Scalable Visible Light Catalyzed Synthesis of Tetrahydroquinoline Intermediates for Pharma
Introduction to Advanced Photocatalytic Methodologies
The landscape of pharmaceutical intermediate synthesis is undergoing a significant transformation driven by the demand for greener, more efficient, and cost-effective methodologies. A pivotal advancement in this domain is detailed in patent CN108017580B, which discloses a novel method for synthesizing 1,2,3,4-tetrahydroquinoline compounds via visible light-catalyzed decarboxylation of amino acids. This technology represents a paradigm shift from traditional thermal or metal-heavy processes to a sustainable organophotocatalytic approach. By utilizing an organic photocatalyst known as DPZ in conjunction with visible light irradiation, this method achieves high conversion rates under exceptionally mild conditions, specifically at temperatures ranging from 20°C to 30°C in an air atmosphere. For R&D directors and process chemists, this offers a compelling alternative to harsh synthetic routes, while supply chain managers will appreciate the simplification of reaction parameters and the elimination of scarce noble metal catalysts. The ability to transform simple N-phenyl amino acid precursors into complex heterocyclic scaffolds with high selectivity underscores the potential of this technology to streamline the manufacturing of high-value active pharmaceutical ingredients (APIs) and fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydroquinoline derivatives has relied heavily on methods that present significant operational and economic challenges for large-scale manufacturing. Traditional routes often involve multicomponent reactions that require precise stoichiometric control and generate complex impurity profiles, making downstream purification difficult and costly. Alternatively, the hydrogenation of quinoline compounds, while effective, typically necessitates the use of high-pressure hydrogen gas and expensive transition metal catalysts, introducing safety hazards and stringent regulatory hurdles regarding residual metal limits in final drug substances. Furthermore, earlier photocatalytic approaches frequently depended on ruthenium or iridium polypyridyl complexes. Although these metal-based catalysts exhibit strong visible light absorption, their high cost, potential toxicity, and the difficulty in completely removing trace metals from the product stream pose severe limitations for GMP-compliant pharmaceutical production. These conventional methods often operate under harsh conditions, requiring elevated temperatures or inert atmospheres, which significantly increase energy consumption and capital expenditure for specialized reactor infrastructure.
The Novel Approach
In stark contrast, the methodology described in patent CN108017580B introduces a streamlined, metal-free protocol that leverages the power of organic photocatalysis. The core innovation lies in the use of the DPZ organic photocatalyst, which facilitates the decarboxylative cyclization of N-phenyl amino acids under ambient air conditions. This approach drastically simplifies the reaction setup by replacing oxygen cylinders or inert gas lines with simple air balloons, thereby reducing operational complexity and safety risks associated with pure oxygen handling. The reaction proceeds efficiently at room temperature (20-30°C), eliminating the need for energy-intensive heating or cooling systems. As illustrated in the specific reaction pathway below, the transformation is direct and atom-economical, converting readily available amino acid derivatives into the desired tetrahydroquinoline core with excellent yields.
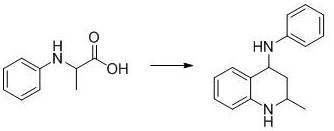
The versatility of this system is further evidenced by its tolerance to various substituents on the aromatic rings, allowing for the synthesis of a diverse library of analogues including those with halogen, alkyl, and alkoxy groups. This flexibility is crucial for medicinal chemists exploring structure-activity relationships (SAR) during drug discovery phases. Moreover, the use of 4Å molecular sieves as an additive ensures the removal of moisture, driving the reaction equilibrium towards product formation and maintaining catalyst activity over extended periods. This novel approach not only addresses the environmental concerns associated with heavy metal waste but also offers a robust platform for the cost reduction in API manufacturing by utilizing inexpensive, commercially available starting materials and catalysts.
Mechanistic Insights into DPZ-Catalyzed Decarboxylative Cyclization
The mechanistic elegance of this transformation centers on the unique photophysical properties of the DPZ organic photocatalyst. Upon irradiation with visible light (specifically blue LEDs with wavelengths of 450-455 nm), the DPZ molecule absorbs photon energy to reach an excited state capable of engaging in single-electron transfer (SET) processes. In this catalytic cycle, the excited photocatalyst interacts with the N-phenyl amino acid substrate, initiating a radical decarboxylation event. This generates a reactive carbon-centered radical intermediate which subsequently undergoes intramolecular cyclization onto the pendant aniline moiety. The overall process is oxidative, utilizing molecular oxygen from the ambient air as the terminal oxidant to regenerate the ground state of the catalyst and close the catalytic cycle. This reliance on air as a green oxidant is a critical feature, as it avoids the use of stoichiometric chemical oxidants that often produce hazardous byproducts. The general reaction scheme highlights the broad substrate scope, where R groups can vary from hydrogen to halogens and alkyl chains, demonstrating the robustness of the radical mechanism against steric and electronic variations.
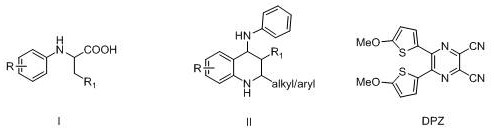
From an impurity control perspective, this mechanism offers distinct advantages over thermal radical initiators. The mild photochemical activation ensures that side reactions typically associated with high-temperature thermal degradation are minimized. The selectivity of the DPZ catalyst promotes the desired cyclization pathway over competing dimerization or polymerization of the radical intermediates. Furthermore, the absence of transition metals eliminates the risk of metal-catalyzed side reactions or the formation of metal-coordinated impurities that are notoriously difficult to purge. The inclusion of 4Å molecular sieves plays a dual role: physically adsorbing water produced during the oxidation steps and preventing hydrolysis of sensitive intermediates. This rigorous control over the reaction environment results in clean reaction profiles, simplifying the workup procedure to a straightforward solvent removal and column chromatography, which is highly desirable for maintaining high purity specifications in pharmaceutical intermediates.
How to Synthesize Tetrahydroquinoline Derivatives Efficiently
Implementing this visible light-catalyzed protocol requires attention to specific operational details to maximize yield and reproducibility. The process begins with the preparation of the reaction mixture in a dry environment, emphasizing the importance of excluding excess moisture prior to the addition of molecular sieves. The substrate, typically an N-phenyl amino acid, is dissolved in a chlorinated solvent such as chloroform or dichloromethane, which provides the optimal polarity for the photocatalytic cycle. The reaction vessel is then sealed and equilibrated with dry air, ensuring a steady supply of oxygen without introducing humidity. Irradiation is performed using low-power blue LED lamps, which are energy-efficient and provide the specific wavelength required to activate the DPZ catalyst without causing thermal stress to the reaction mixture. The detailed standardized synthesis steps, including precise molar ratios and purification techniques, are outlined in the guide below.
- Dissolve organic photocatalyst DPZ (0.2-0.4 mol%) in toluene, dry thoroughly, and add N-phenyl amino acid substrate along with 4A molecular sieves in chloroform.
- Seal the reaction flask with a rubber stopper, insert a balloon filled with dry air, and stir at 20-30°C under irradiation with 1W blue LED lamps (450-455 nm) for 5-15 hours.
- Remove solvent via rotary evaporation and purify the crude residue using silica gel column chromatography (n-hexane/ethyl acetate) to isolate the target tetrahydroquinoline compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic benefits regarding cost stability and supply reliability. The primary driver for cost optimization is the complete elimination of noble metal catalysts. Traditional ruthenium or iridium complexes are subject to volatile market prices and supply constraints due to their geological scarcity. By switching to the synthetic organic DPZ catalyst, manufacturers can decouple their production costs from the fluctuations of the precious metals market. Additionally, the use of air as the oxidant removes the need for purchasing, storing, and handling compressed oxygen or hazardous chemical oxidants, further reducing operational expenditures and safety compliance costs. The mild reaction conditions also imply lower energy consumption for heating or cooling, contributing to a reduced carbon footprint and lower utility bills over the lifecycle of the product.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the simplification of the catalyst system and reaction conditions. By utilizing an organic photocatalyst that is easy to synthesize and stable, the direct material costs are significantly lowered compared to metal-based alternatives. The high yields reported in the patent data, often exceeding 80% for various substrates, mean that less raw material is wasted, improving the overall mass balance and efficiency of the production line. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of purification media and solvents. This holistic reduction in material and processing inputs leads to substantial cost savings in pharmaceutical intermediate manufacturing, allowing for more competitive pricing in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and commodity-grade starting materials. N-phenyl amino acids and common organic solvents like chloroform are produced at a massive global scale, ensuring consistent availability and minimizing the risk of shortages that often plague specialty reagents. The robustness of the reaction to air and ambient temperature means that the process is less sensitive to minor variations in utility supplies, such as steam or chilled water, which can be a bottleneck in crowded manufacturing facilities. This operational flexibility allows for more reliable production scheduling and shorter lead times for high-purity tetrahydroquinoline derivatives, ensuring that downstream API synthesis is not delayed by intermediate supply disruptions.
- Scalability and Environmental Compliance: The scalability of this photochemical process is validated by the successful gram-scale amplification experiments detailed in the patent, which maintained high efficiency even at larger volumes. The absence of heavy metals simplifies the environmental compliance landscape, as there is no need for expensive wastewater treatment systems designed to remove trace ruthenium or iridium. This aligns perfectly with increasingly stringent global environmental regulations regarding heavy metal discharge. The green nature of the process, utilizing visible light and air, positions the manufacturer as a leader in sustainable chemistry, which is a growing requirement for partnerships with major multinational pharmaceutical companies focused on ESG (Environmental, Social, and Governance) goals.
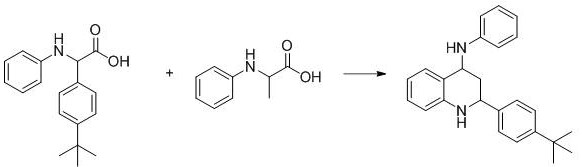
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light-catalyzed synthesis method. These answers are derived directly from the experimental data and technical disclosures within patent CN108017580B, providing a factual basis for evaluating the technology's fit for your specific production needs. Understanding these nuances is essential for R&D teams planning process validation and for procurement teams assessing the long-term viability of this supply route. We encourage stakeholders to review these details to fully appreciate the operational advantages offered by this metal-free photocatalytic platform.
Q: What are the advantages of using DPZ photocatalyst over traditional ruthenium or iridium complexes?
A: The DPZ organic photocatalyst eliminates the need for expensive and toxic noble metals like ruthenium or iridium. It offers lower synthesis costs, easier removal from the final product, and aligns with green chemistry principles by avoiding heavy metal contamination in pharmaceutical intermediates.
Q: Can this visible light catalyzed decarboxylation method be scaled up for industrial production?
A: Yes, the patent data demonstrates successful gram-scale amplification (Example 10) yielding 78% conversion. The mild reaction conditions (20-30°C) and use of air as an oxidant suggest high feasibility for commercial scale-up without requiring high-pressure or high-temperature equipment.
Q: What is the role of 4A molecular sieves in this synthesis?
A: The 4A molecular sieves act as a desiccant to remove trace water generated during the reaction or present in the solvent. This helps drive the equilibrium forward, prevents catalyst deactivation, and ensures high yields and selectivity for the tetrahydroquinoline products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light photocatalysis in modern pharmaceutical synthesis. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the DPZ-catalyzed decarboxylation can be seamlessly translated into robust industrial processes. Our facility is equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required for global regulatory submissions. We are committed to delivering high-purity tetrahydroquinoline intermediates that adhere to the highest quality standards, leveraging our technical expertise to optimize yield and minimize impurities through advanced process control.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free route can enhance your margin structure. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us help you secure a sustainable and cost-effective supply chain for your critical pharmaceutical intermediates.
