Advanced Catalytic Synthesis of Evodiamine: Technical Upgrades for Commercial Scale-Up
Advanced Catalytic Synthesis of Evodiamine: Technical Upgrades for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for producing bioactive alkaloids. A pivotal advancement in this domain is detailed in patent CN101941971B, which discloses a novel organic chemical synthesis method for Evodiamine. This quinoline alkaloid, historically extracted from the fruit of Tetradium ruticarpum (formerly Evodia rutaecarpa), possesses significant pharmacological potential, including anti-inflammatory, vasodilatory, and thermoregulatory properties. The patented process represents a paradigm shift from resource-constrained botanical extraction to efficient chemical synthesis, utilizing indole acetonitrile as a primary feedstock. By leveraging catalytic hydrogenation and optimized condensation reactions, this methodology not only secures a reliable supply chain for this high-value pharmaceutical intermediate but also drastically reduces the environmental footprint associated with its production.
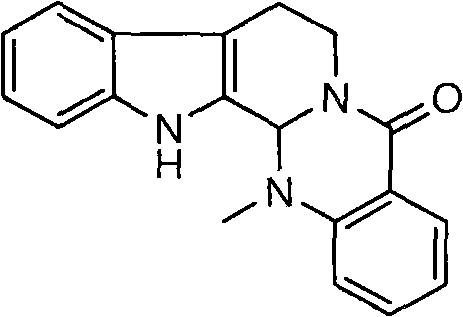
Evodiamine acts as a potent vanilloid receptor agonist and has garnered attention for its potential in treating obesity and cardiovascular disorders. The structural complexity of the molecule, featuring a fused indoloquinazolinone skeleton, has historically posed challenges for total synthesis. However, the approach outlined in the patent data provides a clear, step-wise roadmap that balances chemical elegance with industrial pragmatism. For R&D directors and procurement specialists alike, understanding the nuances of this synthetic route is critical for evaluating potential suppliers and optimizing the cost structure of downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the sourcing of Evodiamine relied heavily on extraction from medicinal plants, a method inherently fraught with variability due to seasonal changes, geographical origin, and agricultural conditions. While early chemical synthesis attempts were made, such as the biomimetic synthesis reported by Asahina and Ohta in 1928, many subsequent methods suffered from severe operational drawbacks. A notable domestic method reported in 2007 utilized 3,4-dihydro-β-carboline and N-methylanthranilic acid anhydride but relied heavily on phosphoryl chloride (POCl3) for cyclization steps. The use of POCl3 is a major liability in modern green chemistry; it generates substantial amounts of acidic wastewater and phosphorus-containing waste, necessitating expensive and complex waste treatment protocols. Furthermore, the overall yield of these conventional synthetic routes was often dismal, reportedly reaching a maximum of only 44%. Such low efficiency translates directly into exorbitant production costs and poor atom economy, rendering these methods unsuitable for the rigorous demands of large-scale industrial production.
The Novel Approach
In stark contrast, the method described in patent CN101941971B introduces a streamlined, five-step sequence that circumvents the use of hazardous phosphoryl chloride entirely. The innovation lies in the strategic selection of starting materials and the optimization of reaction conditions to maximize throughput. The process initiates with the catalytic hydrogenation of indole acetonitrile, a readily available and inexpensive commodity chemical. This is followed by a formylation reaction using ethyl formate, a mild and cost-effective reagent. The subsequent cyclization employs trifluoroacetic acid (TFA) instead of POCl3, significantly mitigating environmental hazards. Finally, the coupling of the carboline intermediate with a mixed anhydride derived from N-methylanthranilic acid ensures high regioselectivity and yield. This holistic redesign of the synthetic pathway results in an overall yield exceeding 65%, a substantial improvement over the 44% ceiling of previous methods, thereby achieving the dual goals of cost reduction and environmental compliance.
Mechanistic Insights into Pd/C Catalyzed Hydrogenation and Anhydride Condensation
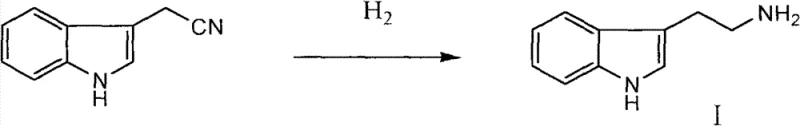
The cornerstone of this synthesis is the initial reduction step, where indole acetonitrile is converted into tryptamine (Intermediate I). This transformation is achieved through catalytic hydrogenation using 5% palladium on carbon (Pd/C). The choice of Pd/C is deliberate; it offers high activity for nitrile reduction while maintaining the integrity of the sensitive indole ring, which can be prone to over-reduction or polymerization under harsher conditions. The reaction is conducted in alcoholic solvents such as methanol or ethanol at temperatures ranging from 25°C to 100°C. The mechanism involves the adsorption of hydrogen gas onto the palladium surface, followed by the sequential addition of hydrogen atoms to the carbon-nitrogen triple bond of the nitrile group. This heterogeneous catalysis allows for easy separation of the catalyst via filtration, facilitating catalyst recovery and reuse, which is a key factor in reducing the cost of goods sold (COGS).
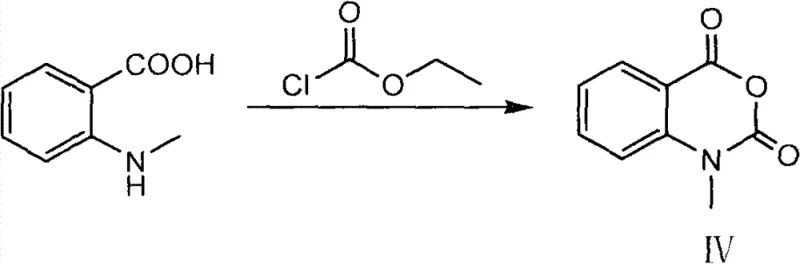
Following the formation of tryptamine, the synthesis proceeds through formylation and acid-catalyzed cyclization to generate the 3,4-dihydro-β-carboline core. Concurrently, the second key fragment, the mixed anhydride (Intermediate IV), is prepared from N-methylanthranilic acid and ethyl chloroformate. This activation strategy converts the carboxylic acid into a highly reactive electrophile without generating the corrosive byproducts associated with thionyl chloride or oxalyl chloride activations. The final condensation step involves the nucleophilic attack of the carboline nitrogen on the activated carbonyl of the mixed anhydride. This reaction is performed in a non-polar solvent system, such as dichloromethane or chloroform, under reflux conditions. The use of a mixed anhydride ensures that the acylation occurs selectively at the desired nitrogen atom, minimizing the formation of regioisomers and simplifying the purification process. This mechanistic precision is vital for ensuring the high purity specifications required for pharmaceutical applications.
Impurity control is rigorously managed throughout this process. The use of trifluoroacetic acid for cyclization, rather than harsher Lewis acids, prevents the degradation of the indole moiety. Furthermore, the recrystallization steps specified for the intermediates, particularly the white crystal formation of the mixed anhydride and the final product, serve as effective purification barriers. The patent data indicates that the final product exhibits a melting point of 270-271°C, consistent with literature values, and spectral data (IR and NMR) matches reference standards, confirming the structural fidelity of the synthesized Evodiamine. This level of characterization provides assurance to quality control teams regarding the identity and purity of the material.
How to Synthesize Evodiamine Efficiently
The synthesis of Evodiamine via this patented route requires precise control over reaction parameters to achieve the reported yields and purity profiles. The process is divided into distinct operational units: hydrogenation, formylation/cyclization, anhydride preparation, and final condensation. Each step builds upon the previous one, necessitating careful monitoring of stoichiometry, temperature, and reaction time. For instance, the hydrogenation step must be monitored for hydrogen uptake to ensure complete conversion of the nitrile group without excessive pressure that could compromise safety. Similarly, the preparation of the mixed anhydride requires strict exclusion of moisture to prevent hydrolysis of the activated species. The following guide outlines the standardized operational framework derived from the patent embodiments, serving as a baseline for process engineers aiming to implement this technology.
- Catalytic Hydrogenation: Reduce indole acetonitrile using 5% Pd/C catalyst in alcohol solvents (methanol/ethanol) at 25-100°C to prepare tryptamine.
- Formylation and Cyclization: React tryptamine with ethyl formate, followed by cyclization with trifluoroacetic acid in dichloromethane to form the carboline intermediate.
- Anhydride Preparation and Condensation: React N-methylanthranilic acid with ethyl chloroformate to form a mixed anhydride, then condense with the carboline intermediate in a non-polar solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits. The primary driver of value is the substitution of scarce, natural raw materials with abundant, petrochemical-derived feedstocks. Indole acetonitrile and N-methylanthranilic acid are commercially available in bulk quantities, insulating the supply chain from the volatility of agricultural harvests. Moreover, the elimination of phosphoryl chloride not only improves safety but also removes the need for specialized corrosion-resistant equipment and extensive neutralization infrastructure, leading to significant capital expenditure (CAPEX) and operational expenditure (OPEX) savings. The simplified waste profile allows for more straightforward regulatory compliance, reducing the administrative burden and potential fines associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The optimization of reaction conditions directly correlates to improved material efficiency. By achieving yields greater than 65% compared to the historical 44%, the consumption of raw materials per kilogram of finished product is drastically reduced. Additionally, the use of recoverable solvents like dichloromethane and ethanol, combined with the ability to filter and reuse the Pd/C catalyst, further drives down variable costs. The avoidance of expensive and hazardous reagents like POCl3 eliminates the hidden costs associated with their handling, storage, and neutralization, resulting in a leaner and more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: Dependence on botanical extraction subjects the supply of Evodiamine to geopolitical and climatic risks. By shifting to a fully synthetic route, manufacturers can guarantee consistent production schedules regardless of external agricultural factors. The robustness of the chemical steps, which utilize standard reactor types and common solvents, ensures that production can be easily scaled or shifted between different manufacturing sites without significant retooling. This flexibility is crucial for maintaining continuity of supply for downstream pharmaceutical clients who require just-in-time delivery of high-purity intermediates.
- Scalability and Environmental Compliance: The process is explicitly designed for large-scale industrial production. The reaction conditions, such as reflux temperatures and atmospheric pressure hydrogenation (or low pressure), are inherently scalable from pilot plant to multi-ton reactors. From an environmental perspective, the "green" nature of the synthesis—characterized by the absence of heavy metal waste (other than recoverable Pd) and phosphorus waste—aligns with increasingly stringent global environmental regulations. This proactive compliance minimizes the risk of production shutdowns due to environmental audits and enhances the corporate social responsibility (CSR) profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Evodiamine. These answers are derived directly from the technical specifications and beneficial effects reported in the patent literature, providing a transparent view of the process capabilities. Understanding these details helps stakeholders assess the feasibility of integrating this intermediate into their broader drug development or manufacturing pipelines.
Q: What are the primary advantages of this Evodiamine synthesis method over traditional extraction?
A: Unlike traditional extraction from Evodia fruit which is limited by natural resources, this chemical synthesis uses abundant raw materials like indole acetonitrile, ensuring supply continuity and significantly lower production costs with yields exceeding 65%.
Q: How does this process address environmental concerns associated with previous synthetic routes?
A: The novel method eliminates the use of highly corrosive and polluting phosphoryl chloride (POCl3) found in older methods, replacing it with safer reagents like trifluoroacetic acid and ethyl chloroformate, making it suitable for modern industrial environmental standards.
Q: Is this synthesis route scalable for large-scale API manufacturing?
A: Yes, the process utilizes standard unit operations such as catalytic hydrogenation and reflux condensation in common solvents like dichloromethane and alcohols, which are easily adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Evodiamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics. Leveraging our expertise in process chemistry, we have mastered the synthesis of complex alkaloids like Evodiamine using advanced catalytic methodologies. Our facility is equipped to handle diverse synthetic pathways, boasting extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of Evodiamine meets the exacting standards required for pharmaceutical applications. Our commitment to quality is matched only by our dedication to safety and environmental stewardship.
We invite pharmaceutical partners to collaborate with us to optimize their supply chains. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us demonstrate how our technical prowess and commercial acumen can drive value and reliability in your Evodiamine sourcing strategy.
