Advanced Synthesis of Polysubstituted Thiopyran Derivatives for Commercial Scale-Up and Drug Discovery
Advanced Synthesis of Polysubstituted Thiopyran Derivatives for Commercial Scale-Up and Drug Discovery
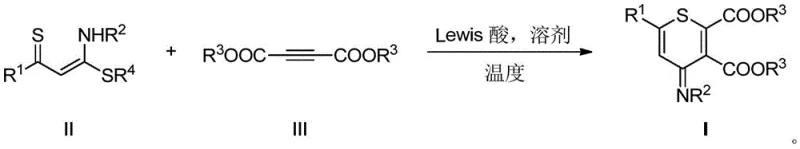
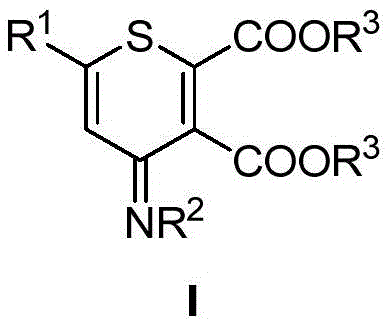
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. A recent technological breakthrough documented in patent CN114380790B introduces a highly efficient route for the synthesis of polysubstituted thiopyran derivatives, a class of six-membered sulfur-containing heterocycles known for their significant biological activity. Unlike traditional approaches that often rely on harsh conditions or scarce resources, this novel method utilizes a Lewis acid-promoted [4+2] cyclization strategy to construct the thiopyran ring in a single operational step. This advancement represents a pivotal shift in how key pharmaceutical intermediates are manufactured, offering a pathway that balances high reaction efficiency with operational simplicity. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for evaluating its potential impact on supply chain resilience and cost structures in the production of high-value agrochemical and pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of thiopyran skeletons has been fraught with significant synthetic challenges that hinder large-scale commercial adoption. Conventional methodologies typically depend heavily on the use of noble metal catalysts, such as palladium or platinum complexes, which not only drive up the raw material costs substantially but also introduce complex downstream processing requirements. The presence of trace heavy metals in the final product is a critical quality attribute that must be strictly controlled, often necessitating expensive purification steps like scavenging or repeated recrystallization to meet regulatory standards for pharmaceutical intermediates. Furthermore, many existing routes require multi-step sequences involving sensitive reagents or extreme reaction conditions, which can lead to lower overall yields and increased generation of hazardous waste. These factors collectively create bottlenecks in the supply chain, extending lead times and reducing the economic viability of producing thiopyran-based active pharmaceutical ingredients or functional materials on a multi-ton scale.
The Novel Approach
In stark contrast to these legacy methods, the innovative process described in the patent data leverages readily available starting materials to achieve a direct and atom-economical transformation. By employing alpha-thiocarbonyl-N,S-ketene ketals and butynoates as substrates, the reaction proceeds through a concerted mechanism facilitated by inexpensive Lewis acids like zinc chloride. This approach eliminates the dependency on precious metals entirely, thereby simplifying the impurity profile and reducing the environmental footprint associated with catalyst disposal. The reaction conditions are notably mild, typically operating within a temperature range of 100-120°C, which enhances process safety and allows for the use of standard stainless steel reactor equipment without the need for specialized lining or high-pressure containment. This streamlined one-step construction of the thiopyran ring not only accelerates the synthesis timeline but also ensures a high degree of functional group tolerance, enabling the rapid generation of diverse derivative libraries for drug discovery programs.
Mechanistic Insights into Lewis Acid-Catalyzed [4+2] Cyclization
From a mechanistic perspective, the success of this synthesis lies in the precise activation of the electron-deficient alkyne by the Lewis acid catalyst. The zinc species coordinates with the carbonyl oxygen atoms of the butynoate ester, increasing the electrophilicity of the triple bond and facilitating the nucleophilic attack by the sulfur and carbon centers of the ketene ketal substrate. This [4+2] cycloaddition proceeds through a well-defined transition state that favors the formation of the six-membered thiopyran ring with high regioselectivity. The use of aprotic polar solvents such as N,N-dimethylformamide (DMF) further stabilizes the charged intermediates involved in the cycle, ensuring smooth progression to the final product. Understanding this electronic interplay is crucial for process chemists aiming to optimize reaction parameters, as slight modifications in the Lewis acid strength or solvent polarity can significantly influence the reaction kinetics and the ratio of desired product to side products.
Furthermore, the structural diversity inherent in the starting materials allows for extensive modulation of the final thiopyran scaffold, which is vital for structure-activity relationship (SAR) studies in medicinal chemistry. The substituents on the aromatic rings and the ester groups can be varied widely without compromising the efficiency of the cyclization, providing a versatile platform for generating analogs with improved pharmacokinetic properties. The mild nature of the reaction conditions also preserves sensitive functional groups that might otherwise be degraded under harsher acidic or basic environments typical of older synthetic routes. This high level of chemoselectivity ensures that the resulting intermediates possess the requisite purity profiles needed for subsequent coupling reactions in the synthesis of complex drug molecules, thereby reducing the risk of batch failures during later stages of manufacturing.
How to Synthesize Polysubstituted Thiopyran Derivatives Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, strict adherence to the optimized molar ratios and atmospheric conditions is paramount for achieving reproducible results. The protocol dictates a specific stoichiometric balance between the ketene ketal and the butynoate, typically favoring a slight excess of the alkyne component to drive the equilibrium towards completion. Maintaining an inert atmosphere, such as nitrogen or argon, is critical during the initial mixing and heating phases to prevent oxidative degradation of the sulfur-containing intermediates, which could lead to the formation of sulfoxides or sulfones that complicate purification. While the general procedure is robust, scaling this reaction requires careful thermal management to ensure uniform heat distribution throughout the reaction mass, preventing localized hot spots that could trigger exothermic runaway scenarios. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining alpha-thiocarbonyl-N,S-ketene ketal and butynoate ester in an aprotic polar solvent such as DMF.
- Add a catalytic amount of Zinc Chloride (ZnCl2) as the Lewis acid promoter under an inert nitrogen atmosphere to initiate the cyclization.
- Heat the reaction mixture to 100-120°C for 5-12 hours, then purify the crude product via silica gel column chromatography to obtain the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Lewis acid-catalyzed methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio, as the key precursors are commodity chemicals that are widely available from multiple global suppliers, mitigating the risk of single-source dependency. This abundance of feedstock ensures a stable supply chain capable of supporting continuous manufacturing campaigns without the interruptions often caused by the scarcity of specialized catalysts or reagents. Moreover, the elimination of noble metals translates directly into substantial cost savings, not only in terms of catalyst purchase price but also in the reduced operational expenditure associated with waste treatment and metal recovery processes. These economic efficiencies allow for more competitive pricing models when sourcing high-purity pharmaceutical intermediates, enhancing the overall margin structure for downstream drug manufacturers.
- Cost Reduction in Manufacturing: The substitution of expensive palladium or platinum catalysts with abundant zinc salts fundamentally alters the cost equation for thiopyran production. By removing the need for rigorous heavy metal clearance steps, manufacturers can reduce solvent consumption and shorten cycle times, leading to significant operational cost reductions. Additionally, the high atom economy of the [4+2] cyclization minimizes waste generation, lowering the costs associated with environmental compliance and hazardous waste disposal. This lean manufacturing approach ensures that the cost of goods sold (COGS) remains optimized even as production volumes scale up to meet commercial demand.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials ensures that production schedules are not vulnerable to the volatility of the precious metals market. Since the reaction does not require moisture-sensitive or air-sensitive reagents that demand specialized logistics, the inbound supply chain becomes more resilient and easier to manage. This reliability is crucial for maintaining consistent inventory levels and meeting just-in-time delivery commitments to pharmaceutical clients who operate on tight development timelines. The robustness of the process also means that technology transfer between different manufacturing sites can be executed with minimal friction, ensuring global supply continuity.
- Scalability and Environmental Compliance: The mild reaction temperatures and ambient pressure conditions make this process inherently safer and easier to scale from kilogram to multi-ton quantities. The use of less toxic zinc-based promoters aligns with green chemistry principles, facilitating easier regulatory approval and reducing the environmental burden on manufacturing facilities. This scalability ensures that the supply of these critical intermediates can grow in tandem with the clinical and commercial success of the final drug products, preventing supply bottlenecks during peak demand periods. Furthermore, the simplified work-up procedure reduces the volume of organic waste, supporting corporate sustainability goals and reducing the total cost of ownership for the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect regarding performance and quality. Understanding these details helps stakeholders make informed decisions about integrating this methodology into their existing production workflows or sourcing strategies. The answers reflect the consensus on the operational parameters and quality outcomes achievable through this advanced Lewis acid-catalyzed route.
Q: What are the advantages of using Lewis acid catalysis over noble metals for thiopyran synthesis?
A: Using Lewis acids like ZnCl2 significantly reduces raw material costs compared to palladium or platinum catalysts, eliminates the need for expensive heavy metal removal steps, and simplifies the purification process for pharmaceutical intermediates.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the reaction operates under mild temperatures (100-120°C) and uses commercially available starting materials, making it highly suitable for scale-up from laboratory to multi-ton commercial production without significant safety hazards.
Q: What is the typical purity and yield range for these derivatives?
A: The patented method reports yields ranging from 35% to 90%, with optimal conditions achieving up to 90% yield. The products exhibit good stereoselectivity and can be purified to high-purity specifications suitable for drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Thiopyran Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a manufacturing partner who can translate complex academic innovations into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of polysubstituted thiopyran derivatives meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality assurance means that we can consistently deliver high-purity intermediates that facilitate smoother downstream synthesis and faster time-to-market for your drug development programs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume needs and supply chain configuration. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate the technical merits of this approach against your internal criteria. Together, we can build a resilient and cost-effective supply chain for next-generation thiopyran-based therapeutics.
