Advanced Catalytic Oxidation for High-Purity Vanillin Production and Commercial Scale-Up
Introduction to Green Catalytic Oxidation Technology
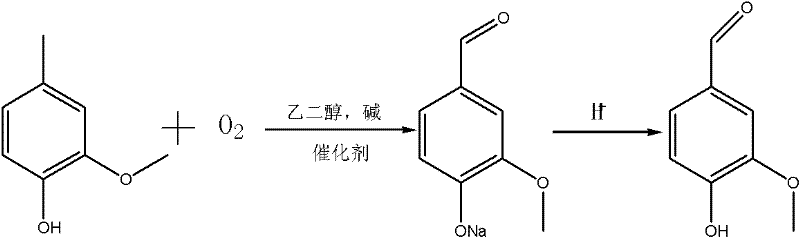
The global demand for high-purity vanillin, a critical ingredient in the food, cosmetic, and pharmaceutical sectors, continues to drive innovation in synthetic methodologies. Patent CN102381950A introduces a transformative approach to vanillin production, addressing the longstanding inefficiencies of traditional p-cresol oxidation routes. This technology leverages salt-group modified Schiff base metal chelates to catalyze the direct oxidation of 4-methylguaiacol, achieving complete substrate conversion while maintaining exceptional selectivity. Unlike conventional methods that rely on homogeneous transition metal salts which are notoriously difficult to separate from the product stream, this novel catalyst system offers a heterogeneous-like behavior that facilitates easy recovery and reuse. By shifting the solvent system from hazardous methanol to safer ethylene glycol mixtures, the process not only enhances operational safety but also aligns with modern green chemistry principles, providing a robust foundation for reliable vanillin supplier operations seeking to minimize environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of vanillin via the p-cresol route has been plagued by significant technical and safety bottlenecks, primarily centered around the oxidation step of 4-methylguaiacol. Conventional protocols typically employ simple transition metal salts, such as cobalt or copper salts, dissolved in strong alkaline methanol solutions. While these systems can achieve moderate conversion rates around 90%, they suffer from severe downstream processing challenges; the homogeneous nature of the catalyst makes separation from the final product arduous, often requiring complex extraction or purification steps that erode overall yield. Furthermore, the reliance on methanol as a solvent introduces substantial safety liabilities due to its low flash point of 11°C and wide explosive limits, necessitating costly explosion-proof equipment and rigorous safety monitoring. The difficulty in recycling these metal salts also leads to increased heavy metal waste discharge, creating environmental compliance burdens that are increasingly untenable for modern chemical manufacturing facilities aiming for sustainability.
The Novel Approach
The methodology outlined in the patent data represents a paradigm shift by integrating advanced ligand design with practical process engineering to overcome these historical barriers. By utilizing salt-group modified Schiff base metal chelates, the invention creates a catalyst that possesses high intrinsic activity for aerobic oxidation while exhibiting tunable solubility properties. The use of ethylene glycol or ethylene glycol-water mixtures as the reaction medium eliminates the fire hazards associated with methanol, allowing reactions to proceed safely at elevated temperatures between 60°C and 120°C under atmospheric pressure. Crucially, the structural modification of the catalyst with ionic liquid-like salt groups ensures that while the catalyst is active during the reaction, it can be precipitated and recovered simply by adding water post-reaction. This innovation transforms the oxidation step from a wasteful, linear process into a circular, efficient cycle, drastically simplifying the workup procedure and enabling the consistent production of high-purity vanillin with yields ranging from 85% to 95%.
Mechanistic Insights into Schiff Base Metal Chelate Catalysis
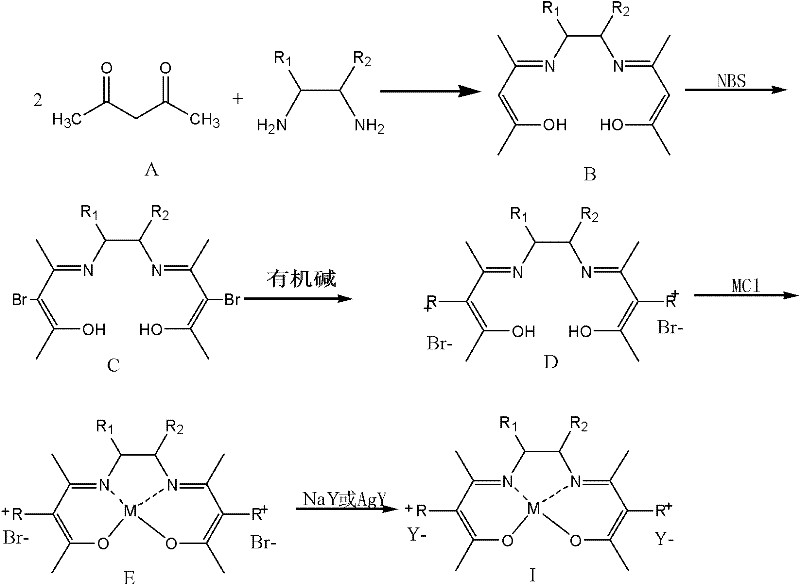
The core of this technological advancement lies in the sophisticated molecular architecture of the catalyst, specifically the salt-group modified Schiff base metal chelates defined by Formula (I). The synthesis begins with the condensation of acetylacetone and various diamines to form a stable Schiff base backbone, which is subsequently brominated and functionalized with organic bases like N-methylimidazole or pyridine to introduce cationic character. This "ionic liquid-supported" structure is then coordinated with transition metal ions such as Fe, Co, Cu, or Mn, creating a highly active center for oxygen activation. The presence of the bulky ionic groups and the specific anion (e.g., PF6-, BF4-) plays a dual role: it stabilizes the metal center against leaching and aggregation, and it modulates the polarity of the complex. During the oxidation of 4-methylguaiacol, the metal center facilitates the transfer of electrons from the substrate to molecular oxygen, generating the aldehyde functionality with high regioselectivity. The robustness of the Schiff base ligand prevents oxidative degradation of the catalyst itself, ensuring that the active species remains intact over multiple cycles, which is a common failure point for simpler metal-ligand systems in harsh alkaline environments.
Impurity control in this system is inherently managed through the precise tuning of the catalyst's steric and electronic environment. In traditional metal salt catalysis, non-selective radical pathways often lead to over-oxidation products or polymerization of the phenolic substrate, resulting in dark-colored tars that are difficult to remove. However, the well-defined coordination sphere of the Schiff base chelate directs the oxidation specifically to the methyl group of the guaiacol ring, suppressing side reactions on the aromatic ring or the methoxy group. The patent data highlights a vanillin selectivity exceeding 85%, indicating that the catalyst effectively discriminates between the desired oxidation pathway and competing degradation routes. Furthermore, the ability to recover the catalyst via centrifugation before the acidification step prevents metal contamination of the final vanillin crystals. This physical separation mechanism ensures that the final product meets stringent purity specifications of 98.0% or higher without the need for extensive chromatographic purification, thereby preserving the integrity of the flavor profile which is critical for food and fragrance applications.
How to Synthesize Vanillin Efficiently
The implementation of this catalytic system requires a disciplined approach to reagent preparation and reaction monitoring to fully realize the reported efficiency gains. The process begins with the meticulous synthesis of the catalyst precursor, where stoichiometry and temperature control during the Schiff base formation and metal chelation steps are critical to ensuring the correct coordination geometry. Once the catalyst is prepared, the oxidation reaction is conducted by suspending 4-methylguaiacol and a stoichiometric amount of base in ethylene glycol, followed by the introduction of the catalyst at loadings as low as 0.001% to 0.1% by weight. Oxygen or air is sparged through the mixture at temperatures between 60°C and 120°C for a duration of 6 to 25 hours, depending on the specific catalyst variant used. Detailed standardized synthesis steps see the guide below.
- Prepare the salt-group modified Schiff base metal chelate catalyst by reacting acetylacetone with diamines, followed by bromination, ionic liquid support, metal coordination, and anion exchange.
- Conduct the oxidation reaction by mixing 4-methylguaiacol, base (NaOH/KOH), ethylene glycol solvent, and the catalyst, then introducing oxygen or air at 60-120°C for 6-25 hours.
- Recover the catalyst via centrifugal filtration after adding water, then isolate vanillin sodium salt through crystallization, acidification, and recrystallization to achieve >98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic oxidation technology translates into tangible strategic advantages that extend far beyond simple yield improvements. The primary value driver is the drastic simplification of the downstream processing workflow; because the catalyst can be recovered via simple filtration rather than complex extraction or distillation, the number of unit operations is significantly reduced. This reduction in processing complexity directly correlates to lower capital expenditure on equipment and reduced operational expenditures related to energy consumption and labor. Additionally, the elimination of methanol removes a major regulatory and safety burden, lowering insurance costs and reducing the need for specialized hazardous material handling infrastructure. The ability to recycle the expensive metal-chelate catalyst multiple times without significant loss of activity further decouples production costs from volatile raw material markets, providing a more predictable and stable cost structure for long-term supply contracts.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the high atom economy and catalyst recyclability. By avoiding the loss of expensive transition metals in the waste stream and eliminating the need for solvent recovery systems associated with volatile methanol, the overall cost of goods sold is substantially optimized. The high conversion rate of 100% ensures that raw material utilization is maximized, meaning less starting material is required to produce the same amount of finished vanillin. Furthermore, the simplified workup reduces the consumption of auxiliary chemicals such as acids and bases needed for pH adjustments and extractions, leading to comprehensive savings across the entire manufacturing value chain without compromising on product quality.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by process upsets related to safety incidents or equipment failures, risks that are mitigated by this safer solvent system. The use of ethylene glycol, a high-boiling and non-flammable solvent, allows for more flexible scheduling and reduced downtime for safety purges compared to methanol-based processes. The robustness of the catalyst system also means that production batches are more consistent, reducing the variance in cycle times and allowing for more accurate forecasting of delivery dates. This reliability is crucial for serving large-scale customers in the food and pharmaceutical industries who require just-in-time delivery of critical ingredients, positioning manufacturers using this technology as preferred partners for mission-critical supply chains.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste generation, but this technology inherently minimizes the environmental footprint through its design. The catalyst recovery loop prevents heavy metal discharge, addressing one of the most stringent aspects of environmental regulation in fine chemical manufacturing. The reduction in volatile organic compound (VOC) emissions due to the absence of methanol simplifies permitting and compliance reporting, facilitating faster approval for capacity expansions. The process generates significantly less hazardous waste, lowering disposal costs and aligning with the increasing corporate sustainability goals of downstream customers who are auditing their suppliers for green chemistry credentials, thus future-proofing the supply relationship against tightening global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced vanillin synthesis route. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D teams planning pilot trials and for procurement teams assessing the long-term viability of this supply source.
Q: How does the salt-group modified catalyst improve separation compared to traditional metal salts?
A: Traditional transition metal salts often remain dissolved in the reaction mixture, making separation difficult. The salt-group modified Schiff base chelates described in CN102381950A are designed to be insoluble in the specific oxidation medium post-reaction (especially after water addition), allowing for simple centrifugal filtration and recycling without complex extraction processes.
Q: What are the safety advantages of using ethylene glycol over methanol in vanillin synthesis?
A: Methanol has a very low flash point (11°C) and wide explosion limits, posing significant fire and toxicity risks in large-scale oxidation. Ethylene glycol has a much higher boiling point and flash point, significantly reducing volatility and flammability hazards, thereby lowering the requirement for expensive explosion-proof infrastructure and improving overall plant safety.
Q: What purity levels can be achieved with this catalytic oxidation method?
A: The patent data indicates that this method achieves a 4-methylguaiacol conversion rate of 100% and a vanillin selectivity of greater than 85%. Following standard workup and recrystallization, the final vanillin product reaches a purity of 98.0% to 98.6%, meeting stringent specifications for food and pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vanillin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic technologies requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising lab-scale results of this Schiff base catalysis can be seamlessly translated into industrial reality. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the 98%+ purity profiles required for high-end flavor and fragrance applications. We understand the critical nature of vanillin in your formulations and are committed to delivering a supply stream that is both chemically superior and commercially resilient.
We invite you to engage with our technical procurement team to discuss how this catalytic oxidation method can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your competitive advantage in the global market for fine chemical intermediates.
