Revolutionizing Bempedoic Acid Intermediate Production via Trityl Protection Strategy
Revolutionizing Bempedoic Acid Intermediate Production via Trityl Protection Strategy
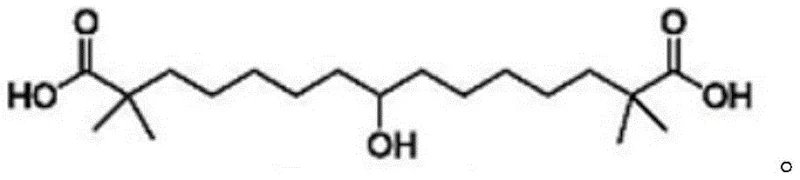
The pharmaceutical landscape for lipid-lowering therapies has been significantly transformed by the advent of Bempedoic Acid, a first-in-class ATP citrate lyase inhibitor approved for treating dyslipidemia. As global demand for this novel non-statin therapy surges, the efficiency and safety of its supply chain have become paramount concerns for multinational pharmaceutical corporations. Patent CN115611739A introduces a groundbreaking preparation method for Bempedoic Acid intermediates that addresses critical bottlenecks in traditional manufacturing. This technical insight report analyzes the proprietary trityl-protection strategy detailed in the patent, offering a comprehensive evaluation of its mechanistic robustness, operational safety, and commercial viability for large-scale API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Bempedoic Acid and its precursors has been plagued by significant safety hazards and purification challenges that hinder industrial scalability. Prior art, such as the route disclosed in WO2004067489, relies heavily on the use of p-toluenesulfonylmethyl isocyanide, a reagent known for its high toxicity and poor atom economy. Furthermore, these conventional processes often necessitate the use of sodium hydride, a dangerously pyrophoric substance that poses severe explosion risks upon contact with moisture or air, thereby complicating reactor engineering and safety protocols. Beyond safety, the downstream processing in these legacy routes frequently generates potential genotoxic impurities, specifically p-toluenesulfonyl derivatives, which require rigorous and costly removal steps to meet stringent regulatory standards for active pharmaceutical ingredients.
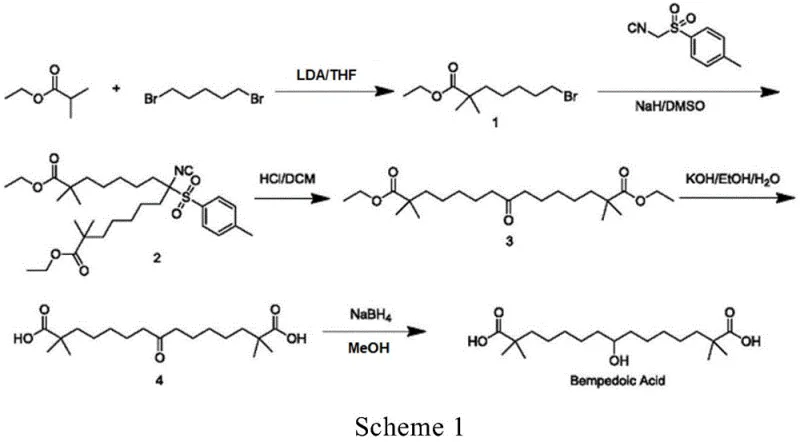
The Novel Approach
In stark contrast to these hazardous legacy methods, the novel approach described in CN115611739A leverages a strategic trityl (triphenylmethyl) protection group to fundamentally alter the physicochemical properties of the intermediates. By employing triphenylmethanol to protect the carboxylic acid functionality early in the synthesis, the resulting intermediates exhibit enhanced crystallinity, allowing for purification via simple recrystallization rather than labor-intensive column chromatography. This shift not only drastically simplifies the isolation process but also significantly improves the overall yield and purity profile of the final intermediate. The use of mild, commercially available reagents such as alkali metal carbonates and standard organic solvents ensures that the process is inherently safer and more amenable to continuous manufacturing environments, effectively eliminating the risks associated with explosive hydrides and toxic isocyanides.
Mechanistic Insights into Trityl-Protection and Alkylation Strategy
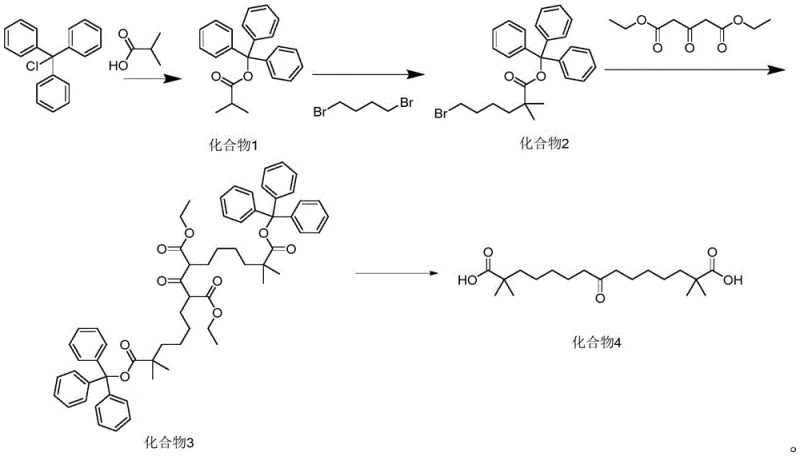
The core innovation of this synthetic route lies in the sequential construction of the carbon backbone using a protected isobutyrate scaffold. The process initiates with the esterification of isobutyric acid with triphenylmethanol using DIC (1,3-diisopropylcarbodiimide) as a coupling agent in dichloromethane. This step forms Compound 1, a sterically hindered ester that serves as a stable platform for subsequent functionalization. The bulky trityl group not only protects the acid moiety but also influences the steric environment around the alpha-carbon, facilitating controlled alkylation. In the subsequent step, Compound 1 undergoes deprotonation with lithium diisopropylamide (LDA) at low temperatures (-10 to 0°C) to generate a kinetic enolate, which then reacts with 1,4-dibromobutane to extend the carbon chain, yielding Compound 2 with high regioselectivity.
Following the chain extension, the synthesis proceeds through a critical condensation reaction where Compound 2 reacts with diethyl 1,3-acetonedicarboxylate. This transformation is catalyzed by alkali metal carbonates (such as cesium or potassium carbonate) in the presence of an iodide source, which acts as a nucleophilic catalyst to enhance the displacement of the bromide leaving group. The reaction conditions are remarkably mild, typically conducted in ethanol at 60-70°C, avoiding the harsh thermal stress often required in traditional Claisen condensations. The final hydrolysis step utilizes aqueous alkali to simultaneously cleave the ethyl esters and remove the trityl protecting group, precipitating the target keto-diacid intermediate (Compound 4) with exceptional purity levels exceeding 99.6%, demonstrating the efficacy of the protection-deprotection strategy in impurity control.
How to Synthesize Bempedoic Acid Intermediate Efficiently
The implementation of this trityl-based synthetic route requires precise control over reaction parameters to maximize yield and minimize byproduct formation. The process is designed to be operationally straightforward, utilizing standard reactor setups common in fine chemical manufacturing. Key to the success of this method is the maintenance of anhydrous conditions during the alkylation phase and the careful selection of solvent systems for the recrystallization steps to ensure optimal crystal growth and impurity rejection. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures required to execute this synthesis at scale, please refer to the standardized protocol outlined below.
- Protect isobutyric acid with triphenylmethanol using DIC in DCM to form Compound 1.
- Alkylate Compound 1 with 1,4-dibromobutane using LDA in THF to generate Compound 2.
- Condense Compound 2 with diethyl 1,3-acetonedicarboxylate using carbonate base and iodide catalyst to yield Compound 3.
- Hydrolyze and deprotect Compound 3 using aqueous alkali to obtain the final keto-diacid intermediate (Compound 4).
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this novel synthetic methodology offers substantial advantages in terms of cost structure and supply chain resilience. By eliminating the need for specialized, hazardous reagents like sodium hydride and toxic isocyanides, manufacturers can significantly reduce the costs associated with hazardous material handling, storage, and waste disposal. The transition from column chromatography to recrystallization for purification represents a major operational efficiency gain, as chromatography is often a bottleneck in large-scale production due to high solvent consumption and low throughput. This process intensification allows for faster batch cycles and reduced solvent recovery loads, directly contributing to a lower cost of goods sold (COGS) for the final intermediate.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the simplification of the purification workflow and the use of commodity chemicals. Eliminating column chromatography removes a capital-intensive and solvent-heavy unit operation, leading to substantial savings in both equipment depreciation and utility costs. Furthermore, the high yields reported in the patent examples (e.g., 95.4% for the protection step and 83-85% for the condensation) indicate a highly efficient atom economy, minimizing raw material waste. The avoidance of expensive and difficult-to-handle reagents also lowers the procurement burden, allowing for more predictable budgeting and reduced exposure to volatile specialty chemical markets.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as triphenylmethanol, isobutyric acid, and 1,4-dibromobutane ensures a robust and diversified supply base. Unlike routes dependent on custom-synthesized toxic building blocks, this method utilizes commodity feedstocks that are less susceptible to supply disruptions. The operational safety improvements also mitigate the risk of production stoppages due to safety incidents or regulatory inspections related to hazardous chemical usage. This stability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery requirements of global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, with reaction conditions that are easily transferable from pilot plants to multi-ton reactors. The use of mild bases and standard solvents aligns well with green chemistry principles, reducing the environmental footprint of the manufacturing process. The ability to recover and recycle the trityl alcohol byproduct further enhances the sustainability profile of the route. These factors collectively facilitate smoother regulatory approvals and environmental permitting, accelerating the time-to-market for generic or biosimilar versions of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trityl-protection strategy for Bempedoic Acid intermediates. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical implications for process development and quality assurance teams.
Q: Why is the trityl protection group advantageous for Bempedoic Acid synthesis?
A: The trityl group allows for purification via recrystallization rather than column chromatography, significantly improving scalability and reducing production costs while maintaining high purity.
Q: Does this method avoid genotoxic impurities found in prior art?
A: Yes, unlike previous methods using p-toluenesulfonylmethyl isocyanide, this route avoids the formation of potential genotoxic sulfonamide derivatives, ensuring better quality control for API manufacturing.
Q: What are the safety benefits of this new synthetic route?
A: The process eliminates the use of hazardous reagents like sodium hydride (which is pyrophoric) and toxic isocyanides, replacing them with safer carbonates and standard organic solvents suitable for industrial operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bempedoic Acid Intermediate Supplier
As the pharmaceutical industry continues to evolve towards safer and more efficient manufacturing paradigms, NINGBO INNO PHARMCHEM stands at the forefront of implementing these advanced synthetic technologies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this trityl-protection route are fully realized in a GMP-compliant environment. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite procurement leaders and R&D directors to engage with us to explore how this optimized synthesis can enhance your supply chain efficiency. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your regulatory filings and accelerate your project timelines.
