Advanced Pd-Catalyzed Synthesis of Fluorinated Nitrogen Heterocycles for Commercial Scale-Up
Introduction to Next-Generation Fluorination Technology
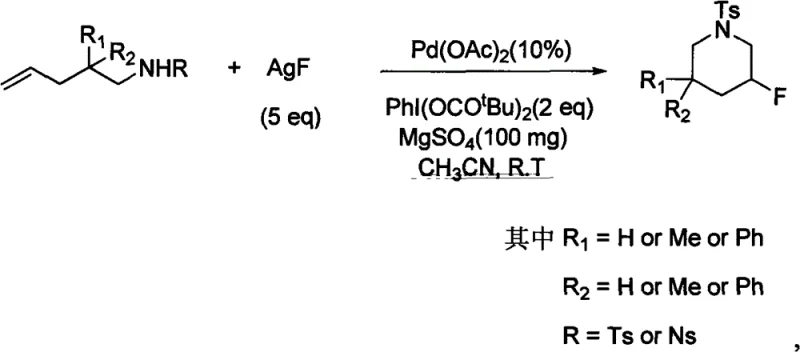
The strategic incorporation of fluorine atoms into organic molecules has become a cornerstone of modern medicinal chemistry, profoundly influencing the metabolic stability, lipophilicity, and bioavailability of active pharmaceutical ingredients (APIs). Patent CN101691319B introduces a groundbreaking methodology for synthesizing fluorinated nitrogen-containing heterocyclic compounds, specifically targeting the efficient construction of fluorinated six-membered rings such as piperidines. This technology represents a significant leap forward from traditional approaches by leveraging a palladium-catalyzed olefin aminofluorination strategy. Unlike conventional methods that often rely on pre-functionalized substrates or harsh fluorinating agents, this invention utilizes simple nitrogen-containing olefins as starting materials, employing a hypervalent iodine compound as an oxidant and an inorganic fluoride salt as the fluorine source. For R&D directors and process chemists seeking reliable pharmaceutical intermediate supplier partnerships, understanding this mechanistic shift is crucial for developing robust, scalable, and cost-effective synthetic routes for next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated nitrogen heterocycles has been fraught with significant challenges, primarily revolving around the limitations of classical fluorination reagents. Traditional protocols frequently employ reagents such as DAST (Diethylaminosulfur trifluoride) to convert alcohol precursors into their corresponding fluorides. However, these classical reactions suffer from inherent selectivity issues, often leading to elimination byproducts or rearrangement side reactions that compromise the overall purity of the final API intermediate. Furthermore, reagents like DAST are thermally unstable and potentially explosive, necessitating stringent safety protocols, specialized handling equipment, and cryogenic conditions to prevent runaway reactions. These factors not only escalate the operational costs but also introduce substantial supply chain vulnerabilities, as the procurement and storage of such hazardous materials require rigorous regulatory compliance. Additionally, the multi-step sequences often required to install the alcohol functionality prior to fluorination add unnecessary complexity, reducing the overall atom economy and generating excessive chemical waste, which is increasingly untenable in modern green chemistry mandates.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN101691319B offers a streamlined, direct difunctionalization pathway that constructs the heterocyclic ring and installs the fluorine atom simultaneously. This novel approach utilizes a palladium catalyst system in conjunction with silver fluoride (AgF) as a benign, inorganic fluorine source, effectively bypassing the need for dangerous covalent fluorinating agents. The reaction operates under remarkably mild conditions, typically at room temperature in acetonitrile solvent, which drastically reduces energy consumption and simplifies reactor requirements. By employing a hypervalent iodine oxidant, such as PhI(OCOtBu)2, the catalytic cycle is efficiently sustained, allowing for high turnover numbers and excellent yields. This method transforms simple, readily available allylamine derivatives directly into valuable fluorinated piperidine scaffolds with high regioselectivity. For procurement managers focused on cost reduction in API manufacturing, this shift eliminates the expensive and hazardous reagent costs associated with DAST while simplifying the workflow to a single-pot transformation.
Mechanistic Insights into Pd-Catalyzed Olefin Aminofluorination
The core of this technological advancement lies in the intricate palladium-catalyzed mechanism that facilitates the simultaneous formation of carbon-nitrogen and carbon-fluorine bonds. The catalytic cycle initiates with the activation of the olefin substrate by the palladium species, likely involving a coordination step that renders the double bond susceptible to nucleophilic attack. The presence of the hypervalent iodine oxidant plays a dual role: it serves as a terminal oxidant to regenerate the active palladium species and potentially assists in the generation of an electrophilic fluorine species from the inorganic silver fluoride salt. This in situ generation of reactive fluorine equivalents allows for the precise installation of the fluorine atom at the desired position on the heterocyclic ring without affecting other sensitive functional groups. The use of silver fluoride is particularly advantageous due to its solubility profile in organic nitrile solvents and its ability to act as a fluoride donor without the extreme basicity associated with alkali metal fluorides, thereby minimizing base-mediated decomposition pathways. This mechanistic elegance ensures that the reaction proceeds with high chemoselectivity, preserving the integrity of the sulfonamide protecting groups (Ts or Ns) and other substituents on the substrate.
Furthermore, the impurity profile of this reaction is exceptionally clean, which is a critical parameter for pharmaceutical manufacturing. The high selectivity observed in the patent examples suggests that competing pathways, such as beta-hydride elimination or non-specific radical fluorination, are effectively suppressed by the specific ligand environment and reaction conditions employed. The inclusion of drying agents like magnesium sulfate further enhances the reaction efficiency by sequestering trace water that could otherwise hydrolyze the hypervalent iodine oxidant or deactivate the catalyst. This level of control over the reaction environment translates directly into simplified downstream processing, as the crude reaction mixtures contain fewer byproducts that require difficult separation. For quality control teams, this means that achieving the stringent purity specifications required for GMP production becomes significantly more manageable, reducing the burden on analytical resources and accelerating the release of batches for clinical or commercial use.
How to Synthesize Fluorinated Piperidine Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and reaction monitoring to maximize yield and reproducibility. The patent outlines a robust protocol where the substrate olefin, palladium catalyst, oxidant, and fluoride source are combined in a specific molar ratio to ensure complete conversion. The process is designed to be operationally simple, avoiding the need for inert atmosphere gloveboxes or specialized high-pressure equipment, which makes it accessible for both laboratory-scale optimization and pilot plant operations. Detailed standard operating procedures for this transformation focus on the precise addition of reagents and the maintenance of ambient temperature to prevent thermal degradation of the sensitive intermediates. For process engineers looking to translate this chemistry to production, the following guide summarizes the critical operational parameters derived from the patent data.
- Prepare the reaction mixture by combining palladium acetate catalyst, hypervalent iodine oxidant (PhI(OCOtBu)2), and inorganic fluoride salt (AgF) in a nitrile solvent.
- Add the nitrogen-containing olefin substrate to the reaction vessel, optionally including a desiccant like magnesium sulfate to enhance yield.
- Stir the mixture at room temperature for 5 to 24 hours, then filter, concentrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this palladium-catalyzed fluorination method offers transformative benefits for supply chain resilience and manufacturing economics. The elimination of hazardous fluorinating reagents like DAST not only improves workplace safety but also removes the logistical complexities associated with transporting and storing Class 4.2 dangerous goods. This simplification of the raw material portfolio allows procurement teams to source stable, commodity-grade chemicals such as silver fluoride and palladium acetate from a broader range of qualified vendors, thereby mitigating the risk of supply disruptions caused by single-source dependencies. Moreover, the ability to conduct the reaction at room temperature significantly lowers the utility costs associated with heating or cooling large-scale reactors, contributing to a reduced carbon footprint and lower overall operating expenses. These factors collectively enhance the economic viability of producing fluorinated intermediates, making it a compelling choice for cost-sensitive generic drug programs as well as high-value novel therapeutics.
- Cost Reduction in Manufacturing: The substitution of expensive, specialized fluorinating agents with inexpensive inorganic salts like silver fluoride drives down the direct material costs per kilogram of product. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, improving the overall mass balance and reducing the cost of goods sold (COGS). The simplified workup procedure, which involves basic filtration and chromatography rather than complex distillation or crystallization steps to remove hazardous byproducts, further reduces labor and processing time. By streamlining the synthetic sequence to a single step from readily available olefins, manufacturers can achieve substantial cost savings compared to multi-step traditional routes that require isolation and purification of intermediate alcohols.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents ensures a consistent supply of critical inputs, shielding production schedules from the volatility often seen with specialty reagent markets. Silver fluoride and hypervalent iodine compounds are commercially available in bulk quantities, allowing for strategic stockpiling and long-term contracting without the degradation concerns associated with moisture-sensitive fluorinating agents. This stability extends to the starting materials as well, as the nitrogen-containing olefins can be synthesized in high volumes using established industrial processes. Consequently, supply chain managers can forecast demand more accurately and maintain leaner inventory levels without compromising production continuity, ensuring timely delivery of APIs to downstream formulation partners.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic volatile organic compounds (VOCs) associated with reagent decomposition make this process inherently safer for scale-up. Engineering controls for exotherm management are less demanding, allowing for the use of standard glass-lined or stainless steel reactors without the need for specialized Hastelloy equipment often required for corrosive fluorination chemistry. From an environmental standpoint, the generation of less hazardous waste streams simplifies effluent treatment and disposal, helping facilities meet increasingly strict environmental regulations. The potential for catalyst recovery and recycling further enhances the sustainability profile of the process, aligning with corporate social responsibility goals and reducing the environmental impact of pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic overview of the method's capabilities and limitations. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into their existing manufacturing pipelines. The answers reflect the consensus on the method's robustness, substrate tolerance, and operational simplicity.
Q: What are the primary safety advantages of this method compared to classical DAST fluorination?
A: This method utilizes inorganic fluoride salts like silver fluoride instead of hazardous reagents like DAST, significantly reducing toxicity risks and eliminating the need for cryogenic conditions often required for sensitive fluorinating agents.
Q: Does this catalytic system tolerate diverse substituents on the olefin substrate?
A: Yes, the patent demonstrates high tolerance for various substituents including phenyl, methyl, and different sulfonamide protecting groups (Ts, Ns), allowing for the synthesis of a broad library of fluorinated heterocyclic scaffolds.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: The reaction proceeds at room temperature with simple operation steps and uses readily available starting materials, making it highly amenable to commercial scale-up without the complex engineering controls needed for exothermic or hazardous fluorination processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Piperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced fluorination technologies play in accelerating drug discovery and development. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methodologies like the one described in CN101691319B can be successfully translated into robust manufacturing processes. We are committed to delivering high-purity fluorinated nitrogen heterocycles that meet the rigorous quality standards demanded by the global pharmaceutical industry. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch, providing our partners with the confidence they need to advance their clinical programs.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget without compromising quality. Please contact us today to request specific COA data for our fluorinated intermediates and to discuss route feasibility assessments for your target molecules. Let us be your trusted partner in navigating the complexities of fluorine chemistry and bringing your life-saving medicines to market faster and more economically.
