Advanced Synthesis of 3,4-Dihydro-2H-quinolizin-2-one Compounds for Commercial API Manufacturing
Introduction to Novel Quinolizinone Scaffold Synthesis
The pharmaceutical industry continuously seeks robust methodologies for constructing nitrogen-containing heterocycles, which serve as privileged scaffolds in a vast array of bioactive molecules. Patent CN114478516B introduces a groundbreaking approach to synthesizing 3,4-dihydro-2H-quinolizin-2-one compounds, a structural motif that has historically presented significant synthetic challenges due to the complexity of forming the fused bicyclic system with high regioselectivity. This innovation leverages an intramolecular electrocyclization dearomatization strategy, transforming readily available 2-pyridine acetate derivatives into complex quinolizinone cores under remarkably mild conditions. The significance of this development lies not only in the novelty of the chemical transformation but also in its potential to streamline the supply chain for high-purity pharmaceutical intermediates. By enabling the construction of these valuable heterocycles at room temperature, the technology reduces energy consumption and eliminates the need for specialized high-pressure equipment, positioning it as a highly attractive route for commercial scale-up.
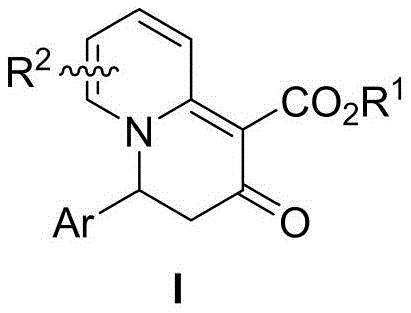
Furthermore, the structural diversity accessible through this method is exceptional, accommodating a wide range of functional groups such as nitro, halogen, and ester moieties without compromising the integrity of the cyclization process. This flexibility is crucial for medicinal chemists aiming to explore structure-activity relationships (SAR) efficiently. The ability to introduce electron-withdrawing groups like nitro or trifluoromethyl directly onto the scaffold opens new avenues for downstream derivatization, allowing for the rapid generation of compound libraries for drug screening. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patent-protected methodologies is essential for offering clients cutting-edge solutions that balance innovation with manufacturability. The following sections will dissect the technical merits and commercial implications of this synthesis in detail.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of quinolizinone derivatives has relied on multi-step sequences involving harsh reaction conditions, such as high temperatures, strong acids, or the use of stoichiometric amounts of toxic reagents. Conventional routes often struggle with poor atom economy and the formation of complex impurity profiles that are difficult to separate, leading to reduced overall yields and increased production costs. Many existing methods require the pre-functionalization of starting materials with sensitive protecting groups, adding unnecessary steps and waste to the process. Additionally, the reliance on transition metal catalysts in some traditional cyclizations introduces the risk of heavy metal residues, which is a critical quality attribute (CQA) concern for API manufacturing. These limitations create bottlenecks in the supply chain, extending lead times and increasing the cost of goods sold (COGS) for final drug products. The inability to perform these transformations under mild conditions also restricts the scope of compatible substrates, limiting the chemical space available for exploration.
The Novel Approach
In stark contrast, the methodology disclosed in CN114478516B offers a streamlined, one-pot solution that operates at room temperature, effectively bypassing the thermal and chemical stressors associated with legacy techniques. The core of this innovation is the utilization of 2-pyridine acetate derivatives that undergo a spontaneous or mildly catalyzed intramolecular cyclization to form the target quinolizinone skeleton. This approach eliminates the need for expensive transition metal catalysts in many instances, relying instead on the unique properties of fluorinated alcohol solvents like hexafluoroisopropanol (HFIP) to promote the reaction. The mildness of the conditions ensures that sensitive functional groups remain intact, thereby expanding the substrate scope to include molecules that would decompose under traditional conditions. This simplicity translates directly into operational efficiency, as the work-up procedure is straightforward, typically involving solvent removal and standard column chromatography. For procurement teams, this represents a significant opportunity for cost reduction in API manufacturing by minimizing raw material complexity and waste disposal costs.
Mechanistic Insights into Intramolecular Electrocyclization Dearomatization
The chemical elegance of this transformation lies in its mechanism, which involves a concerted electrocyclic ring closure followed by a rearomatization or tautomerization step to establish the stable quinolizinone system. The reaction initiates with the nucleophilic attack or orbital overlap facilitated by the specific conformation of the 2-pyridine acetate derivative, driven by the electronic properties of the substituents. The use of hexafluoroisopropanol (HFIP) as a solvent plays a pivotal role; its high ionizing power and hydrogen-bond donating ability stabilize the transition state and intermediate species, lowering the activation energy barrier sufficiently to allow the reaction to proceed at ambient temperature. This solvent effect is critical for achieving the observed yields without the need for external heating. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as concentration and substituent electronics, to optimize outcomes for specific derivatives. The robustness of this mechanistic pathway ensures consistent product quality, a key requirement for regulatory compliance in pharmaceutical production.
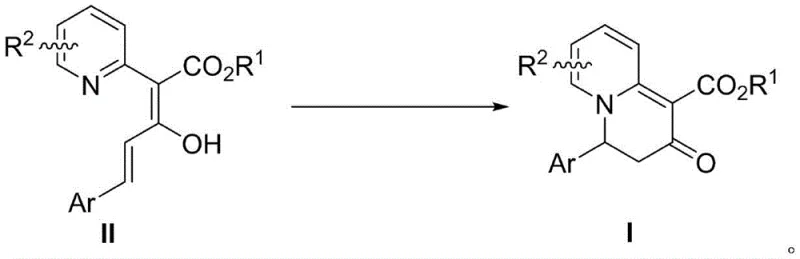
Regarding impurity control, the mild nature of this room-temperature cyclization inherently suppresses the formation of thermal degradation products and polymerization by-products that often plague high-temperature reactions. The selectivity of the electrocyclization ensures that the primary impurity profile is manageable, typically consisting of unreacted starting material or minor regioisomers that are easily removed during purification. The patent data indicates that even with diverse substituents like nitro groups or halogens, the reaction maintains high fidelity, producing the desired scaffold with minimal side reactions. This high level of chemoselectivity is particularly advantageous when scaling up, as it reduces the burden on downstream purification units. For R&D directors, this means a cleaner crude product and a more predictable process validation trajectory. The ability to control the reaction outcome through solvent choice and subtle electronic tuning of the substrate provides a powerful tool for designing efficient synthetic routes for complex drug candidates.
How to Synthesize 3,4-Dihydro-2H-quinolizin-2-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to solvent quality and reaction monitoring, although the operational steps are fundamentally simple. The process begins by dissolving the specific 2-pyridine acetate derivative precursor in hexafluoroisopropanol to achieve a molar concentration of approximately 0.1 mol/L, ensuring optimal molecular interactions for cyclization. While the reaction can proceed without a catalyst, the addition of mild acids like p-toluenesulfonic acid can be employed to accelerate the rate if necessary, though the patent highlights that catalyst-free conditions often yield superior results in terms of purity. The mixture is then stirred at room temperature for an extended period, typically around 7 days, allowing the slow but thermodynamically favorable cyclization to reach completion. Reaction progress is conveniently monitored using thin-layer chromatography (TLC), providing real-time visibility into conversion rates. Upon completion, the solvent is removed under reduced pressure, and the crude residue is purified via column chromatography using standard eluent systems such as petroleum ether and ethyl acetate. The detailed standardized synthesis steps see the guide below.
- Dissolve the 2-pyridine acetate derivative substrate in hexafluoroisopropanol (HFIP) to achieve a concentration of approximately 0.1 mol/L.
- Optionally add a mild catalyst such as p-toluenesulfonic acid or scandium triflate, though the reaction proceeds efficiently without catalysts in optimized solvents.
- Stir the reaction mixture at room temperature for 7 days, monitor via TLC, and purify the resulting solid using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis technology offers compelling advantages that directly address the pain points of modern pharmaceutical supply chains, particularly regarding cost efficiency and operational simplicity. The elimination of expensive transition metal catalysts and the ability to run reactions at room temperature significantly lower the energy footprint and raw material costs associated with production. This aligns perfectly with the industry's push towards greener chemistry and sustainable manufacturing practices. Furthermore, the high tolerance for functional groups means that a single platform technology can be used to generate a wide library of intermediates, reducing the need for developing unique processes for each new candidate. This versatility enhances supply chain reliability by minimizing the number of specialized reagents required and simplifying inventory management. For procurement managers, the ability to source high-quality intermediates produced via such a robust and scalable method ensures a steady flow of materials for downstream API synthesis, mitigating the risk of production delays caused by complex or unstable synthetic routes.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of costly catalytic systems and the reduction of energy consumption due to ambient temperature operation. By avoiding high-pressure reactors and extensive heating cycles, the capital expenditure (CAPEX) and operational expenditure (OPEX) for manufacturing facilities are substantially reduced. Additionally, the high yields reported in the patent, reaching up to 84% for certain derivatives, indicate excellent atom economy and minimal waste generation. This efficiency translates to lower raw material usage per kilogram of product, directly impacting the bottom line. The simplified work-up procedure further reduces labor costs and solvent usage during purification. Consequently, adopting this method can lead to significant cost savings in API manufacturing, making it an economically viable option for both generic and innovative drug production.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to a more resilient supply chain by reducing dependency on scarce or volatile reagents. Since the reaction tolerates a wide range of substituents and conditions, the risk of batch failure due to minor variations in raw material quality is minimized. The use of common organic solvents and standard purification techniques ensures that the process can be easily transferred between different manufacturing sites without significant re-validation efforts. This flexibility is crucial for maintaining continuity of supply in a global market where logistical disruptions are common. Moreover, the scalability of the reaction from milligram to multi-kilogram scales has been demonstrated, assuring partners that commercial quantities can be met without compromising quality. This reliability makes the technology a strategic asset for securing long-term supply agreements for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process is straightforward due to the absence of exothermic hazards associated with high-temperature reactions or reactive metal catalysts. The mild conditions allow for the use of standard glass-lined or stainless steel reactors, facilitating a smooth transition from pilot to commercial scale. From an environmental standpoint, the reduction in hazardous waste and energy usage supports compliance with increasingly stringent environmental regulations. The potential for solvent recycling, particularly for specialized solvents like HFIP, further enhances the sustainability profile of the process. This alignment with green chemistry principles not only reduces environmental impact but also improves the corporate social responsibility (CSR) metrics for manufacturing partners. Ultimately, the combination of scalability and environmental compliance positions this technology as a future-proof solution for the sustainable production of complex nitrogen heterocycles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived from the specific details and advantages outlined in the patent documentation. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating this method into their existing workflows. The answers provided are based on the empirical data and experimental results reported in the patent, ensuring accuracy and relevance for decision-making. These insights cover reaction conditions, substrate scope, and purification strategies, offering a comprehensive overview for technical teams. We encourage further discussion with our technical experts to explore how these solutions can be tailored to your specific project requirements.
Q: What are the key advantages of this synthesis method over traditional quinolizinone preparations?
A: This method utilizes a mild, room-temperature intramolecular electrocyclization that avoids harsh conditions and expensive transition metal catalysts, significantly simplifying purification and reducing heavy metal contamination risks.
Q: Can the substituents on the pyridine ring be varied for different biological activities?
A: Yes, the patent demonstrates high tolerance for various substituents including nitro, halogen, and alkyl groups on the pyridine ring, allowing for extensive SAR studies in drug discovery.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: The process operates at ambient temperature without pressure vessels and uses standard organic solvents, making it highly amenable to scale-up from gram to multi-kilogram batches with consistent yield profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dihydro-2H-quinolizin-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis methods described in CN114478516B for advancing drug discovery and development programs. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into reliable commercial supplies. Our facility is equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates. We understand the critical importance of consistency and quality in the supply of complex heterocyclic building blocks, and our team is committed to delivering products that exceed industry standards. By leveraging our expertise in process optimization and scale-up, we can help you navigate the complexities of bringing new quinolizinone-based therapeutics to market efficiently.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us to request specific COA data and route feasibility assessments for your target compounds. Together, we can accelerate your development timelines and secure a competitive advantage in the marketplace through superior supply chain solutions and technical excellence.
