Revolutionizing Cinacalcet Intermediate Production: A Technical Deep Dive into Patent CN111018734B
Revolutionizing Cinacalcet Intermediate Production: A Technical Deep Dive into Patent CN111018734B
The pharmaceutical industry continuously seeks robust, scalable, and cost-efficient synthetic pathways for critical active pharmaceutical ingredient (API) intermediates. Patent CN111018734B, published in late 2020, introduces a significant technological advancement in the preparation of (R)-N-(1-(naphthalen-1-yl)ethyl)-3-(3-(trifluoromethyl)phenyl)propionamide, a pivotal intermediate for the synthesis of Cinacalcet hydrochloride. This calcimimetic agent is essential for treating secondary hyperparathyroidism in chronic kidney disease patients. The disclosed methodology represents a paradigm shift from traditional transition-metal catalyzed processes to a more sustainable, organic-base mediated approach. By leveraging the unique reactivity of Meldrum's acid derivatives, this innovation addresses long-standing challenges regarding raw material availability, reaction safety, and downstream purification complexity. For R&D directors and procurement strategists, understanding the nuances of this patent is crucial for optimizing supply chains and ensuring the consistent delivery of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cinacalcet intermediates has been plagued by reliance on expensive reagents, hazardous conditions, and cumbersome purification steps. As illustrated in prior art Route 1, conventional methods often employ titanium tetraisopropoxide (Ti(OiPr)4) as a Lewis acid catalyst for condensation reactions.  This approach not only incurs high material costs due to the price of the titanium catalyst but also imposes stringent anhydrous requirements that complicate reactor operations. Furthermore, the subsequent reduction step typically utilizes sodium cyanoborohydride, a toxic reagent that generates hazardous waste streams requiring specialized disposal protocols. Perhaps most critically, many legacy routes fail to establish chirality early in the synthesis, necessitating expensive chiral chromatography for final resolution, which severely limits throughput and scalability. Other routes involving Heck couplings introduce palladium catalysts, leading to potential heavy metal contamination issues that require rigorous and costly removal processes to meet strict regulatory limits for API manufacturing.
This approach not only incurs high material costs due to the price of the titanium catalyst but also imposes stringent anhydrous requirements that complicate reactor operations. Furthermore, the subsequent reduction step typically utilizes sodium cyanoborohydride, a toxic reagent that generates hazardous waste streams requiring specialized disposal protocols. Perhaps most critically, many legacy routes fail to establish chirality early in the synthesis, necessitating expensive chiral chromatography for final resolution, which severely limits throughput and scalability. Other routes involving Heck couplings introduce palladium catalysts, leading to potential heavy metal contamination issues that require rigorous and costly removal processes to meet strict regulatory limits for API manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN111018734B offers a streamlined alternative that bypasses these bottlenecks. The core innovation lies in the utilization of 3-trifluoromethylbenzaldehyde and Meldrum's acid as primary building blocks. 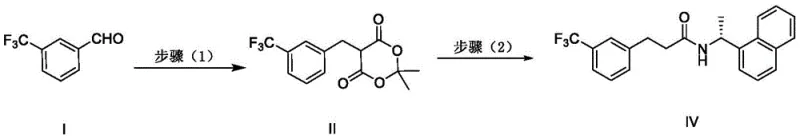 This novel pathway constructs the carbon skeleton through a condensation reaction that avoids the use of transition metals entirely. The process proceeds through a stable intermediate, 2,2-dimethyl-5-(3-(trifluoromethyl)benzyl)-1,3-dioxo-4,6-dione, which serves as an activated acyl equivalent. By reacting this intermediate directly with (R)-1-(1-naphthyl)ethanamine, the synthesis achieves both amide bond formation and decarboxylation in a unified sequence. This strategic design eliminates the need for separate activation steps like acid chloride formation, which are prone to instability and handling difficulties. The result is a significantly shortened synthetic sequence that enhances overall yield while drastically reducing the environmental footprint associated with heavy metal waste and solvent consumption.
This novel pathway constructs the carbon skeleton through a condensation reaction that avoids the use of transition metals entirely. The process proceeds through a stable intermediate, 2,2-dimethyl-5-(3-(trifluoromethyl)benzyl)-1,3-dioxo-4,6-dione, which serves as an activated acyl equivalent. By reacting this intermediate directly with (R)-1-(1-naphthyl)ethanamine, the synthesis achieves both amide bond formation and decarboxylation in a unified sequence. This strategic design eliminates the need for separate activation steps like acid chloride formation, which are prone to instability and handling difficulties. The result is a significantly shortened synthetic sequence that enhances overall yield while drastically reducing the environmental footprint associated with heavy metal waste and solvent consumption.
Mechanistic Insights into Meldrum's Acid Mediated Amidation
The chemical elegance of this process stems from the unique electronic properties of the Meldrum's acid moiety. In the initial step, the aldehyde undergoes a condensation with Meldrum's acid, potentially facilitated by organocatalysts like proline or simple bases, to form a benzylidene derivative which is subsequently reduced or directly converted to the benzyl-substituted dione. The high acidity of the methylene protons in Meldrum's acid facilitates nucleophilic attacks, allowing for efficient carbon-carbon bond formation under mild conditions. When this intermediate encounters the chiral amine, the nucleophilic nitrogen attacks one of the carbonyl carbons of the dione ring. This attack triggers a cascade where the ring opens and subsequently undergoes thermal decarboxylation. This decarboxylation acts as a thermodynamic driving force, pushing the equilibrium towards the desired amide product and preventing reverse reactions. This mechanism ensures high conversion rates without the need for excessive coupling agents or activating reagents that typically generate stoichiometric amounts of salt byproducts.
From an impurity control perspective, this mechanism offers distinct advantages over reductive amination routes. Traditional reductive aminations can suffer from over-alkylation or incomplete reduction, leading to secondary amine impurities that are structurally similar to the product and difficult to separate. In the Meldrum's acid route, the stoichiometry is tightly controlled by the ring-opening event; once the amide bond is formed and CO2 is lost, the molecule is chemically locked into the propionamide structure. There is no risk of over-alkylation since the electrophile is consumed in the ring-opening process. Furthermore, the byproducts of the reaction, primarily acetone and carbon dioxide, are volatile and easily removed during workup, leaving behind a crude product of exceptional purity. This inherent selectivity reduces the burden on downstream purification units, allowing for simpler crystallization or filtration steps to achieve the required specification for API synthesis.
How to Synthesize (R)-N-(1-(Naphthalen-1-yl)ethyl)-3-(3-(Trifluoromethyl)phenyl)propionamide Efficiently
The practical implementation of this synthesis involves a straightforward two-stage protocol that is amenable to standard glass-lined or stainless steel reactors. The first stage focuses on the construction of the functionalized side chain using the Meldrum's acid scaffold, while the second stage couples this fragment with the chiral amine. The detailed standardized synthesis steps are provided in the guide below.
- Condense 3-trifluoromethylbenzaldehyde with Meldrum's acid under catalytic conditions to form the protected benzyl-Meldrum's acid derivative.
- React the resulting intermediate with (R)-1-(1-naphthyl)ethanamine in a solvent system to effect amidation and simultaneous decarboxylation.
- Isolate the final high-purity amide product through simple filtration or extraction workups, avoiding complex chromatographic separation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the substantial reduction of raw material volatility. By shifting away from specialized catalysts like titanium tetraisopropoxide or palladium complexes, which are subject to geopolitical supply constraints and price fluctuations, manufacturers can secure a more stable cost base. The starting materials, specifically 3-trifluoromethylbenzaldehyde and Meldrum's acid, are commodity chemicals produced by multiple global suppliers, ensuring competitive pricing and reliable availability. This diversification of the supply base mitigates the risk of production stoppages due to single-source vendor failures.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and chiral separation columns directly translates to lower variable costs per kilogram. Without the need for precious metal scavengers or high-pressure hydrogenation equipment, capital expenditure for plant setup is also minimized. The simplified workup procedures, often requiring only filtration and washing rather than complex distillations or chromatography, reduce utility consumption and labor hours. These cumulative efficiencies allow for a significantly leaner cost structure, enabling more competitive pricing strategies in the generic pharmaceutical market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to higher batch success rates and consistent cycle times. Unlike moisture-sensitive reactions that demand rigorous drying of solvents and inert atmospheres, this process tolerates a broader range of operational parameters. This flexibility reduces the likelihood of batch failures due to minor deviations in humidity or temperature, ensuring that delivery schedules are met with greater predictability. Furthermore, the avoidance of carcinogenic reagents like ethyl acrylate simplifies regulatory compliance and logistics, removing the need for specialized hazardous material handling and storage facilities.
- Scalability and Environmental Compliance: As production scales from pilot batches to multi-ton commercial runs, the safety profile of this process becomes increasingly valuable. The absence of high-pressure hydrogenation steps and toxic cyanide-based reducing agents aligns with modern green chemistry principles. Waste streams are less hazardous, lowering disposal costs and simplifying environmental permitting. The high atom economy of the decarboxylative amidation step ensures that a larger proportion of input mass ends up in the final product, minimizing waste generation. This sustainability profile is increasingly important for multinational corporations aiming to meet corporate social responsibility goals and reduce their overall carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide clarity for potential partners.
Q: How does this new synthesis route improve upon conventional titanium-catalyzed methods?
A: Unlike conventional routes relying on expensive and moisture-sensitive titanium tetraisopropoxide, this method utilizes readily available Meldrum's acid and avoids heavy metal catalysts, significantly simplifying post-processing and reducing raw material costs.
Q: What are the primary impurity concerns addressed by this patent technology?
A: The process avoids the formation of difficult-to-remove byproducts associated with reductive amination using cyanoborohydride and eliminates the need for chiral column separation, ensuring a cleaner impurity profile suitable for API manufacturing.
Q: Is this synthetic route scalable for industrial production?
A: Yes, the method features mild reaction conditions, inexpensive starting materials like 3-trifluoromethylbenzaldehyde, and simple isolation steps such as filtration, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinacalcet Intermediate Supplier
The technological potential of Patent CN111018734B underscores the importance of partnering with a manufacturer who possesses both the chemical expertise and the infrastructure to execute complex syntheses at scale. NINGBO INNO PHARMCHEM stands at the forefront of this capability, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling the specific thermal and mixing requirements of condensation and decarboxylation reactions. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques such as HPLC and chiral GC to ensure every batch meets the exacting standards required for API manufacturing. Our commitment to quality assurance guarantees that the intermediates supplied are free from critical impurities, facilitating a smooth regulatory filing process for our clients.
We invite global pharmaceutical companies and contract manufacturing organizations to collaborate with us on optimizing their cinacalcet supply chains. By leveraging our technical proficiency in this novel Meldrum's acid route, we can help you achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term production goals and enhance your market competitiveness.
