Scalable Synthesis of Broflanilide Intermediates via Optimized Meta-Diamide Pathway
Introduction to Next-Generation Meta-Diamide Insecticides
The global agrochemical sector is witnessing a paradigm shift towards meta-diamide insecticides, driven by the urgent need to combat pest resistance against established ortho-diamides like chlorantraniliprole. At the forefront of this innovation is Broflanilide, a compound with a unique mode of action targeting GABA-gated chloride channels, offering superior efficacy against lepidoptera and coleoptera pests. The technical feasibility of producing these complex molecules at scale was recently advanced by the disclosure in patent CN112707836A, which outlines a robust preparation method for m-diamide compounds. This patent addresses critical bottlenecks in existing manufacturing processes, proposing a streamlined five-step synthesis that achieves near-quantitative yields while eliminating the need for extreme cryogenic conditions. For R&D directors and supply chain strategists, understanding this pathway is essential for securing a reliable agrochemical intermediate supplier capable of meeting future demand.
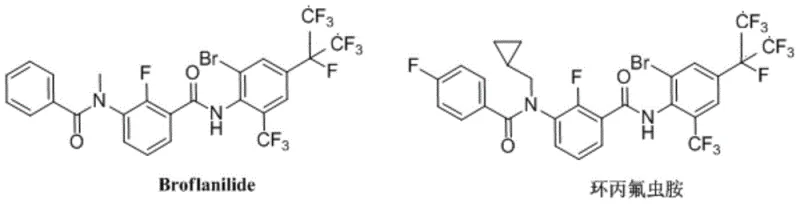
The structural versatility of this class allows for analogues such as Cyflumetofen, which shares the core scaffold but varies in substituents to optimize biological activity. The patent details a unified strategy to access both Broflanilide and its analogues, emphasizing the importance of late-stage functionalization. By deferring the introduction of the bromine atom to the final step, the synthesis minimizes side reactions and simplifies the purification of sensitive intermediates. This approach not only enhances the overall process mass intensity (PMI) but also aligns with green chemistry principles by reducing solvent usage and waste generation. As we delve into the technical specifics, it becomes clear that this methodology represents a significant leap forward in cost reduction in agrochemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industrial synthesis of Broflanilide was plagued by three distinct and problematic routes, each presenting severe scalability challenges. The first reported route suffered from excessive length and poor atom economy, requiring at least 2.4 equivalents of acid relative to amine, which necessitated complex hydrolysis and acid recovery operations. Furthermore, this method generated difficult-to-remove impurities during the condensation of acyl chloride with amine, and the liquid-liquid separation post-hydrolysis was notoriously difficult due to deep coloration and emulsion formation, resulting in a single-step yield of only approximately 75%. 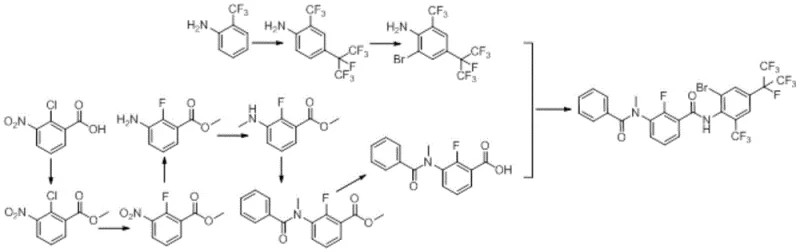
The second route introduced different inefficiencies, particularly in the construction of the heptafluoroisopropyl group, where yields plummeted to merely 63%, creating a significant bottleneck for material throughput. Additionally, this pathway relied on N-bromosuccinimide (NBS) for the final bromination, a reagent that is not only costly but also generates succinimide waste that complicates downstream purification. The third route attempted to utilize lithium diisopropylamide (LDA) for condensation, which mandated deep cooling to -70°C. Such cryogenic requirements are energetically prohibitive on a commercial scale and resulted in a dismal single-step yield of 34%, rendering the process economically unviable for large-scale production.
The Novel Approach
In stark contrast, the methodology disclosed in CN112707836A offers a streamlined, five-step sequence that circumvents these historical hurdles through intelligent reaction design. The new route initiates with a highly efficient condensation between 2-fluoro-3-nitrobenzoyl chloride and 4-(perfluoropropane-2-yl)-2-(trifluoromethyl)aniline, catalyzed by DMAP in toluene at moderate temperatures of 110-140°C. This eliminates the need for cryogenic LDA chemistry while achieving yields exceeding 96%. The subsequent steps involve catalytic hydrogenation, selective alkylation, acylation, and a final regioselective bromination, all optimized for high conversion and ease of isolation. 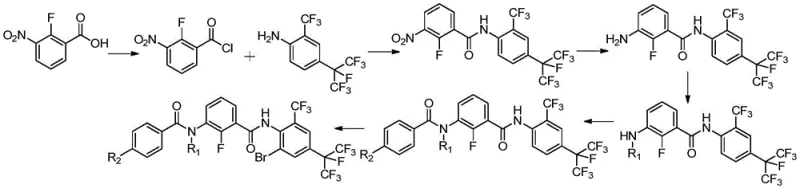
A defining feature of this novel approach is the strategic placement of the bromination step at the very end of the synthesis. By introducing the bromine atom last, the process avoids potential side reactions on the aromatic ring during earlier transformations, thereby preserving the integrity of the sensitive fluoro- and trifluoromethyl-substituted scaffolds. The patent reports that multi-step reactions are almost quantitative with few byproducts, allowing for high-purity products to be obtained via simple solvent separation or recrystallization without the need for column chromatography. This operational simplicity translates directly into enhanced supply chain reliability and reduced lead time for high-purity agrochemical intermediates.
Mechanistic Insights into the Five-Step Cascade Synthesis
The core of this technological breakthrough lies in the precise control of reaction conditions across the five-step cascade, ensuring that each transformation proceeds with maximal efficiency. The initial condensation leverages the nucleophilicity of the aniline derivative, activated by the electron-withdrawing perfluoroalkyl groups, to attack the acyl chloride. The use of DMAP as a nucleophilic catalyst accelerates this acylation, while the choice of toluene or xylene as a solvent facilitates the removal of water and byproducts through azeotropic distillation or hot filtration. Following this, the reduction of the nitro group to an amine is achieved using heterogeneous catalysts like 10% Pd/C or Pt/C under hydrogen pressures of 1.5 to 3.0 MPa. This catalytic hydrogenation is preferred over chemical reduction (e.g., iron/acid) as it generates no heavy metal sludge, aligning with stringent environmental compliance standards.
The alkylation step demonstrates remarkable chemoselectivity, utilizing either formaldehyde or cyclopropanecarboxaldehyde to install the N-alkyl group. When formaldehyde is used, concentrated sulfuric acid acts as both solvent and catalyst, promoting the formation of the iminium ion intermediate which is subsequently reduced or rearranged to the secondary amine. Alternatively, for the cyclopropyl analogue, zinc powder in acetic acid provides a mild reducing environment that preserves the cyclopropane ring from acid-catalyzed ring opening. The final acylation and bromination steps are equally rigorous; the acylation employs benzoyl chloride derivatives under DMAP catalysis to form the diamide backbone, while the bromination utilizes an in situ generated electrophilic bromine species from sodium bromide and an oxidant like sodium chlorate. This ensures specific substitution at the desired position without polybromination, a common issue in electrophilic aromatic substitution of activated rings.
How to Synthesize Broflanilide Efficiently
Implementing this synthesis requires strict adherence to the thermal and stoichiometric parameters outlined in the patent to ensure reproducibility and safety. The process begins with the activation of 2-fluoro-3-nitrobenzoic acid using thionyl chloride or triphosgene, followed by immediate coupling with the fluorinated aniline. Operators must maintain the reaction temperature between 110°C and 140°C to drive the equilibrium forward while preventing thermal decomposition of the nitro group. Subsequent hydrogenation demands careful pressure control within the 2.0 to 2.5 MPa range to balance reaction rate with safety margins in the autoclave. The detailed standardized synthesis steps, including specific workup procedures like hot filtration at 80°C to remove catalyst and impurities, are critical for achieving the reported 99% purity specifications.
- Condense 2-fluoro-3-nitrobenzoyl chloride with 4-(perfluoropropane-2-yl)-2-(trifluoromethyl)aniline using DMAP catalyst in toluene at 110-140°C.
- Perform catalytic hydrogenation on the nitroamide intermediate using Pd/C or Pt/C under 1.5-3.0 MPa hydrogen pressure at 40-100°C.
- Conduct alkylation using formaldehyde or cyclopropanecarboxaldehyde in the presence of acid catalysts (H2SO4 or Zn/AcOH) to introduce the N-alkyl group.
- React the alkylamino intermediate with benzoyl chloride derivatives (Formula II) using DMAP to form the bisamide structure.
- Execute the final regioselective bromination using sodium bromide and an oxidizing agent (e.g., sodium chlorate) to obtain the target meta-diamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits that extend beyond mere technical elegance. The primary advantage lies in the drastic simplification of the purification workflow. By eliminating the need for column chromatography and replacing it with crystallization and hot filtration, the process significantly reduces solvent consumption and processing time. This reduction in unit operations directly correlates to lower utility costs and higher throughput capacity, enabling manufacturers to respond more agilely to market fluctuations. Furthermore, the avoidance of expensive reagents like NBS and cryogenic reagents like LDA results in substantial cost savings in raw material procurement, making the final active ingredient more price-competitive in the global marketplace.
- Cost Reduction in Manufacturing: The elimination of deep cooling requirements (-70°C) removes the need for specialized cryogenic equipment and the high energy consumption associated with maintaining such low temperatures. Additionally, replacing NBS with commodity chemicals like sodium bromide and sodium chlorate drastically lowers reagent costs. The high yields reported (often >90% per step) minimize raw material waste, ensuring that a greater proportion of input costs are converted into saleable product, thereby optimizing the overall cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 2-fluoro-3-nitrobenzoic acid and common solvents like toluene and ethyl acetate mitigates the risk of supply disruptions. Unlike routes dependent on specialized fluorinating agents or unstable intermediates, this pathway utilizes robust chemistry that can be sourced from multiple vendors. The simplified workup procedures also reduce the dependency on highly skilled labor for complex purifications, ensuring consistent production quality even during periods of workforce fluctuation.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, avoiding the heat transfer limitations associated with cryogenic reactions. The use of catalytic hydrogenation instead of stoichiometric metal reductions reduces the generation of hazardous solid waste, simplifying effluent treatment. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site, a critical factor for multinational corporations adhering to strict ESG (Environmental, Social, and Governance) mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this meta-diamide synthesis pathway. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer or licensing agreements.
Q: How does the new bromination method improve upon traditional NBS usage?
A: The patented method utilizes sodium bromide combined with oxidizing agents like sodium chlorate or hypochlorite, avoiding the expensive N-bromosuccinimide (NBS) reagent used in prior art. This allows for precise introduction of bromine atoms at specific sites in the final step with high yield, significantly reducing reagent costs and simplifying waste treatment compared to conventional radical bromination.
Q: What purification techniques ensure high purity without complex chromatography?
A: The process relies on strategic 'hot filtration' and solvent crystallization rather than column chromatography. For instance, after the condensation step, the mixture is stirred at 80°C with alkali solution, separated while hot to prevent precipitation, and then cooled to 0-5°C for crystallization. This physical separation method effectively removes impurities and unreacted starting materials, ensuring high-purity intermediates suitable for direct use in subsequent steps.
Q: Why is this route considered more scalable than previous methods involving LDA?
A: Previous routes often required lithium diisopropylamide (LDA) and deep cooling conditions down to -70°C, which are energy-intensive and difficult to maintain on a multi-ton scale. This new protocol operates at moderate temperatures (40-140°C) and avoids cryogenic conditions entirely. The elimination of deep cooling and the use of robust catalytic hydrogenation make the process inherently safer and easier to scale for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Broflanilide Supplier
As the demand for next-generation insecticides continues to surge, partnering with a manufacturer that possesses deep technical expertise in complex heterocyclic synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific thermal and pressure requirements of this five-step synthesis, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that the transition from lab-scale discovery to commercial reality requires not just chemical knowledge, but engineering excellence.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this optimized route. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-purity Broflanilide and Cyflumetofen intermediates reliably and efficiently, securing your supply chain for the future.
