Scalable Non-Metal Catalyzed Synthesis of 1,3-Diketones for Advanced Pharmaceutical Intermediates
Scalable Non-Metal Catalyzed Synthesis of 1,3-Diketones for Advanced Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate synthesis is undergoing a significant transformation, driven by the urgent need for greener, more cost-effective, and scalable chemical processes. A pivotal advancement in this domain is detailed in Chinese patent CN110746278B, which discloses a novel non-metal-catalyzed method for preparing 1,3-diketone compounds based on alkynones. This technology represents a paradigm shift away from traditional reliance on expensive precious metal catalysts, offering a robust pathway for generating high-purity building blocks essential for drug discovery and development. The core innovation lies in a mild, two-stage reaction sequence that can be executed either as a step-by-step process or a streamlined one-pot procedure, utilizing readily available organic and inorganic bases. For R&D directors and procurement strategists alike, this patent provides a compelling blueprint for optimizing supply chains and reducing the total cost of ownership for complex heterocyclic precursors.
1,3-Diketones are ubiquitous motifs in medicinal chemistry, serving as critical scaffolds for the rapid construction of imidazoles, pyrazoles, pyrimidines, and quinolines. Historically, accessing these structures with high fidelity has been fraught with challenges, often requiring harsh acidic conditions or sophisticated transition metal catalysis. The methodology outlined in CN110746278B circumvents these bottlenecks by employing a nucleophilic addition-hydrolysis strategy that operates under remarkably gentle thermal conditions, typically between 20°C and 80°C. This not only enhances safety profiles but also significantly broadens the functional group tolerance, allowing for the synthesis of diverse derivatives that might otherwise decompose under rigorous traditional protocols. As we delve deeper into the technical specifics, it becomes evident that this approach offers a reliable solution for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 1,3-diketones from alkynones was dominated by methodologies that presented substantial operational and economic drawbacks. Conventional routes often relied on direct hydrolysis using concentrated sulfuric acid or platinum tetrachloride at elevated temperatures, conditions that are inherently hazardous and difficult to control on a multi-kilogram scale. Furthermore, alternative strategies involving gold or silver complexes, while effective in laboratory settings, introduce prohibitive costs due to the price of the catalysts and the stringent requirement for their removal to meet pharmaceutical purity standards. Literature precedents, such as those by Glotova et al. and Nordmann et al., reported yields as low as 20% to 53% or required specific acidic conditions that generated significant by-product burdens. These limitations create a bottleneck for procurement managers seeking cost reduction in API manufacturing, as the downstream purification processes become excessively resource-intensive.
The Novel Approach
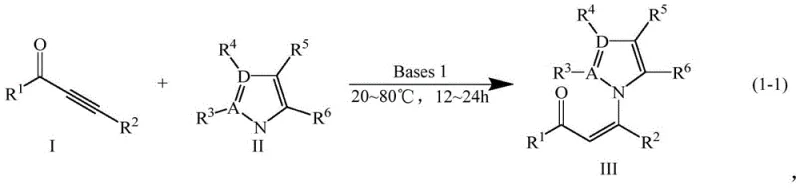
In stark contrast, the novel approach disclosed in the patent leverages a metal-free catalytic system that dramatically simplifies the synthetic workflow. By utilizing a combination of organic bases like 4-dimethylaminopyridine (DMAP) and triethylamine, followed by a hydrolysis step with alkali metal hydroxides, the process achieves yields generally exceeding 80%. This method effectively decouples the reaction efficiency from the need for rare earth or precious metals, thereby stabilizing the supply chain against fluctuations in metal commodity prices. The versatility of this approach is further highlighted by its compatibility with a wide array of substrates, including various substituted phenyl groups, heteroaryl rings, and cycloalkyl moieties. The ability to run this as a one-pot reaction, as illustrated in the general scheme below, minimizes solvent usage and unit operations, directly translating to enhanced process mass intensity (PMI) metrics.
Mechanistic Insights into Non-Metal Catalyzed Hydration
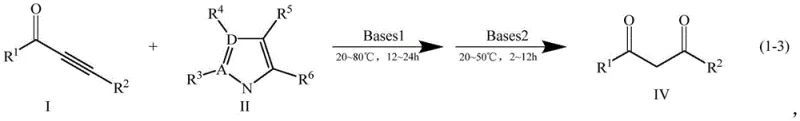
The mechanistic elegance of this transformation lies in the sequential activation of the alkynone triple bond by a nitrogen-containing aromatic nucleophile. In the first stage, the nitrogen heterocycle (such as imidazole or pyrazole) attacks the electron-deficient alkyne carbon, facilitated by the organic base which enhances the nucleophilicity of the nitrogen species. This results in the formation of a stable β-enaminone intermediate (Compound III), a structure that serves as a protected form of the 1,3-dicarbonyl system. This intermediate isolation or in-situ generation is crucial because it prevents the premature decomposition of the alkynone, a common side reaction observed in direct base-catalyzed hydrolysis attempts. The use of DMAP acts not just as a base but potentially as a nucleophilic catalyst, accelerating the addition step without being consumed in the stoichiometry of the final product.
The second stage involves the hydrolytic cleavage of the C-N bond in the enaminone intermediate to reveal the desired 1,3-diketone (Compound IV). This step is mediated by a stronger inorganic base, such as potassium hydroxide or sodium hydroxide, under mild thermal conditions (20-50°C). The mechanism likely proceeds through a hydroxide attack on the electrophilic carbon, followed by the elimination of the amine leaving group. This controlled hydrolysis ensures that the diketone backbone remains intact while avoiding the polymerization or degradation pathways often triggered by strong mineral acids. The result is a clean reaction profile with minimal impurity formation, which is a critical parameter for R&D directors focused on impurity profiling and regulatory compliance. The structural integrity of the final product is maintained, as evidenced by the successful synthesis of sensitive substrates containing halogens and electron-withdrawing groups.
How to Synthesize 1,3-Diketones Efficiently
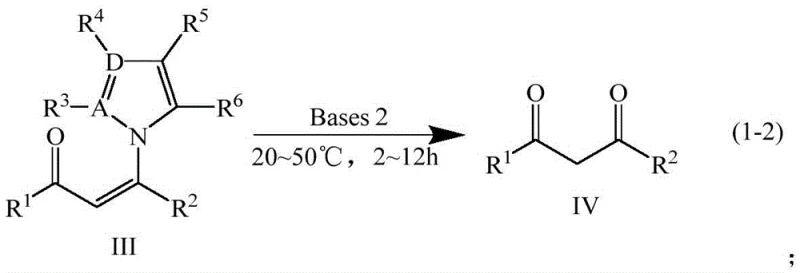
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity. The patent outlines a robust protocol that begins with the dissolution of the alkynone starting material in a polar aprotic solvent such as dimethyl sulfoxide (DMSO). The addition of the nitrogen source and the primary base must be carefully timed to ensure complete conversion to the intermediate before introducing the hydrolysis agent. For optimal results, the reaction temperature should be maintained within the specified 20-80°C window, with room temperature often proving sufficient for many substrates. Detailed standard operating procedures regarding molar ratios, specifically maintaining a base-to-substrate ratio between 1:1 and 1:5, are critical for reproducibility. The following section outlines the standardized synthesis steps derived directly from the patent examples to ensure consistent quality.
- Mix alkynone compound, nitrogen-containing aromatic compound (e.g., imidazole), and a first class base (e.g., DMAP/TEA) in a solvent like DMSO.
- React the mixture at 20-80°C for 12-24 hours to form the intermediate enaminone compound.
- Add a second class base (e.g., KOH, NaOH) directly to the reactor and continue reaction at 20-50°C for 2-12 hours to hydrolyze the intermediate into the final 1,3-diketone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this non-metal catalyzed methodology offers profound advantages for supply chain resilience and cost management. The elimination of gold and silver catalysts removes a significant variable cost component, as these precious metals are subject to volatile market pricing and require specialized recovery infrastructure. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling cycles, contributing to a lower carbon footprint and reduced utility costs. For procurement managers, this translates into a more predictable cost structure and the ability to source raw materials from a broader, more competitive supplier base, as the reagents involved are commodity chemicals rather than specialized proprietary catalysts.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete avoidance of precious metal catalysts. Traditional methods relying on gold or silver complexes incur high material costs and necessitate expensive purification steps to remove trace metal residues to ppm levels required by pharmacopeial standards. By substituting these with inexpensive organic amines and inorganic hydroxides, the direct material cost is drastically simplified. Additionally, the high yields reported (generally above 80%) minimize waste generation and improve the overall atom economy, ensuring that a greater proportion of the starting material is converted into valuable product rather than lost to side reactions or purification losses.
- Enhanced Supply Chain Reliability: The reagents utilized in this synthesis, such as DMSO, triethylamine, and potassium hydroxide, are globally available commodity chemicals with stable supply chains. This contrasts sharply with specialized ligands or metal complexes that may have long lead times or single-source dependencies. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, enhancing batch-to-b consistency. For supply chain heads, this reliability reduces the risk of production stoppages and ensures a continuous flow of high-purity pharmaceutical intermediates to downstream customers, safeguarding against market disruptions.
- Scalability and Environmental Compliance: The mild thermal profile (20-80°C) and the use of standard solvents make this process highly amenable to scale-up from laboratory to pilot and commercial plant scales. The absence of corrosive strong acids like concentrated sulfuric acid reduces the engineering requirements for reactor materials, allowing for the use of standard stainless steel equipment rather than glass-lined or Hastelloy reactors. From an environmental standpoint, the reduction in heavy metal waste simplifies effluent treatment and disposal, aligning with increasingly stringent global environmental regulations. This facilitates smoother regulatory approvals and reduces the long-term liability associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this non-metal catalyzed synthesis. These insights are derived from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, substrate scope, and process safety. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this synthesis method require expensive precious metal catalysts?
A: No, the method described in patent CN110746278B utilizes non-metal organic bases like DMAP and triethylamine, followed by inorganic hydroxides, completely eliminating the need for costly gold or silver catalysts.
Q: What is the typical yield range for this 1,3-diketone preparation?
A: According to the patent data, the reaction generally achieves yields above 80%, with specific examples demonstrating yields ranging from 80.3% to 99.3% depending on the substrate and conditions.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the mild reaction conditions (20-80°C) and the use of common solvents like DMSO make this process highly scalable and safer for industrial manufacturing compared to high-temperature or strong acid methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the non-metal catalyzed synthesis described in patent CN110746278B for the production of high-value pharmaceutical intermediates. Our team of expert chemists has extensively evaluated this pathway and confirmed its viability for large-scale production, ensuring that we can deliver 1,3-diketone compounds with exceptional purity and consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities and rigorous QC labs to meet the most stringent purity specifications required by global regulatory bodies. Our commitment to technological excellence allows us to offer clients a secure and efficient supply of critical building blocks for drug development.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data for our catalog items or to discuss route feasibility assessments for custom synthesis projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving innovation and efficiency in your pharmaceutical manufacturing operations.
