Scalable Synthesis of 4-Substituted Pyridine Derivatives via Novel Phosphorus Chemistry
Scalable Synthesis of 4-Substituted Pyridine Derivatives via Novel Phosphorus Chemistry
The landscape of heterocyclic chemistry is constantly evolving to meet the rigorous demands of modern agrochemical and pharmaceutical development. A pivotal advancement in this field is documented in Chinese Patent CN1898255A, which outlines a robust and versatile process for the preparation of substituted pyridine derivatives. This technology specifically addresses the longstanding challenge of synthesizing pyridines with electron-withdrawing substituents, such as trifluoromethyl groups, at the 4-position of the ring. Traditional synthetic routes often struggle with regioselectivity, frequently yielding the thermodynamically favored 6-substituted isomers instead of the desired 4-substituted targets. By leveraging modified Wittig and Horner-Wadsworth-Emmons (HWE) reactions coupled with innovative cyclization strategies, this patent provides a reliable pathway for producing high-purity intermediates essential for next-generation herbicides and pharmaceutical agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted pyridines has relied heavily on classical condensation reactions such as the Hantzsch synthesis or modifications of the Reformatsky reaction. While these methods are well-established, they exhibit significant limitations when applied to complex molecular architectures required by modern crop protection science. As noted in the background art of the patent, existing literature describes routes for 4-trifluoromethyl substituted pyridines via Reformatsky pathways, yet these often involve harsh conditions and multiple purification steps that erode overall yield. Furthermore, standard electrophilic substitution on pyridine rings is inherently difficult due to the electron-deficient nature of the heterocycle, often necessitating indirect approaches that lack precision. The tendency for side reactions to produce 6-substituted isomers creates a substantial burden on downstream purification, requiring energy-intensive chromatography or recrystallization to achieve the necessary purity standards for regulatory approval in agrochemical applications.
The Novel Approach
The methodology disclosed in CN1898255A represents a paradigm shift by constructing the pyridine ring through the reaction of alpha-beta-unsaturated carbonyl compounds with specialized phosphorus ylides. This approach allows for the precise installation of functional groups prior to ring closure, effectively bypassing the regioselectivity issues plaguing direct substitution methods. The process utilizes readily available starting materials, such as phosphonium salts or phosphonates, which react with enones under mild basic conditions to form linear intermediates. These intermediates then undergo cyclization, which can be catalyzed by Bronsted acids, zeolites, or ammonium salts, offering flexibility in process design. This modularity enables manufacturers to tailor the synthesis to specific substitution patterns, including the critical 4-trifluoromethyl and 3-thioalkyl configurations, ensuring a reliable agrochemical intermediate supplier can meet diverse client specifications without retooling entire production lines.
Mechanistic Insights into Phosphorus-Mediated Cyclization
The core of this technology lies in the generation and reactivity of the phosphorus ylide species. The process begins with the preparation of Wittig or Horner precursors, typically formed by reacting triphenylphosphine or trialkyl phosphites with organic halides containing electron-withdrawing groups like cyano or ester functionalities. Upon treatment with a base—ranging from alkali metal alcoholates to organolithium compounds—these precursors generate the active ylide species capable of nucleophilic attack. The reaction with the alpha-beta-unsaturated carbonyl compound proceeds via a conjugate addition mechanism, forming a new carbon-carbon bond and establishing the carbon skeleton of the future pyridine ring. The stereochemistry and electronic properties of the substituents on both the phosphorus reagent and the enone substrate play a crucial role in determining the efficiency of this coupling step, with optimal molar ratios typically ranging from 1:1 to 1:3 to drive the reaction to completion.
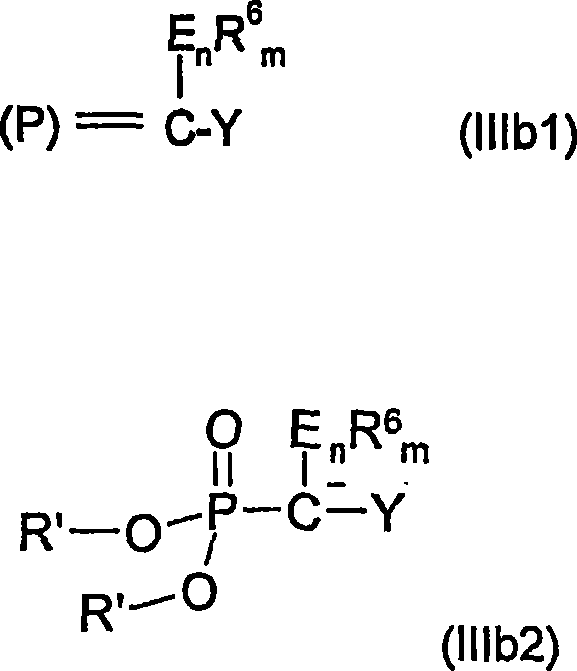
Following the initial coupling, the resulting acyclic intermediates possess the necessary functionality to undergo intramolecular cyclization. The patent details several distinct pathways for this ring-closing step, each offering unique advantages depending on the desired final substitution pattern. For instance, the use of zeolite catalysts at elevated temperatures facilitates a continuous flow process, which is highly advantageous for industrial scale-up. In this scenario, the intermediate passes through a heated tubular reactor where the acidic sites on the zeolite surface promote dehydration and aromatization. Alternatively, Bronsted acid catalysis in batch reactors allows for precise temperature control between -30°C and 150°C, accommodating sensitive functional groups that might degrade under harsher thermal conditions. This mechanistic versatility ensures that the synthesis can be adapted to produce a wide array of derivatives, including those with sulfur-containing groups at the 3-position, which are valuable precursors for sulfonamide herbicides.
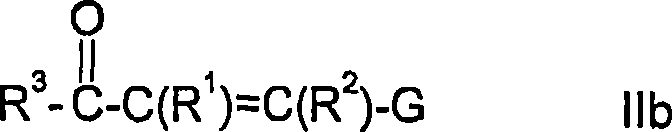
Impurity control is a critical aspect of this mechanistic pathway, particularly given the potential for isomer formation during the ylide addition step. The patent highlights that while mixtures of stereoisomers may form initially, the subsequent cyclization step often converges to the desired aromatic product, simplifying the purification profile. Furthermore, the ability to isolate intermediates via distillation or chromatography before cyclization provides an additional checkpoint for quality assurance. By carefully selecting the leaving group on the alpha-beta-unsaturated carbonyl substrate—such as alkoxy or amino groups—chemists can influence the rate of cyclization and the nature of byproducts. This level of control is essential for maintaining stringent purity specifications, ensuring that the final active pharmaceutical ingredients or agrochemicals derived from these intermediates meet global regulatory standards without requiring excessive reprocessing.
How to Synthesize 4-Substituted Pyridine Derivatives Efficiently
Implementing this synthesis route requires a systematic approach to reagent preparation and reaction condition optimization. The process generally involves three critical stages: the generation of the phosphorus ylide, the coupling with the unsaturated carbonyl substrate, and the final cyclization to form the pyridine ring. Operators must pay close attention to moisture exclusion during the ylide formation, as water can quench the reactive species and lower yields. Solvent selection is also paramount, with aprotic solvents like THF or DMF preferred for Wittig reactions, while alcohols may be suitable for Horner variants. The detailed standardized synthesis steps see the guide below.
- Preparation of phosphorus reagents (Wittig or Horner precursors) by reacting phosphines or phosphites with suitable organic halides.
- Reaction of the generated ylide reagents with alpha-beta-unsaturated carbonyl compounds in organic solvents at controlled temperatures.
- Cyclization of the resulting intermediates using Bronsted acids, zeolite catalysts, or ammonium salts to form the final pyridine ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers significant strategic benefits for procurement managers and supply chain directors seeking to optimize their sourcing of complex heterocyclic building blocks. The primary advantage lies in the substantial cost savings achieved through process intensification and the elimination of expensive transition metal catalysts often required in cross-coupling alternatives. By utilizing commodity phosphorus reagents and avoiding precious metals like palladium or platinum, the raw material costs are drastically reduced, and the risk of heavy metal contamination in the final product is virtually eliminated. This simplification of the supply chain not only lowers the cost of goods sold but also reduces the environmental footprint associated with mining and refining rare earth elements, aligning with increasingly strict corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the ability to perform telescoped reactions where intermediates are not isolated, thereby saving on solvent consumption and processing time. The use of continuous flow reactors with zeolite catalysts further drives down operational expenditures by enabling higher throughput and better heat management compared to traditional batch processes. Additionally, the high atom economy of the Wittig-type coupling minimizes waste generation, reducing the costs associated with waste disposal and environmental compliance. These factors combine to create a highly competitive cost structure for producing high-value pyridine derivatives.
- Enhanced Supply Chain Reliability: Securing a stable supply of critical intermediates is a top priority for any manufacturing organization, and this technology supports that goal by relying on widely available starting materials. The phosphorus precursors and enone substrates are commercially accessible from multiple vendors, mitigating the risk of single-source dependency that often plagues specialty chemical supply chains. Furthermore, the robustness of the reaction conditions allows for production in diverse geographic locations, enhancing supply chain resilience against regional disruptions. This flexibility ensures that downstream manufacturers can maintain consistent production schedules even in volatile market conditions.
- Scalability and Environmental Compliance: The scalability of this process is demonstrated by the patent's inclusion of continuous flow methodologies, which are inherently easier to scale from pilot plant to commercial production than batch operations. The ability to tune reaction parameters such as temperature and residence time in a flow reactor allows for precise control over product quality at large volumes. Moreover, the reduced reliance on hazardous reagents and the potential for solvent recycling contribute to a greener manufacturing profile. This alignment with environmental, social, and governance (ESG) criteria makes the technology attractive for companies aiming to reduce their carbon footprint and adhere to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pyridine synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is crucial for R&D teams evaluating the feasibility of integrating this route into their existing portfolios.
Q: How does this process improve regioselectivity for 4-substituted pyridines?
A: Unlike traditional methods that often yield 6-substituted isomers, this patent utilizes specific alpha-beta-unsaturated carbonyl precursors that direct substitution to the 4-position of the pyridine ring with high fidelity.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the patent explicitly describes continuous flow processes using tubular reactors packed with zeolite catalysts, demonstrating clear feasibility for commercial scale-up beyond batch processing.
Q: What are the key advantages regarding purification?
A: The process allows for the isolation of intermediates via distillation or chromatography if needed, but also supports one-pot cyclization strategies that significantly reduce solvent usage and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a partner who understands both the scientific intricacies and the commercial realities of fine chemical manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market launch is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. Whether you require custom synthesis of complex pyridine intermediates or large-scale supply of established agrochemical building blocks, our infrastructure is designed to support your growth.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis technology can be leveraged for your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your supply chain.
