Industrial Scale-Up of Erlotinib Hydrochloride: A Novel Synthetic Route for High-Purity API Intermediates
The pharmaceutical landscape for oncology treatments continues to demand robust, scalable, and cost-efficient synthetic routes for critical tyrosine kinase inhibitors. Patent CN101463013A, published in June 2009, introduces a significant methodological advancement in the preparation of Erlotinib Hydrochloride, a potent EGFR inhibitor widely known under the trade name Tarceva. Unlike conventional pathways that rely on complex protection-deprotection sequences or harsh demethylation protocols, this invention leverages 3,4-dihydroxybenzaldehyde as a strategic starting material. By fundamentally restructuring the synthetic sequence, the patent addresses critical bottlenecks associated with yield loss and purification difficulties during scale-up. The disclosed methodology not only streamlines the construction of the quinazoline core but also optimizes the handling of intermediates through smart functional group manipulation, specifically the oxidation of nitriles to amides prior to reduction. For global supply chain stakeholders, this represents a pivotal shift towards more sustainable and economically viable manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Erlotinib has been dominated by routes derived from early patents, such as those assigned to Pfizer, which typically initiate from ethyl 3,4-dihydroxybenzoate. As illustrated in the reaction scheme below, this classical approach involves etherification followed by nitration, reduction, and cyclization. However, technical literature and subsequent patent analyses have highlighted severe scalability issues with this pathway. Specifically, the nitration step is highly sensitive to substrate quantity; as the reaction scale increases, both yield and purity suffer significant degradation due to exothermic control challenges. Furthermore, the reduction of the nitro group in these traditional routes often necessitates the use of expensive transition metal catalysts, and the isolation of the free base frequently requires column chromatography, a technique that is notoriously difficult and costly to implement in multi-ton commercial production.
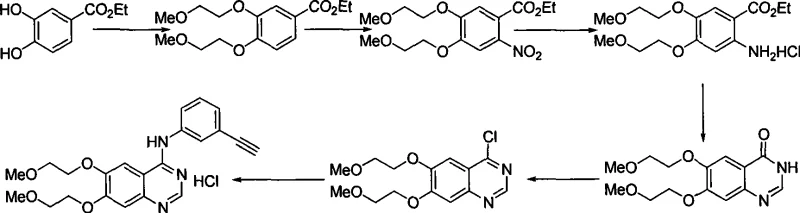
Alternative strategies, such as the one proposed by Jyothi Prasad (WO200706091), attempted to mitigate some issues by employing a demethylation-protection-reprotection sequence starting from 6,7-dimethoxy-3,4-dihydroquinazoline-4-one. While chemically feasible, this approach introduces substantial inefficiency by adding unnecessary synthetic steps. The requirement to first demethylate the methoxy groups, protect the resulting hydroxyls, perform the chlorination and coupling, and finally deprotect the molecule inherently lowers the overall yield and increases the consumption of reagents and solvents. Each additional step in a linear synthesis compounds the risk of yield loss and impurity generation, making such routes less attractive for cost-sensitive generic API manufacturing.
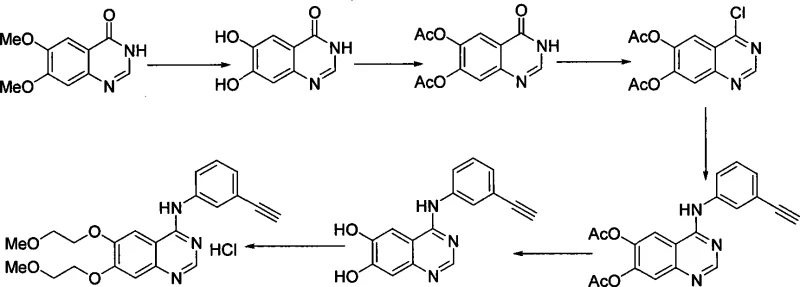
The Novel Approach
In stark contrast to these legacy methods, the process detailed in CN101463013A adopts a convergent and linear strategy that bypasses the need for demethylation entirely. By selecting 3,4-dihydroxybenzaldehyde as the foundational building block, the synthesis immediately establishes the required oxygenation pattern without the need for later-stage modification of the aromatic ring. The route proceeds through a logical sequence of oxime formation, etherification, nitration, and a unique oxidation-cyclization cascade. This structural logic ensures that the expensive and hazardous steps associated with stripping methyl groups are eliminated from the process map. Consequently, the novel approach offers a direct path to the 6,7-disubstituted quinazoline core, significantly reducing the total number of unit operations and minimizing the environmental footprint associated with solvent usage and waste generation.
Mechanistic Insights into the Optimized Quinazoline Synthesis
The core innovation of this patent lies in the precise ordering of functional group transformations, particularly the handling of the nitrile and nitro moieties. The synthesis begins with the conversion of 3,4-dihydroxybenzaldehyde to its oxime derivative (Compound 1) using hydroxylamine hydrochloride, a reaction driven to completion by dehydrating agents like anhydrous magnesium sulfate or acetic anhydride. Subsequent etherification with chloroethyl methyl ether under basic conditions (using potassium carbonate or sodium carbonate) installs the necessary alkoxy side chains to yield Compound 2. The critical nitration step is then performed under strictly controlled low-temperature conditions (0-10°C) using mixed acid systems, ensuring regioselectivity and preventing thermal runaway. Following nitration, the nitrile group is oxidized to a primary amide (Compound 4) using hydrogen peroxide or sodium perborate. This specific transformation is mechanistically vital because the resulting amide is a solid with poor water solubility, facilitating easy isolation via centrifugation rather than complex extraction protocols.
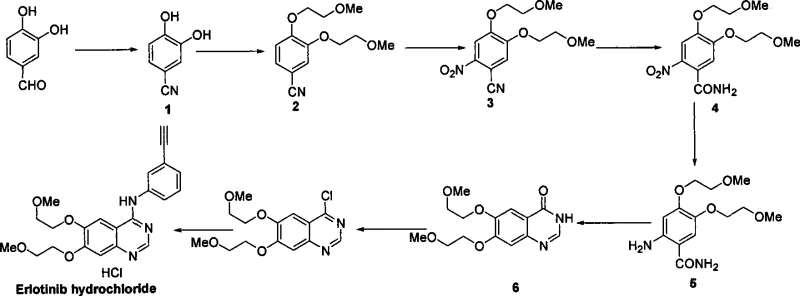
Following the oxidation, the nitro group is reduced to an amine (Compound 5) using catalytic hydrogenation or transfer hydrogenation with ammonium formate and Pd/C. The proximity of the newly formed amine to the amide carbonyl facilitates the subsequent cyclization step upon heating with formamide or formic acid, closing the pyrimidine ring to form the quinazolinone scaffold (Compound 6). The final stages involve chlorination of the 4-position using thionyl chloride, followed by nucleophilic aromatic substitution with 3-ethynylaniline. This sequence demonstrates a high degree of chemoselectivity, where the alkyne functionality remains intact during the acidic chlorination and cyclization steps. The meticulous control over reaction parameters, such as maintaining nitration temperatures below 10°C and utilizing mild oxidants, ensures that the impurity profile remains manageable, directly addressing the purity concerns that plague older synthetic methodologies.
How to Synthesize Erlotinib Hydrochloride Efficiently
The synthesis of Erlotinib Hydrochloride via this patented route requires careful attention to reaction stoichiometry and temperature control, particularly during the nitration and oxidation phases. The process is designed to be operationally simple, utilizing common industrial solvents like toluene, DMSO, and methanol. The following guide outlines the critical operational parameters derived from the patent embodiments, focusing on the transition from bench-scale optimization to pilot-plant execution. For detailed standard operating procedures and safety data sheets regarding these specific transformations, please refer to the technical guidelines below.
- Convert 3,4-dihydroxybenzaldehyde to the corresponding oxime (Compound 1) using hydroxylamine hydrochloride and a dehydrating agent.
- Perform etherification with chloroethyl methyl ether under basic conditions to protect hydroxyl groups, yielding Compound 2.
- Execute controlled nitration at 0-10°C to introduce the nitro group, followed by oxidation of the nitrile to an amide (Compound 4).
- Reduce the nitro group to an amine (Compound 5) and cyclize using formamide/formic acid to form the quinazolinone core (Compound 6).
- Chlorinate the quinazolinone and couple with 3-ethynylaniline to finalize the Erlotinib structure, followed by salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage stems from the drastic simplification of the purification workflow. By engineering the intermediates to be solids that can be separated via centrifugation, the process eliminates the need for large-volume liquid-liquid extractions and, crucially, column chromatography. In an industrial setting, removing column chromatography translates to massive reductions in silica gel costs, solvent consumption, and processing time, leading to substantial cost savings in the final API price. Furthermore, the avoidance of expensive metal catalysts for nitro reduction in favor of more economical alternatives like iron powder or ammonium formate further drives down the raw material costs, enhancing the overall margin potential for generic manufacturers.
- Cost Reduction in Manufacturing: The elimination of demethylation and reprotection steps inherently reduces the consumption of reagents and solvents, directly lowering the variable cost per kilogram of the produced intermediate. Additionally, the ability to isolate key intermediates through simple filtration or centrifugation rather than complex distillation or chromatographic separation significantly reduces utility costs and waste disposal fees. This streamlined approach ensures that the manufacturing process remains economically competitive even when raw material prices fluctuate, providing a stable cost structure for long-term supply contracts.
- Enhanced Supply Chain Reliability: Starting from 3,4-dihydroxybenzaldehyde, a commodity chemical with a robust global supply chain, mitigates the risk of raw material shortages that can occur with specialized esters or protected heterocycles. The mild reaction conditions, particularly the low-temperature nitration and ambient pressure reductions, reduce the dependency on specialized high-pressure or cryogenic equipment, allowing for production across a wider range of qualified manufacturing sites. This flexibility enhances supply continuity and reduces the lead time for high-purity pharmaceutical intermediates, ensuring that downstream API production schedules are met without delay.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial suitability, with reaction exotherms managed through controlled addition rates and temperatures that are easily maintained in standard stainless steel reactors. The reduction in solvent usage and the avoidance of heavy metal catalysts simplify the wastewater treatment process, aiding in compliance with increasingly stringent environmental regulations. This 'green chemistry' aspect not only reduces the environmental footprint but also minimizes the regulatory burden associated with solvent residue limits in the final drug substance, facilitating faster regulatory approval and market entry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, purity profiles, and scalability factors. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: How does this novel route improve upon the traditional Pfizer synthesis method?
A: The traditional route starting from ethyl 3,4-dihydroxybenzoate often suffers from yield declines during nitration when scaling up and requires expensive metal catalysts for nitro reduction. This novel method utilizes 3,4-dihydroxybenzaldehyde, avoiding the need for subsequent demethylation and reprotection steps found in other routes, thereby shortening the synthetic sequence and improving overall throughput.
Q: What are the purification advantages of the intermediate oxidation step?
A: A key innovation in this process is the oxidation of the nitrile group to an amide prior to nitro reduction. The resulting amide intermediate is a solid with poor water solubility, which allows for easy separation via centrifugation. This eliminates the need for large-scale solvent extractions and column chromatography, significantly reducing solvent waste and processing time.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the reaction conditions are notably mild, with nitration temperatures controlled between 0-10°C and cyclization occurring under reflux in formamide/formic acid. The use of readily available starting materials and the avoidance of harsh demethylation reagents make this route highly adaptable for multi-kilogram to ton-scale production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Erlotinib Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel Erlotinib synthesis are fully realized in practice. We operate state-of-the-art facilities equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the exacting standards required for oncology drug manufacturing.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthetic technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive efficiency and reliability in your Erlotinib production.
