Scalable Manufacturing of 3'-Methoxyguanosine via Stereoselective Carbohydrate Engineering
Scalable Manufacturing of 3'-Methoxyguanosine via Stereoselective Carbohydrate Engineering
The rapid advancement of mRNA therapeutics has placed an unprecedented demand on the supply chain for high-purity cap analogs, specifically 3'-methoxyguanosine, which serves as a critical building block for Anti-Reverse Cap Analogs (ARCA). Patent CN112661802B, published in May 2022, discloses a groundbreaking synthetic methodology that addresses the long-standing bottlenecks in producing this valuable pharmaceutical intermediate. Unlike traditional routes that rely on hazardous reagents and complex enzymatic steps, this novel approach leverages a rational carbohydrate engineering strategy starting from 1,2-O-isopropylidene-alpha-D-xylofuranose. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this patent represents a paradigm shift towards safer, more economical, and scalable manufacturing processes that can support the burgeoning needs of the biotechnology sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3'-methoxyguanosine has been fraught with significant safety hazards and purification challenges that render it unsuitable for modern GMP manufacturing. As illustrated in the prior art, conventional methods typically initiate from guanosine, requiring a conversion to 2-amino adenosine followed by methylation using diazomethane. This reliance on diazomethane is a critical liability; it is an extremely toxic, carcinogenic, and explosive gas that poses severe risks to personnel and requires specialized, costly infrastructure to handle safely. Furthermore, the methylation step lacks regioselectivity, generating a mixture of 2'-O-methyl and 3'-O-methyl isomers that possess very similar polarities, making their separation via chromatography notoriously difficult and yield-limiting. Finally, the subsequent conversion of the amino group back to the carbonyl functionality often necessitates the use of adenosine deaminase, an expensive enzymatic catalyst that adds substantial cost and complexity to the supply chain, effectively pricing out large-scale commercial application.
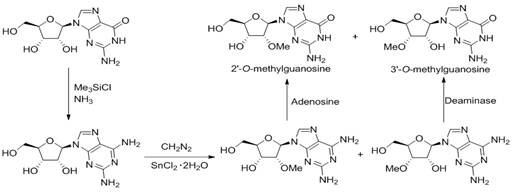
The Novel Approach
In stark contrast, the methodology outlined in CN112661802B circumvents these pitfalls by constructing the target molecule from a simpler sugar precursor rather than modifying a complex nucleoside. This bottom-up approach allows for precise control over stereochemistry and functional group installation before the nucleobase is even introduced. By starting with 1,2-O-isopropylidene-alpha-D-xylofuranose, the synthesis strategically inverts the configuration at the C-3' position through a robust oxidation-reduction sequence, thereby installing the hydroxyl group in the exact orientation required for the final 3'-O-methyl product. This eliminates the formation of difficult-to-separate regioisomers entirely. Moreover, the methylation is performed on a protected sugar intermediate using standard alkylating agents like methyl iodide, completely removing the need for dangerous diazomethane. The final coupling with N2-acetylguanine is achieved under mild Lewis acid conditions, ensuring high yields and simplifying the downstream purification process, which is a decisive advantage for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Stereoselective Carbohydrate Modification
The core innovation of this synthetic route lies in its elegant manipulation of the furanose ring stereochemistry. The process begins with the selective protection of the primary hydroxyl group at the C-5' position using tert-butyldiphenylchlorosilane (TBDPSCl) in the presence of imidazole. This bulky silyl group not only protects the primary alcohol but also imparts significant steric bulk that influences subsequent reactions. The pivotal mechanistic step involves the oxidation of the secondary C-3' hydroxyl group to a ketone using Swern oxidation conditions (oxalyl chloride and DMSO). This transient ketone intermediate destroys the original stereocenter, allowing for a thermodynamic or kinetic reset. Upon reduction with sodium borohydride in an ethanol-water mixture, the hydride attack occurs preferentially from the less hindered face, effectively inverting the configuration from the xylo-series to the ribo-series. This stereochemical inversion is the key to accessing the 3'-position with the correct geometry for methylation, demonstrating a sophisticated understanding of conformational analysis in carbohydrate chemistry.
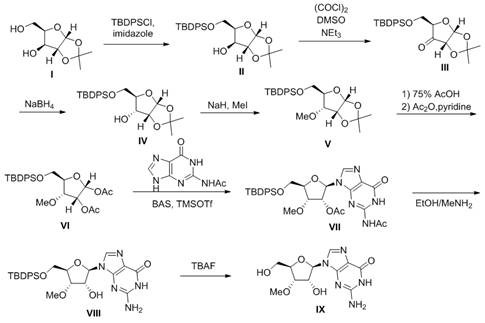
Following the stereochemical setup, the methylation step utilizes sodium hydride as a strong base to deprotonate the newly formed C-3' hydroxyl group, followed by quenching with methyl iodide. This SN2 reaction proceeds cleanly due to the absence of competing hydroxyl groups, which have been either protected or are sterically inaccessible. The subsequent removal of the acetal protecting group using aqueous acetic acid, followed by acetylation, prepares the sugar for nucleophilic substitution. The coupling reaction with N2-acetylguanine is catalyzed by trimethylsilyl trifluoromethanesulfonate (TMSOTf), a potent Lewis acid that activates the anomeric acetate for glycosylation. This Vorbrüggen-type coupling ensures the formation of the beta-N9-glycosidic bond with high fidelity. Finally, the global deprotection strategy employs methylamine to remove the acetyl groups and tetrabutylammonium fluoride (TBAF) to cleave the silyl ether, yielding the target 3'-methoxyguanosine with exceptional purity, addressing the critical杂质谱 concerns of R&D teams.
How to Synthesize 3'-Methoxyguanosine Efficiently
The execution of this synthesis requires careful attention to reaction conditions, particularly during the oxidation and coupling stages, to maximize yield and minimize byproduct formation. The protocol detailed in the patent provides a robust framework that has been optimized for reproducibility, utilizing common solvents like dichloromethane and DMF which are easily sourced and recycled in an industrial setting. The stepwise progression from the protected xylofuranose to the final nucleoside allows for intermediate quality control checks, ensuring that any deviations in stereochemistry are caught early before valuable reagents are consumed. For process chemists looking to implement this technology, the following standardized workflow outlines the critical operational parameters derived directly from the patent examples, ensuring a smooth transition from benchtop discovery to pilot plant operations.
- Selectively protect the primary alcohol of the starting xylofuranose derivative using TBDPSCl and imidazole to form the silyl ether intermediate.
- Perform Swern oxidation to convert the secondary alcohol to a ketone, followed by stereoselective reduction with NaBH4 to invert the configuration to the ribo-form.
- Methylate the inverted secondary hydroxyl group using NaH and MeI, then remove the acetal protecting group and acetylate the remaining hydroxyls.
- Couple the sugar intermediate with N2-acetylguanine using BSA and TMSOTf, followed by deprotection of acetyl and silyl groups to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits that extend far beyond simple yield improvements. The elimination of diazomethane removes a major regulatory and safety hurdle, significantly lowering the barrier to entry for manufacturing facilities that cannot support high-hazard operations. This shift inherently reduces the capital expenditure required for specialized containment systems and waste treatment protocols associated with explosive gases. Furthermore, by replacing expensive enzymatic catalysts with commodity chemicals like methyl iodide and sodium borohydride, the raw material costs are drastically simplified and stabilized against market volatility. The ability to source these reagents from multiple global suppliers enhances supply chain resilience, mitigating the risk of single-source bottlenecks that often plague biotech manufacturing timelines.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the removal of high-cost biological catalysts and the simplification of purification workflows. Traditional methods often suffer from low overall yields due to the difficulty in separating regioisomers, leading to significant material loss and increased cost per gram. By contrast, this novel route achieves high regioselectivity through stereochemical design rather than separation, meaning that a much larger proportion of the starting material is converted into the desired product. The use of robust chemical reagents instead of sensitive enzymes also extends the shelf-life of inventory and reduces the need for cold-chain logistics, contributing to substantial cost savings in storage and handling.
- Enhanced Supply Chain Reliability: Dependence on specialized enzymes like adenosine deaminase introduces a single point of failure in the supply chain, as these biologics can vary in activity between batches and have limited availability. This new chemical synthesis relies entirely on small-molecule reagents that are produced at massive scales for various industries, ensuring consistent availability and pricing. The robustness of the chemical steps, such as the silyl protection and Swern oxidation, allows for flexible scheduling and batch sizing, enabling manufacturers to respond rapidly to fluctuating demand from mRNA vaccine and therapeutic developers without compromising on lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior. The avoidance of diazomethane eliminates the generation of toxic nitrogenous waste streams, simplifying effluent treatment and reducing the environmental footprint of the manufacturing process. The solvents used, such as ethyl acetate and ethanol, are greener alternatives to the chlorinated solvents often required in older nucleoside syntheses. This alignment with green chemistry principles facilitates easier regulatory approval and permits for commercial scale-up of complex pharmaceutical intermediates, ensuring long-term operational continuity in an increasingly regulated global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived from the specific experimental data and comparative advantages highlighted in patent CN112661802B, providing clarity for stakeholders evaluating this process for potential licensing or procurement partnerships. Understanding these nuances is essential for making informed decisions about integrating this methodology into existing production pipelines.
Q: Why is the new synthetic route for 3'-methoxyguanosine safer than previous methods?
A: The novel route described in patent CN112661802B eliminates the use of highly toxic and explosive diazomethane, which was required in traditional methylation steps. Additionally, it avoids the use of expensive and unstable enzymatic catalysts like adenosine deaminase, replacing them with robust chemical reagents suitable for industrial scale-up.
Q: How does this process ensure high stereochemical purity?
A: The process utilizes a precise oxidation-reduction sequence on a protected xylofuranose scaffold. By oxidizing the C-3' hydroxyl to a ketone and subsequently reducing it with sodium borohydride, the stereochemistry is inverted from the xylo-configuration to the desired ribo-configuration with high selectivity, ensuring the correct 3'-O-methyl orientation.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the method is explicitly designed for scalability. It employs common, inexpensive reagents such as tert-butyldiphenylchlorosilane and methyl iodide, and avoids difficult chromatographic separations of polar isomers that plagued earlier methods, thereby facilitating cost-effective manufacturing at the metric ton scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3'-Methoxyguanosine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated stereochemical controls described in this patent are maintained at every stage of growth. We operate stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3'-methoxyguanosine meets the exacting standards required for mRNA cap analog synthesis, providing our clients with the confidence needed to advance their therapeutic programs.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this safer, more efficient methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the development of next-generation RNA therapeutics.
