Advanced Enzymatic Synthesis of Chloramphenicol: Scalable Solutions for Global Pharmaceutical Supply Chains
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of essential antibiotics, and the technological landscape has shifted significantly with the introduction of advanced biocatalytic methods. Patent CN106566851B details a groundbreaking preparation method for Chloramphenicol compounds that leverages the specificity of ketoreductase enzymes to overcome longstanding inefficiencies in traditional chemical synthesis. This innovation represents a pivotal move away from harsh chemical reagents towards environmentally benign biological catalysts, offering a robust solution for manufacturers aiming to optimize their production lines. By utilizing a buffer solution system combined with specific enzymatic activity, this method achieves exceptional stereocontrol and conversion rates that were previously difficult to attain on an industrial scale. For global stakeholders, this patent data underscores a critical opportunity to enhance the reliability of Chloramphenicol supply chains while simultaneously addressing increasing regulatory pressures regarding environmental sustainability and waste management in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Chloramphenicol has relied heavily on multi-step chemical syntheses that suffer from inherent thermodynamic and economic inefficiencies. One prevalent traditional route involves the chiral resolution of racemates, a process that is fundamentally limited by a theoretical maximum yield of only 50 percent because the unwanted enantiomer must be discarded or recycled through energy-intensive processes. Furthermore, conventional chemical reduction methods often utilize aluminum isopropoxide systems which generate substantial amounts of difficult-to-treat solid waste, creating significant environmental burdens and increasing disposal costs for manufacturers. These legacy processes also frequently require harsh reaction conditions, including ultra-low temperatures or the use of expensive chiral catalysts like copper trifluoromethanesulfonate, which drive up the overall cost of goods and complicate the engineering requirements for safe operation. The accumulation of these factors results in a manufacturing profile that is increasingly untenable in a modern market focused on green chemistry and cost-effective production of high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a highly specific ketoreductase (KRED) to catalyze the asymmetric reduction of chloramphenicol intermediates directly within a mild aqueous buffer system. This biocatalytic strategy effectively bypasses the need for chiral resolution by dynamically converting the substrate into the desired stereoisomer with exceptional precision, thereby theoretically enabling yields that approach 100 percent rather than being capped at 50 percent. The reaction proceeds under温和 conditions, typically between 25-35°C, which drastically reduces energy consumption associated with heating or cooling large-scale reactors and minimizes the risk of thermal degradation of sensitive intermediates. By replacing toxic heavy metal catalysts and volatile organic solvents with enzymes and water-based buffers, this method not only simplifies the downstream purification process but also aligns with stringent global environmental regulations, offering a clear pathway for cost reduction in antibiotic manufacturing without compromising on product quality or safety standards.
Mechanistic Insights into Ketoreductase-Catalyzed Asymmetric Reduction
The core of this technological advancement lies in the precise mechanistic action of the ketoreductase enzyme, which facilitates a dynamic kinetic resolution during the reduction of the ketone group on the chloramphenicol intermediate. Unlike chemical reductants that may attack the carbonyl group from multiple angles leading to a mixture of stereoisomers, the enzyme's active site imposes strict steric constraints that favor the formation of the (R,R) configuration exclusively. This high level of stereocontrol is critical because Chloramphenicol's biological activity is strictly dependent on its specific three-dimensional structure, and the presence of other isomers such as (S,S) or (R,S) can act as impurities that complicate purification and potentially affect safety profiles. The patent data indicates that by carefully selecting the specific ketoreductase variant and optimizing the cofactor regeneration system, manufacturers can achieve an enantiomeric excess (Ee) and diastereomeric ratio (Dr) both exceeding 99 percent, ensuring that the final product meets the rigorous purity specifications required by international pharmacopoeias.
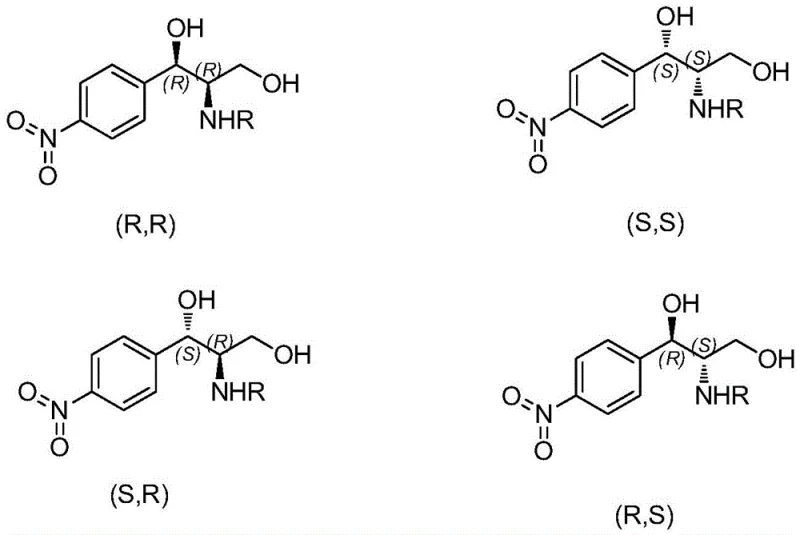
Furthermore, the impurity control mechanism inherent in this enzymatic process is superior to chemical methods because it prevents the formation of byproducts at the source rather than relying on post-reaction filtration. In traditional chemical synthesis, side reactions often generate structurally similar impurities that are chemically difficult to separate from the target molecule, requiring multiple crystallization steps that reduce overall yield. The enzymatic route, however, operates with such high specificity that the reaction mixture primarily contains the desired product and benign byproducts like gluconic acid from the cofactor regeneration system. This simplifies the workup procedure significantly, often allowing for direct isolation of the product through simple extraction or crystallization, which in turn reduces the solvent usage and processing time. For R&D directors, this mechanistic advantage translates to a more robust and predictable process that is easier to validate and scale, reducing the technical risks associated with technology transfer from the laboratory to commercial production facilities.
How to Synthesize Chloramphenicol Efficiently
The implementation of this synthesis route involves a streamlined sequence of operations that begins with the preparation of a buffered aqueous solution containing the chloramphenicol intermediate and a cofactor source such as glucose. Detailed standardized synthetic steps see the guide below.
- Prepare a buffered aqueous solution containing the chloramphenicol intermediate and a cofactor regeneration system such as glucose.
- Introduce specific ketoreductase enzymes and necessary dehydrogenases to the mixture under controlled pH and temperature conditions.
- Monitor the stereoselective reduction until completion, followed by hydrolysis and dichloroacetylation to finalize the active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this enzymatic technology offers profound strategic advantages that extend beyond mere technical feasibility into the realm of significant operational efficiency and risk mitigation. The elimination of chiral resolution steps fundamentally alters the material balance of the production process, meaning that less raw starting material is required to produce the same amount of active pharmaceutical ingredient, which directly correlates to substantial cost savings in raw material procurement. Additionally, the shift to aqueous-based reactions reduces the dependency on volatile organic solvents, which are subject to fluctuating market prices and increasingly strict transportation and storage regulations, thereby enhancing the stability and predictability of the supply chain. The mild reaction conditions also imply lower energy costs and reduced wear and tear on manufacturing equipment, contributing to a lower total cost of ownership for the production facility while ensuring a more consistent and reliable Chloramphenicol supplier capability for long-term contracts.
- Cost Reduction in Manufacturing: The transition from chemical resolution to enzymatic dynamic kinetic resolution removes the inherent 50 percent yield loss associated with discarding unwanted isomers, effectively doubling the theoretical output from the same mass of starting material. This dramatic improvement in atom economy means that the cost per kilogram of the final API is significantly reduced, as the expense of the starting intermediate is amortized over a much larger quantity of saleable product. Furthermore, the removal of expensive transition metal catalysts and the reduction in solvent consumption lower the variable costs of production, allowing for more competitive pricing strategies in the global market without sacrificing margin. These efficiencies collectively drive down the manufacturing cost base, providing a strong buffer against raw material price volatility and enabling more aggressive bidding for large-scale supply tenders.
- Enhanced Supply Chain Reliability: The simplicity of the enzymatic process, which operates at ambient temperatures and pressures, reduces the complexity of the manufacturing infrastructure required, thereby minimizing the risk of unplanned downtime due to equipment failure or utility fluctuations. Because the process does not rely on scarce or geographically concentrated specialty chemicals like chiral metal catalysts, the supply chain is more resilient to disruptions in the upstream chemical market. The use of robust enzymes and common buffer components ensures that production can be maintained consistently across different manufacturing sites, facilitating a diversified supply strategy that protects against regional instabilities. This reliability is crucial for maintaining continuous availability of high-purity antibiotics, ensuring that downstream formulation partners can meet their own production schedules without interruption.
- Scalability and Environmental Compliance: Scaling this biocatalytic process is inherently safer and more straightforward than scaling exothermic chemical reductions, as the heat generation is manageable and the reaction medium is non-flammable water. This ease of scale-up allows manufacturers to respond quickly to surges in demand, such as during public health emergencies, without the need for extensive re-engineering of the production line. Moreover, the reduction in hazardous waste generation simplifies compliance with environmental regulations, reducing the administrative burden and potential fines associated with waste disposal. This environmental stewardship enhances the corporate reputation of the manufacturer and aligns with the sustainability goals of major pharmaceutical buyers, making the supply partnership more attractive and durable in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis technology, derived directly from the patent specifications and industry best practices. These answers provide clarity on how this method compares to existing standards and what stakeholders can expect regarding performance and integration. Understanding these details is essential for making informed decisions about adopting this advanced manufacturing route for Chloramphenicol production.
Q: How does the enzymatic route improve atom economy compared to chemical resolution?
A: Traditional chemical resolution theoretically limits yield to 50% due to the discarding of unwanted isomers. The enzymatic method described in CN106566851B utilizes dynamic kinetic resolution, allowing for conversion rates exceeding 99% and significantly reducing raw material waste.
Q: What are the stereochemical advantages of using ketoreductase in this synthesis?
A: The specific ketoreductase enzymes exhibit high stereoselectivity, preferentially producing the desired (R,R) configuration. This results in an enantiomeric excess (Ee) and diastereomeric ratio (Dr) both exceeding 99%, ensuring high-purity Chloramphenicol without complex purification steps.
Q: Is this biocatalytic process suitable for large-scale industrial manufacturing?
A: Yes, the process operates under mild conditions (25-35°C) in aqueous buffer systems, eliminating the need for hazardous organic solvents and extreme temperatures. This simplifies reactor requirements and waste treatment, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chloramphenicol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis routes described in recent patent literature and have integrated similar advanced biocatalytic capabilities into our CDMO service offerings. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Chloramphenicol or its intermediates meets the highest global standards for safety and efficacy. Our commitment to technological excellence allows us to offer partners a secure and high-quality source for critical antibiotic ingredients, leveraging the latest advancements in green chemistry to deliver superior value.
We invite you to engage with our technical procurement team to discuss how we can tailor this enzymatic technology to your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into how switching to our optimized manufacturing processes can reduce your overall procurement costs. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate our capability to support your long-term production goals with reliability and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
