Scalable Synthesis of 3'-Methoxyguanosine for Advanced mRNA Cap Analog Manufacturing
Scalable Synthesis of 3'-Methoxyguanosine for Advanced mRNA Cap Analog Manufacturing
The rapidly evolving landscape of mRNA therapeutics has placed unprecedented demand on the supply chain for high-purity cap analogs, specifically requiring efficient access to modified nucleosides like 3'-methoxyguanosine. Patent CN112661802B, published in May 2022, discloses a groundbreaking synthetic methodology that addresses critical bottlenecks in the production of this essential building block. Unlike traditional approaches that rely on hazardous reagents or biocatalysts with limited scalability, this invention utilizes a chemically robust pathway starting from readily available xylofuranose derivatives. For R&D directors and procurement strategists in the biopharmaceutical sector, this patent represents a pivotal shift towards safer, more cost-effective manufacturing of anti-reverse cap analogs (ARCAs). The disclosed route not only simplifies the stereochemical control required for the 3'-O-methyl modification but also ensures that the final product meets the stringent purity specifications demanded by modern translational medicine applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 3'-methoxyguanosine was fraught with significant safety and economic challenges that hindered its widespread commercial adoption. As illustrated in the background art, conventional methods typically initiated from guanosine, necessitating a complex transformation into 2-amino adenosine followed by methylation using diazomethane. This reliance on diazomethane is particularly problematic for industrial settings due to its extreme toxicity, explosive nature, and the substantial regulatory burden associated with its handling and storage. Furthermore, the methylation step often resulted in a mixture of 2'-O and 3'-O methylated products with very similar polarities, making separation arduous and yield-limiting. The final conversion step required adenosine deaminase, an expensive biocatalyst that introduces variability and complicates the supply chain continuity. These factors collectively rendered previous methods unsuitable for the multi-kilogram or ton-scale production required by the booming mRNA vaccine and therapeutic market.
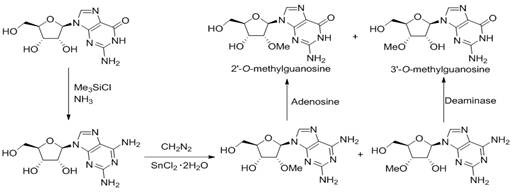
The Novel Approach
In stark contrast, the methodology described in CN112661802B circumvents these hazards by adopting a carbohydrate engineering strategy that builds the nucleoside from a protected sugar precursor rather than modifying a complete nucleoside. This approach begins with 1,2-O-isopropylidene-alpha-D-xylofuranose, a stable and inexpensive starting material. By selectively protecting the primary alcohol and then manipulating the secondary hydroxyl groups through oxidation and reduction, the synthesis achieves the necessary stereochemical inversion without generating toxic byproducts. The subsequent methylation is performed on a simplified sugar intermediate, avoiding the polarity issues associated with nucleoside methylation. Finally, the glycosidic bond is formed via coupling with N2-acetylguanine under controlled conditions. This linear, fully chemical synthesis offers a predictable, scalable, and safe alternative that aligns perfectly with the needs of a reliable pharmaceutical intermediates supplier seeking to optimize their portfolio.
Mechanistic Insights into Stereoselective Sugar Modification and Coupling
The core innovation of this patent lies in the precise manipulation of the furanose ring to establish the correct 3'-methoxy configuration. The process initiates with the selective silylation of the primary hydroxyl group using tert-butyldiphenylchlorosilane (TBDPSCl), which provides robust protection against subsequent reaction conditions. The pivotal mechanistic step involves the oxidation of the C-3' secondary alcohol to a ketone using Swern oxidation conditions (oxalyl chloride/DMSO), followed by a stereoselective reduction using sodium borohydride. This oxidation-reduction sequence effectively inverts the stereochemistry at the C-3' position, converting the xylo-configuration to the desired ribo-configuration required for the final product. This chemical inversion is far more controllable than enzymatic methods and avoids the formation of inseparable isomers. Following inversion, the hydroxyl group is methylated using methyl iodide and sodium hydride, locking in the 3'-O-methyl motif before the nucleobase is introduced.
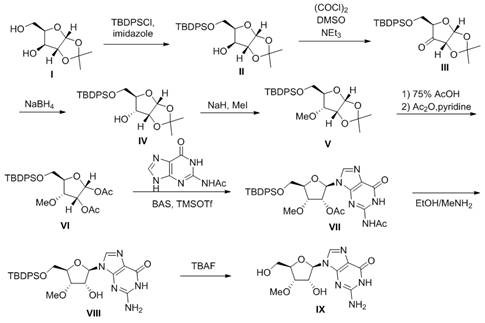
The final stage of the synthesis involves the coupling of the modified sugar intermediate with N2-acetylguanine. This step utilizes bis-trimethylsilyl acetamide (BSA) to activate the nucleobase and trimethylsilyl trifluoromethanesulfonate (TMSOTf) as a Lewis acid promoter to facilitate the formation of the N-glycosidic bond. The use of acetyl protecting groups on the guanine base ensures regioselectivity during coupling and prevents side reactions. The mechanism proceeds through the formation of an oxocarbenium ion intermediate, which is attacked by the silylated nucleobase to form the beta-anomer predominantly. Subsequent deprotection steps remove the acetyl and silyl groups under mild conditions, yielding the target 3'-methoxyguanosine with high purity. This detailed understanding of the reaction pathway allows process chemists to fine-tune conditions for maximum yield and minimal impurity generation, ensuring a consistent supply of high-purity pharmaceutical intermediates.
How to Synthesize 3'-Methoxyguanosine Efficiently
The execution of this synthetic route requires careful attention to reaction conditions, particularly during the oxidation and coupling stages, to maintain high stereochemical integrity. The patent outlines a clear eight-step protocol that transitions from simple sugar protection to complex nucleoside assembly. Operators must ensure anhydrous conditions during the silylation and methylation steps to prevent hydrolysis of sensitive intermediates. The oxidation step requires strict temperature control at -60°C to avoid over-oxidation or side reactions, while the reduction step benefits from a mixed solvent system of ethanol and water to facilitate the stereoselective delivery of hydride. For a comprehensive guide on executing this process in a GMP environment, please refer to the standardized operating procedures below.
- Selective protection of the primary alcohol of 1,2-O-isopropylidene-alpha-D-xylofuranose using TBDPSCl to form intermediate II.
- Oxidation of the secondary alcohol to a ketone (III) followed by stereoselective reduction with NaBH4 to invert configuration to alcohol IV.
- Methylation of the inverted alcohol, deprotection, acetylation, coupling with N2-acetylguanine, and final global deprotection to yield 3'-methoxyguanosine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic advantages beyond mere technical feasibility. The elimination of diazomethane removes a major safety liability and reduces the need for specialized explosion-proof infrastructure, leading to substantial cost savings in facility operations and insurance. Furthermore, replacing expensive enzymatic steps with standard chemical reagents significantly lowers the raw material costs and mitigates the risk of supply disruptions associated with biological catalysts. The simplicity of the purification processes, primarily relying on standard column chromatography and recrystallization, enhances throughput and reduces solvent consumption, contributing to a more sustainable and economically viable manufacturing model. These factors collectively position this method as a superior choice for long-term sourcing strategies.
- Cost Reduction in Manufacturing: The replacement of hazardous and expensive reagents with commodity chemicals drastically reduces the direct material costs associated with production. By avoiding the use of adenosine deaminase and diazomethane, manufacturers can eliminate the high procurement costs and waste disposal fees linked to these specialized substances. Additionally, the improved yield and simplified purification steps minimize material loss, further driving down the cost per kilogram of the final API intermediate. This economic efficiency allows for more competitive pricing models in the global market for mRNA components.
- Enhanced Supply Chain Reliability: Utilizing a fully chemical synthesis route decouples production from the volatility of the biocatalyst market, ensuring a more stable and predictable supply chain. The starting materials, such as xylofuranose derivatives and standard silylating agents, are widely available from multiple global vendors, reducing the risk of single-source dependency. This diversification of the supply base enhances resilience against geopolitical or logistical disruptions, guaranteeing continuous availability of critical nucleoside intermediates for downstream drug manufacturing.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals or explosive gases make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The reduced environmental footprint, achieved through lower solvent usage and the avoidance of hazardous waste streams, aligns with increasingly stringent global environmental regulations. This compliance facilitates faster regulatory approvals and smoother audits, accelerating the time-to-market for new therapeutic candidates relying on this cap analog technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing frameworks. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: Why is the new synthetic route for 3'-methoxyguanosine safer than previous methods?
A: The novel route eliminates the use of highly toxic and explosive diazomethane and expensive enzymatic processes found in prior art, utilizing standard chemical reagents instead.
Q: What is the key stereochemical strategy used in this synthesis?
A: The process employs an oxidation-reduction sequence on the xylofuranose scaffold to invert the configuration of the C-3' hydroxyl group, enabling precise methylation.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights mild reaction conditions, simple purification via column chromatography or recrystallization, and high yields suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3'-Methoxyguanosine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality cap analogs play in the success of next-generation mRNA therapies. Our team of expert process chemists has thoroughly analyzed the methodology disclosed in CN112661802B and is fully prepared to implement this advanced synthesis route at commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3'-methoxyguanosine delivers the performance required for your research and development initiatives.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive value and efficiency for your organization.
