Advanced Cu-OMS-2 Catalyzed Synthesis of Diaryl Sulfides for Pharmaceutical Applications
Advanced Cu-OMS-2 Catalyzed Synthesis of Diaryl Sulfides for Pharmaceutical Applications
The development of efficient, scalable, and environmentally benign synthetic routes for sulfur-containing heterocycles remains a paramount challenge in modern medicinal chemistry. Patent CN111978219B introduces a groundbreaking methodology for the synthesis of diaryl sulfide compounds utilizing a heterogeneous manganese-oxygen octahedral molecular sieve doped with copper (Cu-OMS-2). This technology represents a significant leap forward from traditional homogeneous catalysis, offering a robust platform for the production of high-purity pharmaceutical intermediates. The core innovation lies in the unique redox properties of the OMS-2 framework, which, when modified with copper ions, exhibits superior catalytic activity for Carbon-Sulfur (C-S) bond formation. For R&D directors and process chemists, this patent outlines a pathway to access complex biaryl thioethers under mild conditions, minimizing the reliance on expensive noble metals and simplifying downstream purification processes significantly.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-S bonds to form diaryl sulfides has relied heavily on transition-metal catalyzed cross-coupling reactions, often employing palladium or copper salts in homogeneous systems. These traditional methodologies, while effective on a small scale, suffer from severe drawbacks when translated to industrial manufacturing. The primary issue is the difficulty in separating the toxic metal catalysts from the final product, which necessitates rigorous and costly purification steps to meet stringent pharmaceutical purity standards. Furthermore, homogeneous catalysts are typically single-use, leading to substantial accumulation of heavy metal waste and increased environmental burden. The requirement for specialized ligands to stabilize these metal centers further escalates the raw material costs, making the overall process economically inefficient for large-scale production of commodity intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN111978219B utilizes a heterogeneous Cu-OMS-2 catalyst that fundamentally alters the process economics and operational simplicity. As illustrated in the general reaction scheme below, the coupling of substituted thiophenols with aryl halides proceeds efficiently in dimethyl sulfoxide (DMSO) using cesium carbonate as a base.
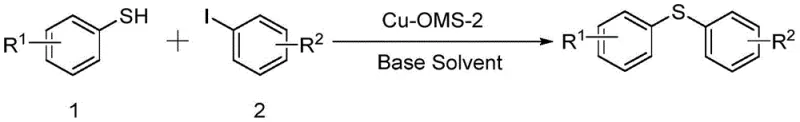
This novel approach eliminates the need for complex ligand systems and allows for the catalyst to be recovered via simple filtration after the reaction is complete. The heterogeneous nature of Cu-OMS-2 ensures that the active copper sites are accessible for the catalytic cycle while remaining physically distinct from the reaction mixture, thereby preventing metal contamination of the product. This shift from homogeneous to heterogeneous catalysis not only streamlines the workflow but also aligns perfectly with green chemistry principles by reducing waste generation and enabling catalyst recycling, a critical factor for sustainable manufacturing.
Mechanistic Insights into Cu-OMS-2 Catalyzed C-S Coupling
The efficacy of the Cu-OMS-2 catalyst stems from the synergistic interaction between the manganese oxide framework and the doped copper species. The OMS-2 structure possesses mixed-valence manganese ions (Mn2+, Mn3+, Mn4+), which provide excellent redox flexibility. When copper is doped into this lattice, primarily as Cu(II) oxide species, it creates highly active sites that facilitate the oxidative addition and reductive elimination steps essential for the C-S coupling mechanism. The copper centers activate the aryl iodide substrate, while the basic environment generated by the carbonate base deprotonates the thiophenol to form a reactive thiolate nucleophile. This precise orchestration of electronic effects within the molecular sieve pore structure enhances the reaction kinetics, allowing the transformation to proceed at moderate temperatures ranging from 60°C to 110°C.
From an impurity control perspective, the heterogeneous mechanism offers distinct advantages over solution-phase catalysis. In homogeneous systems, metal leaching often leads to trace metal impurities that are notoriously difficult to remove and can catalyze unwanted side reactions during storage. With Cu-OMS-2, the rigid framework stabilizes the copper ions, minimizing leaching into the solvent. This stability ensures that the crude reaction mixture contains significantly lower levels of heavy metal residues, simplifying the purification strategy. For a reliable pharmaceutical intermediates supplier, this means a more consistent impurity profile and a higher probability of passing rigorous quality control specifications without extensive recrystallization or chromatography, directly impacting the cost of goods sold.
How to Synthesize Diaryl Sulfides Efficiently
The practical implementation of this technology involves a straightforward protocol that is amenable to standard reactor setups found in most pilot and production plants. The process begins with the charging of substituted thiophenol, the aryl iodide coupling partner, and the base into a reactor containing the solvent, typically DMSO. The Cu-OMS-2 catalyst is then added, and the mixture is heated to the optimal temperature of 95°C. Reaction progress is monitored via TLC or HPLC, typically reaching completion within 3 to 8 hours depending on the electronic nature of the substituents. Upon completion, the solid catalyst is removed by filtration, and the filtrate is worked up using standard extraction techniques with dichloromethane. The detailed standardized synthesis steps, including specific molar ratios and workup parameters, are provided in the guide below.
- Charge the reactor with substituted thiophenol, solvent (DMSO), base (Cs2CO3), Cu-OMS-2 catalyst, and aryl iodide at room temperature.
- Heat the mixture to 60-110°C (optimally 95°C) and stir for 3-8 hours under reflux conditions.
- Cool the reaction, filter to recover the heterogeneous catalyst, extract with dichloromethane, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the Cu-OMS-2 catalytic system presents a compelling value proposition centered on cost reduction and operational resilience. The transition to a heterogeneous catalyst system fundamentally changes the cost structure of diaryl sulfide manufacturing by eliminating the recurring expense of expensive ligands and reducing the consumption of precious metals. Since the catalyst can be recovered and reused multiple times with minimal loss in activity, the effective cost per kilogram of the catalyst drops drastically over time. This durability translates into significant long-term savings, making the process highly competitive against traditional methods that rely on stoichiometric or non-recoverable catalytic loads.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of the downstream processing train. By removing the need for complex metal scavenging resins or extensive washing protocols required for homogeneous palladium systems, manufacturers can reduce solvent consumption and labor hours. The ability to operate at atmospheric pressure and moderate temperatures further lowers utility costs associated with heating and cooling. Additionally, the high atom economy of the direct C-S coupling minimizes the generation of byproduct waste, reducing disposal fees and enhancing the overall sustainability profile of the manufacturing site.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the volatility of precious metal markets and the geopolitical risks associated with sourcing specialized ligands. The Cu-OMS-2 catalyst relies on abundant and inexpensive base metals—manganese and copper—which are readily available globally, insulating the supply chain from price shocks. Furthermore, the robustness of the catalyst allows for flexible production scheduling; the reaction tolerates a wide range of functional groups, meaning the same catalytic system can be used to produce a diverse library of intermediates without changing the core process infrastructure. This versatility ensures that production lines can be quickly adapted to meet fluctuating market demands for different API precursors.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen challenges, particularly regarding heat transfer and mixing in heterogeneous slurries. However, the Cu-OMS-2 system has demonstrated excellent stability and performance in batch reactors, indicating strong potential for seamless scale-up. The heterogeneous nature of the catalyst also simplifies compliance with increasingly strict environmental regulations regarding heavy metal discharge. By retaining the copper within the solid matrix, the aqueous waste streams generated during workup contain negligible metal content, facilitating easier wastewater treatment and reducing the regulatory burden on the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational parameters and expected outcomes for potential licensees or manufacturing partners.
Q: What are the advantages of Cu-OMS-2 over traditional homogeneous catalysts?
A: Cu-OMS-2 is a heterogeneous catalyst that can be easily filtered and reused multiple times without significant loss of activity, unlike homogeneous palladium or copper salts which require complex removal steps and generate heavy metal waste.
Q: What is the optimal reaction temperature for this C-S coupling?
A: According to patent optimization data, 95°C in DMSO provides the highest yield (up to 89%), balancing reaction rate and energy consumption effectively.
Q: Can this method tolerate diverse functional groups?
A: Yes, the method demonstrates excellent functional group tolerance, successfully synthesizing derivatives with electron-withdrawing groups like cyano, nitro, and halogens (Cl, F) on the aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Cu-OMS-2 catalytic technology in streamlining the production of high-value sulfur-containing intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of diaryl sulfide meets the exacting standards required for pharmaceutical applications. We are committed to leveraging advanced catalytic methodologies to deliver superior quality products while maintaining cost competitiveness.
We invite you to collaborate with us to evaluate the feasibility of this route for your specific project needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this heterogeneous catalytic system. Please contact our technical procurement team today to request specific COA data for our existing diaryl sulfide portfolio or to discuss route feasibility assessments for your custom synthesis requirements. Together, we can optimize your supply chain and accelerate the delivery of life-saving medicines to patients worldwide.
