Optimizing Capecitabine Intermediate Production via Novel Inosine Halogenation Routes
The global demand for high-purity nucleoside analogs continues to surge, driven primarily by the widespread clinical adoption of oral fluoropyrimidine carbamate prodrugs such as capecitabine. As a critical precursor in the synthesis of this blockbuster antineoplastic agent, the intermediate 1,2,3-O-triacetyl-5-deoxy-beta-D-ribose represents a bottleneck in the supply chain due to historically complex manufacturing processes. Patent CN102260298B introduces a transformative methodology that shifts the synthetic paradigm from traditional sugar-based starting materials to a more efficient inosine-derived pathway. This technical breakthrough not only addresses the longstanding issues of low yield and poor stereocontrol but also aligns perfectly with the modern pharmaceutical industry's push for greener, more cost-effective manufacturing protocols. By leveraging selective halogenation and catalytic hydrogenolysis, this route offers a robust alternative for any reliable pharmaceutical intermediate supplier seeking to optimize their production capabilities.
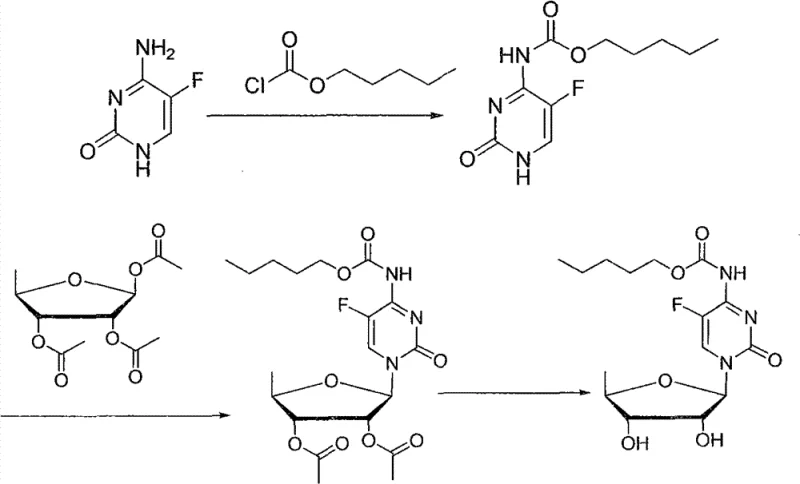
The strategic importance of this intermediate cannot be overstated, as its cost and quality directly dictate the economic viability of the final API. Historically, the market supply of 1,2,3-O-triacetyl-5-deoxy-beta-D-ribose has been constrained by inefficient synthetic routes that rely on expensive reagents or generate substantial hazardous waste. The technology disclosed in CN102260298B fundamentally re-engineers this landscape by utilizing inosine, a readily available and inexpensive nucleoside, as the primary feedstock. This shift allows manufacturers to bypass the multi-step protection and deprotection sequences associated with pentose sugars, thereby streamlining the entire value chain. For procurement managers and supply chain heads, understanding the mechanistic advantages of this patent is crucial for securing long-term contracts and ensuring the continuity of high-purity API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic strategies for accessing 5-deoxy-ribose derivatives have long been plagued by inherent inefficiencies that render them economically uncompetitive on an industrial scale. The most common historical approach, exemplified by US Patent No. 4340729, initiates from D-ribose and involves a lengthy sequence to install the deoxy functionality. As illustrated in the reaction pathways below, this method suffers from a critically low total recovery rate, typically hovering between 25% and 30%, primarily due to the formation of oily intermediates that are notoriously difficult to purify. Furthermore, the final acetylation step often yields a poor ratio of the desired beta-anomer to the alpha-anomer, necessitating costly chromatographic separations or recrystallizations that further erode yield. These technical hurdles result in a high cost of goods sold (COGS) and inconsistent batch-to-batch quality, making this route unsuitable for the rigorous demands of modern GMP manufacturing.
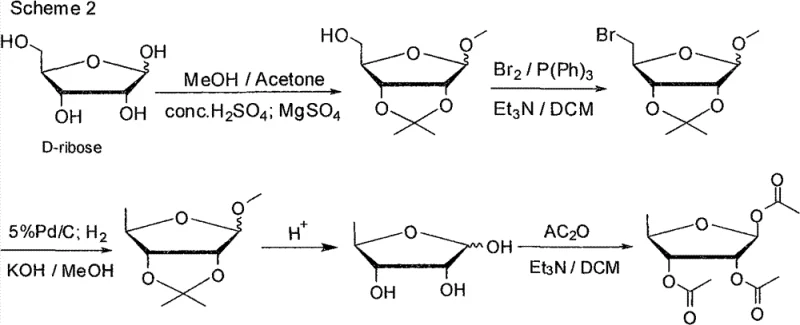
Even attempts to improve upon the D-ribose route by modifying the deoxygenation step, such as those reported in chemical defense research literature, failed to resolve the fundamental cost structure issues. Another significant prior art approach, detailed in Patent CN100432088, utilized inosine as a starting material but relied heavily on iodine as the halogenating agent. While this represented a conceptual improvement by using a nucleoside scaffold, it introduced severe environmental and economic drawbacks. The use of stoichiometric amounts of iodine and triphenylphosphine generates large volumes of black, iodine-containing wastewater and triphenylphosphine oxide byproducts, which are extremely difficult to remove from the product stream. Consequently, despite using a better starting material, the operational complexity and waste treatment costs kept the total yield below 60%, failing to provide a truly scalable solution for the industry.
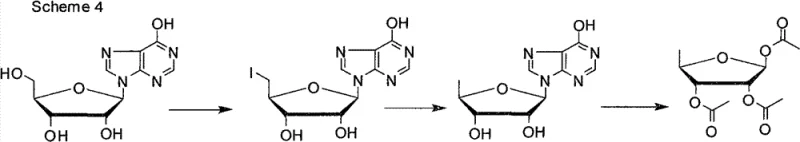
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in CN102260298B leverages a sophisticated three-step sequence that maximizes atom economy and minimizes waste generation. By replacing the problematic iodine reagents with cost-effective chlorinating or brominating agents such as phosphorus tribromide or phosphorus oxychloride, the new route eliminates the formation of persistent organic pollutants and heavy halogen waste. The process begins with a highly selective halogenation of the 5'-hydroxyl group of inosine in an aprotic polar solvent, utilizing sodium tripolyphosphate as an acid binding agent to prevent degradation of the sensitive nucleobase. This is followed by a tandem acetylation and glycosidic bond cleavage step catalyzed by boron trifluoride, which remarkably achieves a beta/alpha anomeric ratio of 95:5, drastically reducing the need for isomeric purification. The final step employs catalytic hydrogenolysis to remove the halogen atom, delivering the target 1,2,3-O-triacetyl-5-deoxy-beta-D-ribose with a total yield exceeding 65% and purity greater than 99.5%.
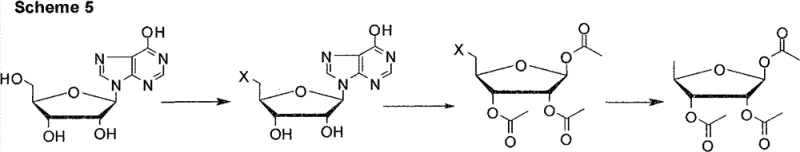
Mechanistic Insights into Selective Halogenation and Catalytic Cleavage
The success of this synthetic strategy lies in the precise control of reaction conditions during the initial functionalization of the inosine scaffold. The use of an aprotic polar solvent like dimethyl sulfoxide (DMSO) is critical, as it solubilizes the polar inosine starting material while facilitating the nucleophilic substitution of the primary hydroxyl group by the phosphorus halide species. The inclusion of an acid binding agent, specifically sodium tripolyphosphate (STPP), plays a dual role: it neutralizes the hydrogen halide generated during the reaction, preventing acid-catalyzed depurination of the nucleoside, and it likely acts as a phase transfer catalyst or complexing agent to enhance the selectivity for the 5'-position over the secondary hydroxyls. This selectivity is paramount, as halogenation at the 2' or 3' positions would lead to irreversible byproducts that cannot be converted into the desired 5-deoxy sugar, thereby preserving the carbon skeleton integrity required for the subsequent steps.
Furthermore, the mechanism of the acetylation and cleavage step represents a significant advancement in carbohydrate chemistry applied to nucleosides. The use of boron trifluoride etherate as a Lewis acid catalyst promotes the formation of an oxocarbenium ion intermediate at the anomeric center, which is then trapped by the acetate nucleophile. The thermodynamic stability of the beta-anomer under these specific acidic conditions drives the equilibrium heavily towards the desired 95:5 ratio, a feat that is difficult to achieve with conventional acetylation reagents like pyridine/acetic anhydride alone. Finally, the catalytic hydro-reduction step utilizes palladium on carbon or Raney nickel to effect a clean reductive dehalogenation. This heterogeneous catalysis ensures that the halogen atom is replaced by hydrogen without affecting the ester protecting groups or the furanose ring structure, resulting in a final product that is chemically pure and ready for the subsequent coupling reactions required to synthesize capecitabine.
How to Synthesize 1,2,3-O-triacetyl-5-deoxy-beta-D-ribose Efficiently
The implementation of this patented process requires careful attention to temperature control and reagent stoichiometry to replicate the high yields reported in the examples. The initial halogenation must be conducted at low temperatures, typically between -5°C and 0°C, to suppress side reactions, before allowing the mixture to warm to room temperature for completion. Following the isolation of the 5'-halo-inosine intermediate, the acetylation step requires reflux conditions in excess acetic anhydride to ensure complete conversion and cleavage of the hypoxanthine base.
- Perform selective halogenation of inosine using phosphorus tribromide or oxychloride in DMSO with an acid binding agent to form 5'-halo-5'-deoxyinosine.
- React the halo-intermediate with acetic anhydride and a boron trifluoride catalyst to achieve simultaneous acetylation and glycosidic bond cleavage.
- Execute catalytic hydro-reduction using palladium on carbon or Raney Nickel to remove the halogen atom and yield the final triacetyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers profound strategic advantages that extend far beyond simple yield improvements. The transition from iodine-based reagents to phosphorus halides results in a drastic simplification of the waste treatment infrastructure, as the effluent no longer contains recalcitrant organo-iodine compounds or phosphine oxides that require specialized disposal methods. This reduction in environmental compliance burden translates directly into lower operational expenditures and reduced risk of regulatory shutdowns, ensuring a more stable supply of this critical intermediate. Moreover, the reliance on inosine, a fermentation-derived commodity chemical, insulates the production process from the volatility of petrochemical-derived sugar prices, providing a more predictable cost structure for long-term budgeting.
- Cost Reduction in Manufacturing: The elimination of expensive iodine and triphenylphosphine reagents, coupled with the high total yield of over 65%, leads to a substantial decrease in raw material costs per kilogram of finished product. By avoiding the formation of difficult-to-remove impurities like triphenylphosphine oxide, the downstream purification process is significantly streamlined, reducing solvent consumption and labor hours associated with chromatography or extensive recrystallization. This efficiency gain allows manufacturers to offer competitive pricing for high-purity pharmaceutical intermediates without sacrificing margin, creating a compelling value proposition for generic drug developers.
- Enhanced Supply Chain Reliability: The robustness of the three-step sequence, characterized by crystalline intermediates rather than oils, ensures consistent batch-to-batch quality and simplifies inventory management. The use of widely available catalysts such as palladium on carbon and standard industrial solvents like DMSO and methylene chloride means that the supply chain is not dependent on niche or single-source reagents that could cause bottlenecks. This resilience is critical for maintaining continuous production schedules for life-saving oncology medications, minimizing the risk of stockouts that could impact patient therapy.
- Scalability and Environmental Compliance: The process is inherently designed for large-scale industrial production, utilizing unit operations such as filtration, distillation, and crystallization that are easily transferred from pilot plant to commercial manufacturing suites. The significant reduction in hazardous waste generation aligns with global sustainability goals and stricter environmental regulations, future-proofing the manufacturing asset against evolving compliance standards. This scalability ensures that the technology can meet the growing global demand for capecitabine as its indications expand and patent exclusivities expire in various markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of this method.
Q: Why is the inosine-based route superior to the traditional D-ribose method?
A: The traditional D-ribose route suffers from low total yields (25-30%) and difficult purification of oily intermediates. The inosine-based approach described in CN102260298B achieves a total yield exceeding 65% with product purity greater than 99.5%, utilizing stable crystalline intermediates that simplify isolation.
Q: How does this patent address the environmental issues of previous inosine methods?
A: Previous inosine routes relied on expensive iodine reagents and triphenylphosphine, generating difficult-to-treat black wastewater containing iodine and phosphine oxides. This novel method utilizes cost-effective chlorinating or brominating agents (like PBr3 or POCl3), eliminating heavy halogen waste and simplifying the three-waste treatment process significantly.
Q: What is the stereochemical selectivity of the acetylation step?
A: By employing acetic anhydride in the presence of a boron trifluoride catalyst, the process achieves a highly favorable beta/alpha anomeric ratio of approximately 95:5. This high stereoselectivity minimizes the formation of unwanted isomers, reducing the burden on downstream purification and increasing overall process efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3-O-triacetyl-5-deoxy-beta-D-ribose Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology drugs relies on the availability of high-quality, cost-effective intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative route described in CN102260298B can be seamlessly integrated into your supply chain. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 1,2,3-O-triacetyl-5-deoxy-beta-D-ribose meets the exacting standards required for API synthesis. Our commitment to process excellence means we can deliver this critical building block with the consistency and reliability that global pharmaceutical partners demand.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can drive value for your specific projects. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the profitability and sustainability of your pharmaceutical manufacturing operations.
