequently necessitates the use of strong mineral acids and elevated temperatures, leading to severe selectivity issues and the generation of complex impurity profiles that are difficult to separate. Furthermore, palladium-catalyzed approaches like the Larock method, while effective, introduce the burden of expensive noble metal catalysts and stringent requirements for removing trace heavy metals from the final active pharmaceutical ingredient. These factors collectively result in prolonged processing times, increased waste generation, and inflated production costs, creating a substantial barrier to the commercial scale-up of complex heterocycles.
The Novel Approach
In stark contrast, the methodology outlined in the patent data introduces a streamlined, four-step sequence that circumvents these historical inefficiencies through a clever combination of protection strategies and copper catalysis. The process initiates with the regioselective bromination of substituted anilines, followed by a transient protection of the amine functionality to direct subsequent reactivity. The core innovation lies in the copper-catalyzed coupling of the protected bromoaniline with diethyl malonate, facilitated by a CuI/L-Proline/Cs2CO3 system in polar aprotic solvents. This approach operates at significantly milder temperatures, typically between 0°C and 60°C, thereby preserving sensitive functional groups and minimizing thermal degradation. The final intramolecular cyclization and deprotection occur seamlessly, yielding the target oxindole with exceptional purity after a simple recrystallization. This holistic view of the synthesis is best visualized in the reaction scheme below, which maps the transformation from starting material to the final oxidized indole target.
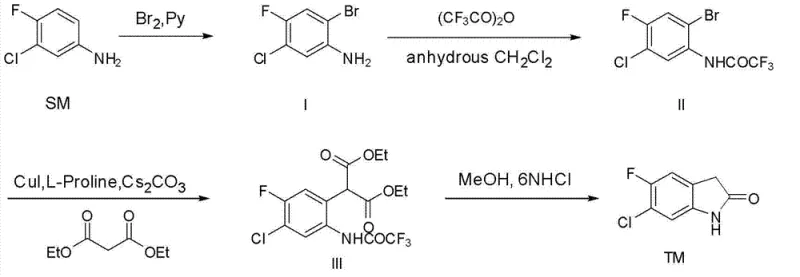
Mechanistic Insights into CuI/L-Proline Catalyzed Arylation
The success of this synthetic route hinges on the precise orchestration of the Ullmann-type coupling reaction, where the choice of ligand and base plays a pivotal role in determining both yield and selectivity. The mechanism likely proceeds through the formation of a soluble copper-ligand complex, where L-Proline acts as a bidentate ligand to stabilize the copper center and facilitate the oxidative addition of the aryl bromide. The presence of cesium carbonate serves a dual purpose: it acts as a base to deprotonate the beta-dicarbonyl compound, generating the reactive enolate nucleophile, and it helps to scavenge the hydrogen bromide byproduct, driving the equilibrium forward. This specific catalytic system is notable for its tolerance to various substituents, including the fluoro and chloro groups present on the aromatic ring, which are often sensitive to harsher nucleophilic aromatic substitution conditions. The detailed reaction conditions for this critical coupling and subsequent cyclization step are illustrated in the following diagram, highlighting the specific reagents and solvent systems employed.
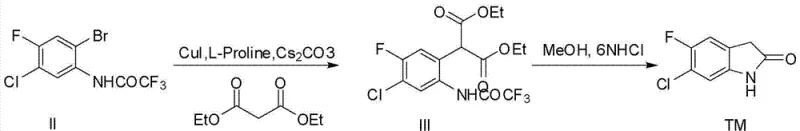
From an impurity control perspective, the mechanistic pathway offers inherent advantages by minimizing side reactions such as homocoupling of the aryl halide or over-alkylation of the malonate. The use of a transient trifluoroacetyl protecting group on the aniline nitrogen is a strategic masterstroke; it prevents the nitrogen from competing as a nucleophile during the copper-catalyzed step, ensuring that the carbon-carbon bond formation occurs exclusively at the desired position. Subsequent removal of this protecting group under acidic conditions (6N HCl in methanol) triggers the spontaneous intramolecular cyclization, effectively locking the structure into the thermodynamically stable oxindole ring system. This tandem deprotection-cyclization sequence eliminates the need for isolating unstable intermediates, thereby reducing material loss and exposure to potential contaminants, ultimately contributing to the high HPLC purity of >99% reported in the experimental data.
How to Synthesize 5-Chloro-4-fluoro-1,3-dihydro-2H-indol-2-one Efficiently
For process chemists and R&D teams looking to replicate or adapt this technology, the patent provides a clear roadmap that balances reaction efficiency with operational simplicity. The synthesis begins with the careful control of exothermic bromination, followed by a straightforward acylation, before moving to the critical copper-catalyzed coupling in DMSO. The protocol emphasizes the importance of maintaining anhydrous conditions during the coupling phase to prevent hydrolysis of the reactive intermediates, while the final workup utilizes standard extraction and recrystallization techniques familiar to any manufacturing facility. Detailed standardized operating procedures regarding stoichiometry, temperature ramps, and quenching protocols are essential for ensuring batch-to-batch consistency. For the complete, step-by-step technical execution of this synthesis, please refer to the structured guide below.
- Brominate 3-chloro-4-fluoroaniline using Br2 and pyridine in DCM at 0-5°C to form the o-bromoaniline intermediate.
- Protect the amine group with trifluoroacetic anhydride in anhydrous DCM to prevent side reactions during coupling.
- Perform CuI/L-Proline catalyzed coupling with diethyl malonate in DMSO, followed by acid-mediated cyclization and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
Beyond the technical elegance of the chemistry, this synthetic route offers profound economic and logistical benefits that resonate deeply with procurement managers and supply chain directors. The shift from precious metal catalysts to earth-abundant copper represents a fundamental change in the cost structure of the molecule, removing the volatility associated with palladium pricing and the regulatory burden of heavy metal clearance. Furthermore, the reliance on commodity chemicals such as diethyl malonate, cesium carbonate, and substituted anilines ensures a robust supply chain that is less susceptible to geopolitical disruptions or raw material shortages. The simplification of the purification process—replacing column chromatography with a single recrystallization from ethyl acetate—drastically reduces solvent consumption and processing time, directly translating to lower manufacturing overheads and a smaller environmental footprint.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the use of inexpensive ligands like L-Proline significantly lower the direct material costs per kilogram of product. Additionally, the ability to purify the final product via simple recrystallization rather than complex chromatographic separation reduces solvent usage and labor hours, leading to substantial overall cost savings in the production budget.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials and avoiding specialized reagents that may have long lead times, this process mitigates the risk of supply interruptions. The mild reaction conditions also reduce the strain on equipment and utilities, allowing for more flexible scheduling and faster turnaround times, which is critical for reducing lead time for high-purity intermediates in a just-in-time manufacturing environment.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing solvents and conditions that are easily managed in large-scale reactors without requiring exotic high-pressure or cryogenic equipment. The reduced generation of hazardous waste and the avoidance of toxic heavy metals align with increasingly stringent global environmental regulations, facilitating smoother regulatory approvals and sustainable operations.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patented technology, we have compiled a set of frequently asked questions based on the specific data points and claims found within the patent documentation. These answers address common concerns regarding reaction safety, impurity profiles, and the adaptability of the method to different substrates. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this route into their existing production portfolios or for those seeking a high-purity oxindole compound for their drug development programs.
Q: What are the advantages of this copper-catalyzed method over classical indole synthesis?
A: Unlike classical methods like Larock or Fischer synthesis which often require toxic heavy metals or harsh acidic conditions, this patent utilizes a mild CuI/L-Proline system. It avoids expensive palladium catalysts and simplifies purification to a single recrystallization step, significantly reducing environmental impact and processing time.
Q: How is product purity controlled in this synthetic route?
A: The process achieves high purity (over 99%) through a combination of selective catalytic coupling and a final recrystallization step using ethyl acetate. The use of specific additives like L-Proline and Cs2CO3 minimizes byproduct formation during the arylation stage.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights industrial suitability due to the use of cheap, commercially available raw materials, mild reaction temperatures (0-60°C), and the elimination of complex chromatographic purification, making it ideal for multi-kilogram to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Chloro-4-fluoro-1,3-dihydro-2H-indol-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in patent CN102875444A are fully realized at the plant level. Our facilities are equipped with state-of-the-art reactor trains capable of handling the specific solvent systems and temperature profiles required for this copper-catalyzed process, all while adhering to stringent purity specifications and maintaining comprehensive documentation for regulatory submissions. Our rigorous QC labs utilize advanced analytical techniques to monitor every critical parameter, guaranteeing that every batch of oxindole intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite you to leverage our technical expertise to optimize your supply chain and accelerate your project timelines. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, helping you identify further opportunities for efficiency within this synthetic route. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our manufacturing capabilities can support your long-term strategic goals for oxindole-based therapeutics.
