Scaling Green Diaryl Sulfide Production: A Transition-Metal-Free Catalytic Breakthrough
The landscape of organic synthesis for sulfur-containing scaffolds is undergoing a significant transformation, driven by the urgent need for greener and more sustainable manufacturing processes. Patent CN110452141B introduces a groundbreaking methodology for the construction of diaryl thioether compounds, a structural motif ubiquitous in bioactive molecules and functional materials. Unlike traditional approaches that rely heavily on toxic and expensive transition metals, this invention leverages a riboflavin-based organocatalytic system coupled with an iodine source and molecular oxygen. This shift represents a paradigm change in how fine chemical manufacturers approach C-S bond formation, offering a pathway that aligns perfectly with modern regulatory demands for reduced heavy metal residues in active pharmaceutical ingredients (APIs). The technology promises not only environmental benefits but also substantial operational efficiencies for large-scale production facilities.
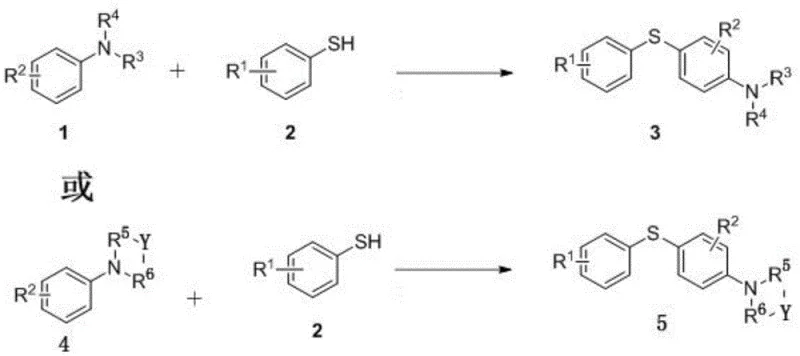
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-sulfur bonds to form diaryl sulfides has been dominated by transition-metal-catalyzed cross-coupling reactions. Prominent examples include palladium-catalyzed couplings which often necessitate harsh reaction conditions, such as temperatures exceeding 140°C and the use of specialized phosphine ligands. These methods suffer from significant drawbacks, including the generation of hazardous waste streams containing heavy metals that are difficult and costly to remove to ppm levels required by pharmacopeial standards. Furthermore, alternative non-metallic oxidant systems reported in prior art, such as those utilizing di-tert-butyl peroxide, pose serious safety risks due to the potential for explosive decomposition and often require excessive amounts of oxidants. These legacy processes frequently exhibit narrow substrate scope, failing to accommodate sensitive functional groups or N-substituted anilines, thereby limiting their utility in the diverse landscape of drug discovery and process chemistry.
The Novel Approach
The methodology disclosed in CN110452141B circumvents these critical bottlenecks by employing a biomimetic riboflavin catalyst system that operates under remarkably mild conditions. By utilizing air or oxygen as the terminal oxidant and elemental iodine as a co-catalyst, the reaction proceeds efficiently at temperatures as low as 60°C, drastically reducing energy consumption compared to conventional thermal protocols. This transition-metal-free strategy eliminates the inherent risk of metal contamination, simplifying the downstream purification process and ensuring the final product meets stringent purity specifications without the need for complex scavenging resins. The robustness of this system is evidenced by its broad compatibility with various substrates, including electron-rich and electron-deficient anilines and thiophenols, delivering high yields that rival or exceed those of traditional metal-catalyzed counterparts while maintaining a superior safety and environmental profile.
Mechanistic Insights into Riboflavin-Catalyzed C-S Bond Formation
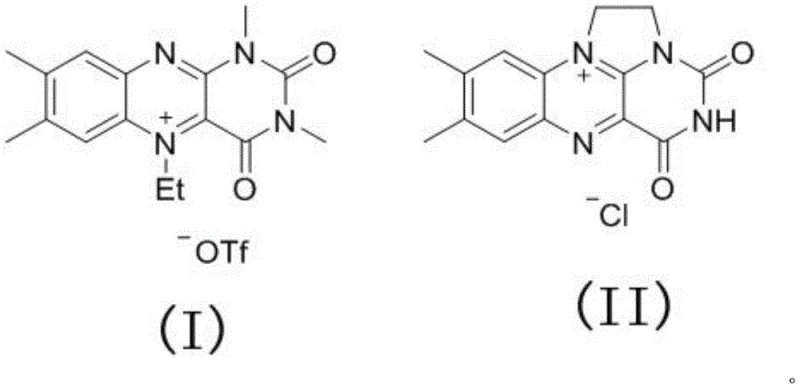
The core innovation of this synthesis lies in the unique redox properties of the riboflavin derivatives, specifically Formula (I) and Formula (II), which act as potent organocatalysts. In the presence of an iodine source, the riboflavin catalyst facilitates the generation of reactive sulfur species from the thiophenol substrate through a single-electron transfer (SET) or hydrogen atom transfer (HAT) mechanism. The isoalloxazine ring system of the riboflavin undergoes reversible reduction and oxidation cycles, effectively shuttling electrons between the substrate and the molecular oxygen oxidant. This catalytic cycle activates the sulfur-hydrogen bond of the thiophenol, generating a thiyl radical or sulfenyl iodide intermediate in situ, which subsequently attacks the electron-rich aromatic ring of the aniline derivative. The mild nature of this radical or electrophilic substitution pathway allows for the preservation of sensitive functional groups that would otherwise be degraded under the harsh acidic or basic conditions typical of classical nucleophilic aromatic substitution.
Impurity control in this process is inherently superior due to the absence of transition metal complexes that can coordinate with product molecules or form stable side-products. The primary byproducts of the reaction are water and reduced iodine species, which are easily removed during the aqueous workup phase involving sodium thiosulfate quenching. The selectivity of the riboflavin catalyst ensures that over-oxidation of the sulfur atom to sulfoxides or sulfones is minimized, a common issue in oxidative coupling reactions using strong chemical oxidants. Furthermore, the use of acetonitrile as the preferred solvent provides an optimal polarity balance that stabilizes the charged intermediates involved in the catalytic cycle while remaining easy to recover and recycle. This mechanistic elegance translates directly into a cleaner crude reaction profile, reducing the burden on purification teams and increasing the overall throughput of the manufacturing line.
How to Synthesize Diaryl Sulfides Efficiently
The practical implementation of this synthesis route is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins with the dissolution of the aniline and thiophenol starting materials in acetonitrile, followed by the sequential addition of the riboflavin catalyst and elemental iodine. The reaction mixture is then heated to a moderate temperature of 60°C and stirred under an atmosphere of air for approximately 20 hours. Upon completion, the reaction is quenched with a saturated aqueous solution of sodium thiosulfate to reduce any residual iodine, followed by extraction with ethyl acetate. The organic layer is washed, dried, and concentrated, with the final product isolated via standard column chromatography. For detailed standardized operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the technical guide below.
- Mix aniline derivatives and thiophenol compounds in acetonitrile solvent at room temperature.
- Add riboflavin catalyst (Formula I or II) and elemental iodine source sequentially to the reaction mixture.
- Heat the reaction to 60°C for 20 hours under air, then quench with sodium thiosulfate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this transition-metal-free technology offers compelling economic and logistical advantages that extend far beyond simple reagent costs. By eliminating the dependence on precious metals like palladium or platinum, manufacturers can insulate their supply chains from the volatile pricing fluctuations characteristic of the precious metals market. The removal of heavy metal catalysts also obviates the need for expensive metal scavenger resins and the associated validation testing required to prove residual metal levels are within acceptable limits, leading to significant reductions in both material costs and quality control overhead. Additionally, the use of air as the oxidant removes the cost and safety liability associated with storing and handling hazardous chemical oxidants like peroxides or hypervalent iodine reagents.
- Cost Reduction in Manufacturing: The economic impact of switching to this organocatalytic system is profound, primarily driven by the drastic simplification of the bill of materials. Without the need for expensive ligands, inert gas protection, or specialized reactor linings to prevent metal corrosion, the capital expenditure for setting up production lines is significantly lowered. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without requiring high-pressure ratings, further reducing infrastructure costs. Moreover, the high atom economy and reduced waste generation lower the costs associated with waste disposal and environmental compliance, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of commodity chemicals such as iodine and riboflavin derivatives, which are readily available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction against moisture and oxygen means that strict anhydrous conditions are not required, simplifying logistics and storage requirements for raw materials. This resilience ensures consistent production schedules and minimizes the risk of batch failures due to minor deviations in reaction conditions, thereby securing a steady flow of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: From a sustainability perspective, this process aligns perfectly with green chemistry principles, facilitating easier regulatory approval in environmentally stringent markets. The absence of toxic heavy metals simplifies the environmental impact assessment and reduces the regulatory burden associated with discharging process wastewater. The scalability is enhanced by the exothermic nature of the oxidation being manageable at 60°C, allowing for safe scale-up from kilogram to multi-ton batches without the thermal runaway risks associated with peroxide-based oxidations. This makes the technology an ideal candidate for continuous flow processing, offering further opportunities for efficiency gains in large-scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this riboflavin-catalyzed synthesis. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational parameters and expected outcomes for potential adopters of this technology.
Q: What are the advantages of using riboflavin catalysts over palladium for C-S bond formation?
A: Riboflavin catalysts eliminate the risk of heavy metal contamination, remove the need for expensive ligand systems, and operate under significantly milder temperatures (60°C vs 140°C), reducing energy costs and safety hazards associated with high-pressure reactors.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes air as the terminal oxidant and common solvents like acetonitrile, avoiding hazardous peroxides. The simple workup procedure involving standard extraction and chromatography makes it highly suitable for commercial scale-up.
Q: What is the substrate scope for this diaryl thioether synthesis?
A: The method demonstrates excellent universality, accommodating various substituents including alkyl, alkoxy, halogens, and nitro groups on both the aniline and thiophenol rings, as well as heterocyclic thiols, yielding products with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this transition-metal-free catalytic technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are fully realized in a commercial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of diaryl sulfide delivered is free from heavy metal contaminants and meets the highest international quality standards.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this innovative synthesis route for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this metal-free approach can optimize your budget. Contact us today to obtain specific COA data and comprehensive route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your critical sulfur-containing building blocks.
