Scalable Production of Optically Pure Shikonin and Alkannin via Novel Intermediate Resolution
Scalable Production of Optically Pure Shikonin and Alkannin via Novel Intermediate Resolution
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for high-value natural product derivatives, particularly those with potent biological activities like Shikonin and Alkannin. A significant breakthrough in this domain is detailed in Chinese Patent CN102399139B, which discloses a novel preparation method for optically pure Shikonin and Alkannin via a strategic intermediate resolution approach. Unlike traditional extraction methods that struggle with limited natural resources and variable purity, or previous synthetic routes plagued by expensive reagents and low yields, this patented technology leverages the formation of diastereomeric amides to achieve exceptional stereochemical control. By reacting a carboxyl-containing intermediate with a chiral amine, specifically (S)-(-)-Alpha-Methyl benzylamine, the process generates diastereomers that can be efficiently separated using standard column chromatography or recrystallization techniques. This innovation not only ensures an optical purity of ≥99% ee but also utilizes raw materials that are inexpensive and readily available, addressing the critical pain points of cost and supply continuity for global procurement teams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of chiral Shikonin and Alkannin has been hindered by significant technical and economic barriers that render many academic routes unsuitable for industrial application. Early reports, such as those by Braun and Couladouros, relied heavily on asymmetric synthesis using reagents like DIP-Cl (diisopinocampheylchloroborane), which are not only prohibitively expensive but also result in disappointingly low overall yields, often ranging between 1.3% and 7.9%. Furthermore, the optical purity achieved in these legacy processes was frequently insufficient for pharmaceutical grade standards, hovering around 40% ee to 82% ee, necessitating additional, yield-eroding purification steps. Other approaches, such as the route described by Nicolaou, while achieving higher optical purity, depended on Weinreb amides and asymmetric reduction steps that are difficult to scale due to the sensitivity of the reagents and the complexity of the electrooxidation deprotection steps required. These conventional methods create a bottleneck for supply chain managers, as the reliance on scarce chiral catalysts and multi-step sequences with poor atom economy drastically inflates the cost of goods sold (COGS) and introduces significant supply risk.
The Novel Approach
In stark contrast, the methodology outlined in patent CN102399139B introduces a paradigm shift by moving away from direct asymmetric catalysis towards a classical yet highly effective resolution strategy at the intermediate stage. This novel approach capitalizes on the physical property differences between diastereomers formed by condensing a racemic carboxylic acid intermediate with a cheap, industrially available chiral amine. This allows for the separation of enantiomers early in the synthesis tree, ensuring that all subsequent steps proceed with a single, pure stereoisomer. The process begins with a Reformatsky-type reaction on 1,4,5,8-tetramethoxy naphthalene-2-formaldehyde, a readily accessible starting material, followed by hydrolysis and amidation. The resulting diastereomeric amides are separated with high efficiency, setting the stereochemistry for the final product without the need for exotic catalysts. This strategic pivot simplifies the operational complexity, enhances the overall yield at each step, and fundamentally alters the cost structure of the synthesis, making it a viable candidate for reliable pharmaceutical intermediate supplier networks aiming for commercial scale-up.
Mechanistic Insights into Diastereomeric Amide Resolution
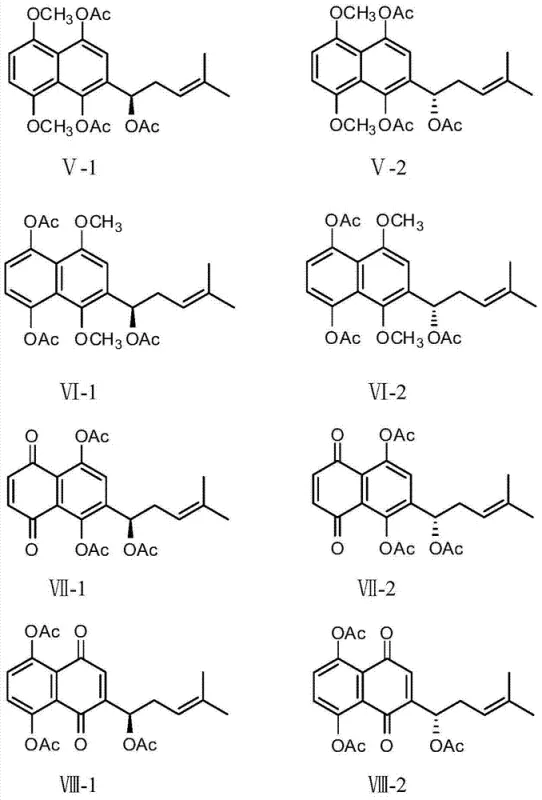
The core mechanistic advantage of this process lies in the thermodynamic and kinetic differentiation of the diastereomeric intermediates formed during the amidation step. When the racemic 3-hydroxy-3-(1,4,5,8-tetramethoxy-2-naphthyl) propionic acid reacts with a single enantiomer of Alpha-Methyl benzylamine, it generates two distinct diastereomers: the (R,S)-amide and the (S,S)-amide. Unlike enantiomers, which possess identical physical properties in an achiral environment, these diastereomers exhibit different polarities, solubilities, and adsorption affinities on silica gel. This difference is exploited through column chromatography or recrystallization, allowing for the physical isolation of the desired stereoisomer with high fidelity. The patent data indicates that this separation is robust and reproducible, providing a solid foundation for the downstream synthesis. Once the chiral center is locked in via this resolution, the subsequent transformations—including silyl protection of the hydroxyl group, reduction of the ester to an aldehyde, and Wittig olefination to install the isohexenyl side chain—proceed without racemization, preserving the optical integrity established in the early stages.
Furthermore, the impurity profile of this route is significantly cleaner compared to asymmetric catalytic methods. In catalytic asymmetric synthesis, trace metals or unreacted chiral ligands can persist as difficult-to-remove impurities, posing regulatory hurdles for API manufacturing. In this resolution-based approach, the chiral auxiliary (the amine) is covalently bound and subsequently removed during the hydrolysis steps, or separated along with the unwanted diastereomer. The use of ceric ammonium nitrate (CAN) for the oxidation of the naphthalene core to the naphthoquinone system is also well-controlled, minimizing over-oxidation byproducts. The final hydrolysis step under alkaline conditions effectively removes the acetyl protecting groups to reveal the active phenolic hydroxyls of Shikonin and Alkannin. This mechanistic clarity ensures that the final product meets the stringent purity specifications required for clinical applications, with the patent explicitly claiming an enantiomeric excess of ≥99% ee for both Shikonin (I-1) and Alkannin (I-2).
How to Synthesize Optically Pure Shikonin Efficiently
The synthesis of these high-value naphthoquinones involves a carefully orchestrated sequence of twelve distinct chemical transformations, beginning with the construction of the chiral side chain on the naphthalene scaffold. The process initiates with the zinc-mediated coupling of ethyl bromoacetate to the tetramethoxy naphthaldehyde, followed by hydrolysis to the acid. The critical resolution step follows, where the acid is coupled with the chiral amine, and the resulting diastereomers are separated. The purified amide is then protected, reduced to an aldehyde, and subjected to a Wittig reaction to extend the carbon chain. Subsequent steps involve deprotection of the silyl ether, oxidation to the quinone, acetylation, and a final global deprotection to yield the target molecule. For R&D teams looking to implement this, the detailed standardized synthesis steps are provided in the guide below, ensuring reproducibility and adherence to the patent's high-yield protocols.
- Perform a Reformatsky-type reaction on 1,4,5,8-tetramethoxy naphthalene-2-formaldehyde followed by hydrolysis to generate the racemic carboxylic acid intermediate.
- Condense the racemic acid with (S)-(-)-Alpha-Methyl benzylamine to form diastereomeric amides, then separate them via silica gel column chromatography or recrystallization.
- Process the separated chiral amides through protection, reduction, Wittig olefination, oxidation, and final hydrolysis to yield optically pure Shikonin or Alkannin.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented resolution method offers substantial advantages that directly address the priorities of procurement managers and supply chain heads, specifically regarding cost reduction in pharmaceutical intermediate manufacturing and supply reliability. The most significant value driver is the substitution of expensive, specialized chiral catalysts with commodity chiral amines. Alpha-Methyl benzylamine is produced on a massive industrial scale for various applications, ensuring a stable supply chain and a fraction of the cost of reagents like DIP-Cl or specialized phosphine ligands. This switch eliminates the volatility associated with sourcing rare earth or precious metal-based catalysts, thereby stabilizing the long-term cost structure of the production process. Additionally, the high yields reported at each step of the synthesis—from the initial 92% yield in the Reformatsky reaction to the 95% yield in the final hydrolysis—mean that less raw material is wasted, further driving down the effective cost per kilogram of the final API intermediate.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and expensive chiral auxiliaries significantly lowers the direct material costs. By utilizing a resolution strategy with recyclable or cheap chiral amines, the process avoids the costly purification steps often required to remove trace metals from pharmaceutical products. The high atom economy and robust yields across the twelve-step sequence ensure that the overall process mass intensity (PMI) is optimized, leading to substantial cost savings in solvent usage and waste disposal. This economic efficiency makes the commercial scale-up of complex pharmaceutical intermediates financially viable even in competitive markets.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 1,4,5,8-tetramethoxy naphthalene-2-formaldehyde and common reagents like ethyl bromoacetate reduces the risk of supply disruptions. Unlike processes dependent on single-source custom synthesizers for exotic reagents, this route can be executed by multiple contract manufacturing organizations (CMOs) globally. The simplicity of the separation techniques (column chromatography/recrystallization) means that the technology transfer to manufacturing sites is straightforward, reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent availability for downstream drug formulation.
- Scalability and Environmental Compliance: The process avoids the use of electrooxidation, which can be challenging to scale safely and efficiently in large reactors. Instead, it employs standard chemical oxidants like ceric ammonium nitrate under controlled conditions. The avoidance of heavy metal catalysts also simplifies the environmental compliance landscape, reducing the burden of wastewater treatment and hazardous waste management. This green chemistry aspect aligns with modern sustainability goals, making the process more attractive for companies aiming to reduce their environmental footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution-based synthesis for Shikonin and Alkannin. These answers are derived directly from the technical specifications and experimental data provided in patent CN102399139B, offering clarity on the feasibility and performance of the method for potential partners and licensees.
Q: How does this resolution method compare to asymmetric catalysis for Shikonin production?
A: Unlike asymmetric catalysis which often requires expensive, scarce chiral ligands (like DIP-Cl) and suffers from lower yields, this method uses industrially cheap chiral amines to form separable diastereomers, offering higher overall yields and easier scalability.
Q: What is the optical purity achievable with this synthetic route?
A: The patent data demonstrates that the final Shikonin and Alkannin products achieve an enantiomeric excess (ee) of greater than or equal to 99%, meeting the stringent requirements for pharmaceutical applications.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes readily available raw materials and avoids complex electrooxidation steps found in prior art. The high yields at each step and the use of standard separation techniques like column chromatography make it highly suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Shikonin Supplier
The technological potential of this resolution-based synthesis represents a significant opportunity for the commercial production of high-purity Shikonin and Alkannin derivatives. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like this can be translated from the laboratory bench to the manufacturing plant with precision. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation capable of verifying the stringent purity specifications, including the critical ≥99% ee requirement, demanded by top-tier pharmaceutical clients. We understand the nuances of handling sensitive naphthoquinone systems and can guarantee the stability and quality of the final product throughout the supply chain.
We invite global partners to collaborate with us to leverage this efficient synthetic route for your anticancer or antiviral drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply of these critical pharmaceutical intermediates is secure, cost-effective, and compliant with the highest international standards.
