Advanced Multicomponent Synthesis Of Carbonyl-Bridged Biheterocycles For Commercial API Production
Introduction To The Novel Multicomponent Synthesis Technology
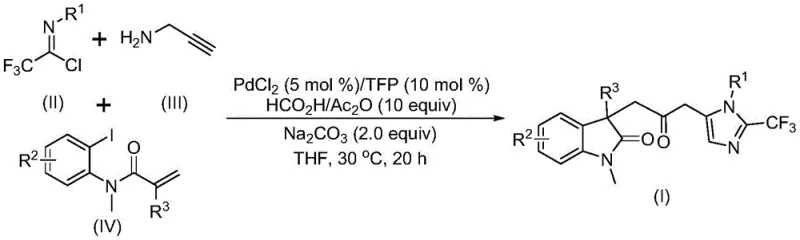
The pharmaceutical industry constantly seeks efficient pathways to construct complex heterocyclic scaffolds, which serve as the core backbone for numerous bioactive molecules. Patent CN115353511A introduces a groundbreaking preparation method for carbonyl-bridged biheterocyclic compounds, specifically targeting the synthesis of indolinone-imidazole derivatives that are prevalent in drug discovery pipelines. This technology represents a significant leap forward in organic synthesis by employing a transition metal palladium-catalyzed carbonylation cascade reaction. Unlike traditional methods that often require harsh conditions or toxic gases, this innovative approach utilizes cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives. The process operates under remarkably mild conditions, typically around 30°C, and avoids the direct use of hazardous carbon monoxide gas by employing a safe CO surrogate system. For R&D directors and process chemists, this patent offers a robust platform for generating diverse chemical libraries with high atom economy and excellent functional group compatibility, addressing the critical need for scalable and safe synthetic routes in modern medicinal chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems containing a carbonyl bridge has been fraught with synthetic challenges that hinder efficient manufacturing. Conventional strategies often rely on the direct coupling of two pre-formed heterocyclic substrates, which frequently suffers from low regioselectivity and poor yields due to steric hindrance and electronic deactivation. Alternative approaches involving oxidative cyclization of substrates with dual nucleophiles often necessitate the use of stoichiometric oxidants and harsh reaction conditions that are incompatible with sensitive functional groups commonly found in advanced intermediates. Furthermore, traditional carbonylation reactions typically require the handling of toxic carbon monoxide gas under high pressure, posing severe safety risks and requiring specialized, capital-intensive equipment that increases the barrier to entry for many manufacturing facilities. These limitations result in prolonged development timelines, increased waste generation, and higher overall production costs, making the search for a superior methodology a top priority for procurement and supply chain teams looking to optimize their API manufacturing processes.
The Novel Approach
The method disclosed in patent CN115353511A overcomes these historical bottlenecks through an elegant multicomponent reaction design that constructs the complex biheterocyclic framework in a single operational step. By leveraging a palladium-catalyzed cascade sequence, this novel approach simultaneously forms multiple chemical bonds, including C-C and C-N bonds, thereby drastically reducing the number of isolation and purification steps required. A key innovation is the replacement of gaseous carbon monoxide with an in-situ generated CO source derived from a mixture of formic acid and acetic anhydride, which not only enhances safety but also simplifies the reaction setup. The reaction proceeds efficiently at a mild temperature of 30°C in common organic solvents like tetrahydrofuran (THF), demonstrating exceptional substrate compatibility. As illustrated in the reaction scheme below, the convergence of trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives allows for the rapid assembly of the target scaffold with high structural diversity.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific analogues. The reaction is believed to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This is followed by an intramolecular Heck-type reaction that establishes the initial cyclic framework. Subsequently, the carbon monoxide released from the formic acid/acetic anhydride mixture inserts into the palladium-carbon bond to form an acyl-palladium intermediate, effectively installing the crucial carbonyl bridge. In parallel, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine species, which undergoes isomerization. The final cyclization step is catalyzed by the activated acyl-palladium intermediate, which facilitates the intramolecular nucleophilic attack to close the second heterocyclic ring, yielding the final carbonyl-bridged biheterocyclic compound. This intricate dance of catalytic cycles ensures high efficiency and selectivity, minimizing the formation of unwanted byproducts.
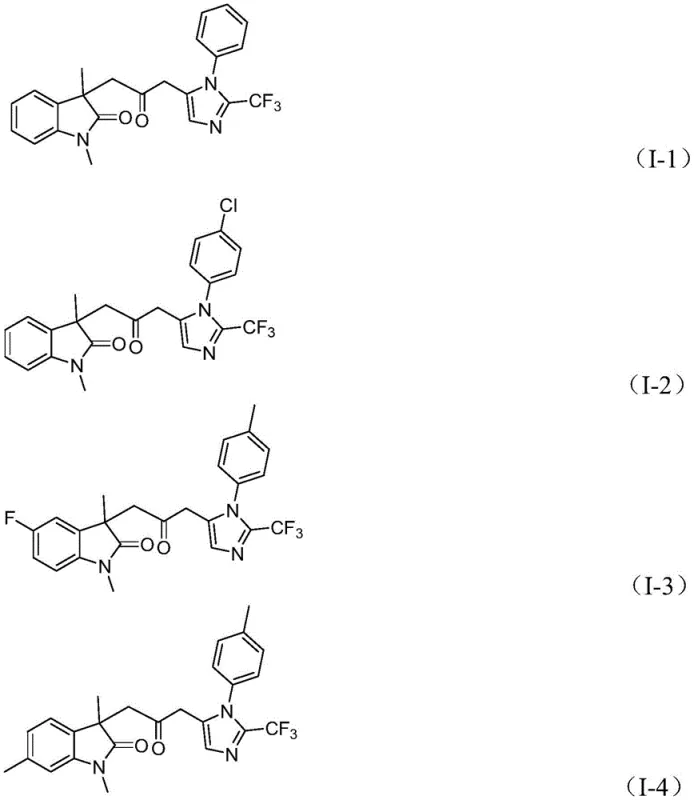
The impurity profile of this reaction is notably clean due to the high specificity of the palladium catalyst and the mild reaction conditions. The use of sodium carbonate as a base helps to neutralize acidic byproducts without promoting degradation of the sensitive imidazole or indolinone moieties. Furthermore, the choice of ligands, such as trifurylphosphine (TFP), plays a pivotal role in stabilizing the palladium center and enhancing the turnover number, which is critical for minimizing residual metal content in the final product. The patent data indicates that the reaction tolerates a wide range of substituents, including electron-withdrawing groups like nitro and trifluoromethyl, as well as electron-donating groups like methoxy and methyl, without significant loss in yield. This robustness suggests that the catalytic cycle is resilient to electronic variations in the substrate, providing a reliable platform for synthesizing diverse libraries of high-purity pharmaceutical intermediates.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
The experimental procedure outlined in the patent provides a clear roadmap for laboratory and pilot-scale synthesis, emphasizing simplicity and reproducibility. The process involves charging a reaction vessel with the palladium catalyst system, base, and CO surrogate, followed by the sequential addition of the three organic substrates in an aprotic solvent. The mixture is then stirred at a controlled temperature for a defined period to ensure complete conversion. While the patent details specific molar ratios and workup procedures involving silica gel treatment and column chromatography, the following section outlines the standardized operational steps required to achieve optimal results in a GMP-compliant environment.
- Prepare the reaction mixture by combining palladium chloride catalyst, TFP ligand, sodium carbonate base, and the CO surrogate system (formic acid/acetic anhydride) in an organic solvent like THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative into the Schlenk tube under inert atmosphere.
- Stir the reaction at 30°C for 12 to 20 hours, followed by filtration, silica gel treatment, and column chromatography purification to isolate the final biheterocyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the significant reduction of raw material costs and operational complexity. By utilizing commercially available and inexpensive starting materials such as propargylamine and acrylamide, the method avoids the reliance on exotic or custom-synthesized precursors that often carry high price tags and long lead times. Furthermore, the elimination of high-pressure carbon monoxide gas removes the need for specialized autoclaves and rigorous safety monitoring systems associated with toxic gas handling, leading to substantial capital expenditure savings and reduced insurance costs. The mild reaction temperature of 30°C also translates to lower energy consumption compared to processes requiring reflux or cryogenic conditions, contributing to a more sustainable and cost-effective manufacturing footprint.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of a palladium catalyst system that operates with high efficiency, potentially allowing for lower catalyst loading in optimized large-scale runs. The avoidance of toxic CO gas infrastructure significantly lowers the barrier to entry for manufacturing sites, while the high yields reported (up to 92% in specific examples) minimize material waste and maximize throughput. Additionally, the simplified workup procedure reduces the consumption of solvents and stationary phases during purification, further driving down the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term drug production, and this method excels by relying on commodity chemicals that are widely available from multiple global suppliers. The robustness of the reaction against functional group variations means that supply chain disruptions for specific substituted precursors can often be mitigated by switching to alternative analogues without re-optimizing the entire process. This flexibility ensures continuous production capability and reduces the risk of stockouts, providing a secure supply line for critical pharmaceutical intermediates needed for clinical and commercial stages.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of this method to gram-level quantities, indicating a clear path toward kilogram and tonne-scale production. The use of a CO surrogate instead of gas simplifies the engineering controls required for scale-up, making it easier to transfer the process from R&D labs to pilot plants and eventually to commercial manufacturing suites. Moreover, the reduced hazard profile aligns with increasingly stringent environmental, health, and safety (EHS) regulations, facilitating smoother regulatory approvals and minimizing the environmental impact of the manufacturing process through reduced waste generation and energy usage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this route for their specific projects.
Q: What are the safety advantages of this carbonylation method compared to traditional CO gas methods?
A: This method utilizes a formic acid and acetic anhydride mixture as a carbon monoxide surrogate, eliminating the need for handling toxic, high-pressure carbon monoxide gas cylinders, which significantly enhances operational safety and reduces regulatory compliance burdens in manufacturing facilities.
Q: What is the substrate scope for the R1 and R3 groups in this synthesis?
A: The reaction demonstrates excellent functional group tolerance, accommodating various substituents such as alkyl, halogen (chloro, bromo, fluoro), alkoxy, nitro, and trifluoromethyl groups on the aromatic rings, allowing for the diversification of the final pharmaceutical intermediate library.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability to gram-level reactions with high efficiency. The use of cheap, commercially available starting materials and mild reaction temperatures (30°C) makes it highly viable for cost-effective commercial scale-up without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed multicomponent synthesis for accelerating drug discovery and development programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle complex organometallic reactions with the highest standards of safety and quality, supported by our rigorous QC labs that guarantee stringent purity specifications for every batch of carbonyl-bridged biheterocyclic intermediates we produce. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting demands of the global healthcare industry.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, helping you identify opportunities for process optimization and budget efficiency. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and comprehensive quotations. Let us help you secure a reliable supply chain for your critical API intermediates while maximizing value through innovative chemistry.
