Advanced Asymmetric Hydrogenation Of Cyclic N-Alkylimines For Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to access high-purity chiral building blocks, which serve as the foundational scaffolds for numerous bioactive compounds. Patent CN102304007B discloses a groundbreaking method for the asymmetric catalytic hydrogenation of cyclic N-alkylimines, offering a robust pathway to generate endocyclic chiral amines with exceptional stereocontrol. This technology addresses the critical demand for reliable pharmaceutical intermediate supplier capabilities by utilizing chiral diamine metal catalysts that operate under relatively mild conditions. The invention specifically targets the synthesis of structurally diverse cyclic amines, which are prevalent in drugs treating cardiovascular diseases, central nervous system disorders, and tobacco dependence. By leveraging transition metals such as Ruthenium, Rhodium, or Iridium coordinated with monosulfonyl-substituted chiral diamine ligands, the process achieves enantiomeric excess values reaching up to 98% ee. This level of precision is paramount for regulatory compliance in drug manufacturing, where impurity profiles must be strictly controlled to ensure patient safety and efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing chiral cyclic amines often rely on classical resolution techniques or stoichiometric chiral auxiliaries, which are inherently inefficient and generate substantial chemical waste. In many legacy processes, the cyclic imine intermediates are unstable and prone to hydrolysis or polymerization, necessitating complex isolation and purification steps that drastically reduce overall yield. Furthermore, conventional catalytic systems frequently suffer from catalyst poisoning by the basic amine products formed during the reaction, leading to incomplete conversion and requiring excessive catalyst loading to drive the reaction to completion. These inefficiencies translate into higher production costs and longer lead times, creating bottlenecks for procurement managers seeking cost reduction in pharmaceutical intermediate manufacturing. The inability to perform these reactions in a telescoped manner often means multiple unit operations are required, increasing the capital expenditure and operational complexity associated with scaling these processes to commercial volumes.
The Novel Approach
The methodology outlined in the patent introduces a transformative approach by enabling the direct asymmetric hydrogenation of cyclic N-alkylimines using highly active chiral metal complexes. A key innovation is the implementation of a one-pot reductive amination strategy, where the imine is generated in situ from a Boc-protected amino ketone and subsequently hydrogenated without isolation. This seamless integration of steps minimizes handling of sensitive intermediates and significantly streamlines the workflow. The use of additives such as di-tert-butyl dicarbonate plays a crucial role in this novel approach by trapping the amine product as a carbamate, effectively shielding the catalyst from deactivation. This strategic modification allows the reaction to proceed with high turnover numbers and maintains high enantioselectivity even at lower catalyst loadings. Consequently, this approach offers a viable solution for the commercial scale-up of complex pharmaceutical intermediates, providing a competitive edge through improved process mass intensity and reduced environmental footprint.
Mechanistic Insights into Chiral Diamine Metal-Catalyzed Hydrogenation
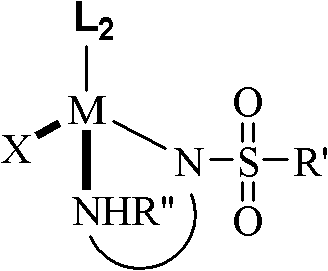
The catalytic cycle operates through a sophisticated metal-ligand cooperative mechanism, typical of Noyori-type asymmetric hydrogenations, where the chirality of the final product is dictated by the stereogenic centers on the diamine ligand backbone. The active catalyst species features a metal center, preferably Ruthenium, coordinated with an eta-6-arene ligand and a chiral monosulfonyl diamine. The mechanism involves the heterolytic cleavage of molecular hydrogen across the metal-amido bond, generating a metal-hydride and an ammonium species that simultaneously transfer a proton and a hydride to the C=N double bond of the substrate. This outer-sphere mechanism avoids direct coordination of the substrate to the metal, which is beneficial for sterically hindered cyclic imines. The sulfonyl group on the ligand enhances the acidity of the NH proton, facilitating the hydrogen activation step and stabilizing the transition state through hydrogen bonding interactions. Understanding these electronic and steric nuances is essential for R&D directors aiming to optimize reaction parameters for new substrate classes.
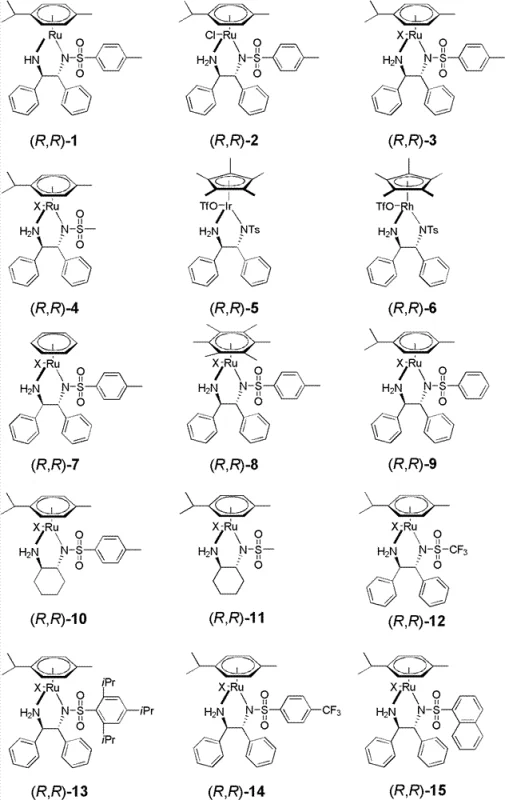
Impurity control is intrinsically built into the catalyst design, as the rigid chiral pocket created by the bulky aryl groups on the diamine ligand prevents the formation of the undesired enantiomer. The patent data indicates that varying the counter-anion, such as using tetraarylborate or phosphate anions, can further tune the electrophilicity of the metal center, thereby influencing both reaction rate and selectivity. For instance, catalysts with bulky non-coordinating anions like BArF4 demonstrated superior performance in terms of enantiomeric excess compared to simple halide salts. This tunability allows chemists to tailor the catalyst system to specific substrate electronic properties, ensuring consistent quality across different batches. By minimizing side reactions such as over-reduction or racemization, the process ensures a clean impurity profile, which simplifies downstream purification and reduces the burden on quality control laboratories during commercial production.
How to Synthesize Endocyclic Chiral Amines Efficiently
The synthesis protocol described in the patent provides a standardized framework for producing high-value chiral amines suitable for industrial application. The process begins with the preparation of the chiral catalyst, followed by the setup of the hydrogenation reaction in a pressure vessel. Detailed operational parameters regarding solvent choice, hydrogen pressure, and temperature are critical to replicating the high yields and selectivity reported in the examples. The following guide summarizes the key procedural steps derived from the patent specifications to assist technical teams in implementing this technology.
- Prepare the chiral metal catalyst by reacting a monosulfonyl chiral diamine ligand with a metal precursor such as [RuCl2(p-cymene)]2 in the presence of a base.
- In a high-pressure reactor, combine the cyclic N-alkylimine substrate, the chiral catalyst, and an additive like di-tert-butyl dicarbonate in a solvent such as dichloromethane.
- Charge the reactor with hydrogen gas to a pressure of 50 atm and maintain the temperature at 40°C for approximately 10 hours to achieve high conversion and enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric hydrogenation technology presents significant opportunities for optimizing the supply of critical chiral intermediates. The ability to utilize a one-pot synthesis route fundamentally alters the cost structure of manufacturing by eliminating intermediate isolation steps, which are often the most resource-intensive parts of a chemical process. This reduction in unit operations directly correlates to lower utility consumption, reduced solvent usage, and decreased labor requirements, driving substantial cost savings in pharmaceutical intermediate manufacturing without compromising on product quality. Furthermore, the robustness of the catalyst system under moderate temperatures and pressures enhances process safety and reliability, reducing the risk of unplanned downtime due to equipment failure or hazardous conditions.
- Cost Reduction in Manufacturing: The elimination of expensive chromatographic purification steps for unstable imine intermediates leads to a drastic simplification of the production workflow. By converting crude reaction mixtures directly into protected amine products, the process minimizes material loss and reduces the consumption of silica gel and eluents. This efficiency gain allows for a more competitive pricing structure for the final chiral amine products, making it an attractive option for large-scale API production where margin compression is a constant challenge.
- Enhanced Supply Chain Reliability: The use of readily available starting materials such as Boc-protected amino ketones and common transition metal precursors ensures a stable supply of raw materials. The catalyst system's tolerance to various functional groups means that a single catalytic platform can be adapted for multiple product lines, reducing the need for specialized inventory. This flexibility enhances supply chain resilience, allowing manufacturers to respond quickly to fluctuations in market demand for different chiral building blocks without retooling entire production lines.
- Scalability and Environmental Compliance: The reaction conditions, operating at temperatures around 40°C and hydrogen pressures of 50 atm, are well within the capabilities of standard industrial hydrogenation reactors, facilitating easy scale-up from pilot to commercial tonnage. Additionally, the high atom economy of hydrogenation reactions combined with the reduced solvent waste from the one-pot procedure aligns with green chemistry principles. This environmental compatibility simplifies regulatory approvals and waste disposal logistics, ensuring long-term sustainability and compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of the one-pot method described in the patent?
A: The one-pot method eliminates the need for isolating and purifying the unstable cyclic N-alkylimine intermediate, significantly reducing processing time and material loss while maintaining high enantiomeric excess.
Q: Which metal centers are most effective for this asymmetric hydrogenation?
A: The patent identifies Ruthenium (Ru), Rhodium (Rh), and Iridium (Ir) as effective metal centers, with specific Ruthenium complexes showing superior enantioselectivity up to 98% ee.
Q: How does the additive improve the reaction efficiency?
A: Additives like di-tert-butyl dicarbonate capture the amine product in situ, preventing it from poisoning the catalyst and thereby sustaining high catalytic activity and selectivity throughout the reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclic N-Alkylimines Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced catalytic technologies to deliver high-purity chiral intermediates to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in laboratory patents are successfully translated into robust manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral amine meets the exacting standards required by top-tier pharmaceutical companies. Our commitment to quality and consistency makes us a trusted partner for organizations looking to secure their supply of critical chiral building blocks.
We invite you to engage with our technical procurement team to discuss how this asymmetric hydrogenation technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalytic route for your target molecules. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence and our proven track record in process development and commercial manufacturing.
