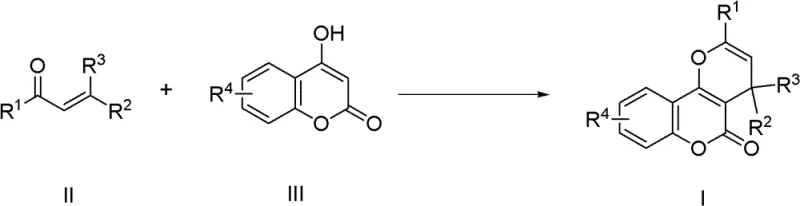
Advanced Gold-Catalyzed Synthesis of Pyranocoumarin Derivatives for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex heterocyclic scaffolds, particularly those exhibiting potent biological activities. Patent CN102336763B introduces a groundbreaking synthesis method for pyranocoumarin derivatives, a class of compounds renowned for their antimalarial, antibacterial, and anti-HIV properties. This technology leverages a sophisticated gold-catalyzed tandem reaction between alpha,beta-unsaturated ketones and 4-hydroxycoumarin compounds. Unlike conventional approaches that often suffer from harsh conditions and poor atom economy, this novel route utilizes a AuCl3/3AgOTf catalytic system to achieve high yields under relatively mild thermal conditions. For R&D directors and procurement specialists, this patent represents a significant leap forward in the reliable production of high-purity pharmaceutical intermediates, offering a pathway that balances synthetic efficiency with environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functional pyranocoumarins has relied heavily on acid-catalyzed condensation reactions, such as those utilizing sulfuric acid or iodine in acetic acid media. While these methods established the foundational chemistry, they are plagued by significant drawbacks that hinder modern commercial scale-up. The use of strong mineral acids generates substantial corrosive waste streams, creating severe environmental disposal challenges and increasing the operational overhead for waste treatment facilities. Furthermore, these traditional protocols frequently exhibit low reaction yields and poor regioselectivity, leading to complex impurity profiles that require extensive and costly downstream purification. The reliance on stoichiometric oxidants in some alternative methods further exacerbates the atom economy issues, making these processes economically unviable for large-scale manufacturing of high-value drug intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN102336763B employs a transition metal-catalyzed strategy that fundamentally reshapes the reaction landscape. By utilizing a dual catalytic system of gold(III) chloride and silver triflate, the process activates the unsaturated ketone substrate through a distinct mechanistic pathway that avoids the pitfalls of brute-force acid catalysis. This approach not only dramatically improves the reaction yield, with specific examples demonstrating efficiencies up to 98%, but also simplifies the operational workflow. The reaction proceeds smoothly in common organic solvents like toluene, eliminating the need for specialized or hazardous reaction media. This shift towards a catalytic, high-selectivity process ensures that the resulting pyranocoumarin derivatives are obtained with superior purity, directly addressing the stringent quality requirements of the global pharmaceutical supply chain.
Mechanistic Insights into AuCl3-Catalyzed Cyclization
The core innovation of this synthesis lies in the unique activation mode provided by the gold catalyst. The AuCl3/3AgOTf system functions as a potent Lewis acid that coordinates with the carbonyl or alkene moiety of the alpha,beta-unsaturated ketone, thereby increasing its electrophilicity. This activation facilitates a nucleophilic attack by the 4-hydroxycoumarin, initiating a cascade sequence that involves carbon-hydrogen bond activation and subsequent deprotonation. The presence of the silver salt serves to regenerate the active gold species and stabilize the intermediate complexes, ensuring the catalytic cycle continues efficiently. This concerted mechanism allows for the formation of the pyran ring fused to the coumarin core with exceptional regiocontrol, minimizing the formation of structural isomers that typically complicate purification in non-catalytic routes.
From an impurity control perspective, this mechanistic precision is paramount. Traditional acid-catalyzed methods often lead to polymerization of the unsaturated ketone or over-oxidation of the coumarin ring, generating difficult-to-remove byproducts. The gold-catalyzed pathway, however, directs the reaction specifically towards the desired intramolecular cyclization. The mild reaction conditions, typically maintained between 80°C and 100°C, prevent thermal degradation of sensitive functional groups on the aromatic rings. This results in a cleaner crude reaction mixture, which significantly reduces the burden on the purification team. For manufacturing partners, this means a more predictable process with consistent batch-to-batch quality, a critical factor when supplying intermediates for regulated drug substances.
How to Synthesize Pyranocoumarin Derivatives Efficiently
Implementing this synthesis protocol requires careful attention to the preparation of the catalytic system and the control of reaction parameters. The process begins with the in situ generation of the active catalyst by mixing gold(III) chloride and silver triflate in a dry organic solvent, typically toluene, under inert atmosphere conditions to prevent catalyst deactivation. Once the catalyst is activated, the substrates—specifically the alpha,beta-unsaturated ketone and the 4-hydroxycoumarin derivative—are introduced in a precise molar ratio, generally ranging from 1:1.0 to 1:1.2. The reaction mixture is then heated to the optimal temperature window and stirred for a duration sufficient to drive the conversion to completion, usually between 6 to 8 hours. Detailed standardized synthesis steps follow below.
- Prepare the catalytic system by mixing AuCl3 and AgOTf in toluene solvent under magnetic stirring at room temperature.
- Dissolve the alpha,beta-unsaturated ketone and 4-hydroxycoumarin substrates in toluene and add to the catalyst mixture.
- Heat the reaction mixture to 80-100°C for 6-8 hours, then purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this gold-catalyzed methodology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the post-reaction workup. Unlike acid-catalyzed processes that require neutralization, extensive washing, and treatment of acidic wastewater, this protocol allows for direct purification via silica gel chromatography after simple solvent removal. This reduction in unit operations translates directly into lower processing times and reduced consumption of auxiliary chemicals. Furthermore, the high atom economy of the reaction ensures that raw material costs are optimized, as a greater proportion of the starting mass is converted into the valuable final product rather than lost as waste.
- Cost Reduction in Manufacturing: Although gold catalysts are inherently expensive, their usage in this protocol is catalytic rather than stoichiometric, and the high turnover frequency justifies the initial investment. The elimination of corrosive acids removes the need for specialized acid-resistant reactor linings and expensive waste neutralization protocols, leading to substantial long-term operational savings. Additionally, the high selectivity reduces the loss of valuable starting materials to side reactions, effectively lowering the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of this reaction conditions makes it highly suitable for scale-up from laboratory to commercial production. The use of stable, commercially available starting materials like chalcones and 4-hydroxycoumarins ensures a secure supply base, mitigating the risk of raw material shortages. The simplified purification process also shortens the overall manufacturing cycle time, allowing for faster turnaround on customer orders and improved responsiveness to market demand fluctuations without compromising product quality.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions intensify, this green chemistry approach provides a significant compliance advantage. By avoiding the generation of heavy acidic waste and utilizing a recyclable catalytic system, manufacturers can meet stringent environmental standards more easily. The process is inherently scalable, as the heat management and mixing requirements are less demanding than exothermic acid-catalyzed reactions, facilitating a smoother transition from pilot plant to multi-ton production scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: What are the primary advantages of the AuCl3/AgOTf catalytic system over traditional acid catalysts?
A: The AuCl3/3AgOTf system offers significantly higher regioselectivity and yield compared to traditional H2SO4 or I2 catalyzed methods, while avoiding the environmental pollution associated with strong mineral acids.
Q: What is the typical reaction temperature and time for this synthesis?
A: The reaction is optimally conducted at temperatures between 80°C and 100°C, with a reaction time ranging from 6 to 8 hours to ensure complete conversion.
Q: How is the final product purified in this protocol?
A: Purification is achieved through a straightforward workup involving the addition of silica gel, solvent removal under reduced pressure, and final isolation via TLC or column chromatography using cyclohexane/ethyl acetate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyranocoumarin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the pharmaceutical intermediate market. Our technical team has thoroughly analyzed the potential of the gold-catalyzed route described in CN102336763B and is fully prepared to leverage this methodology for custom manufacturing projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial supply is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of pyranocoumarin derivatives meets the exacting standards required for downstream drug synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic impact of switching to this catalytic process for your supply chain. We encourage you to request specific COA data and route feasibility assessments to verify the compatibility of this technology with your current development pipelines. Let us help you secure a reliable, high-quality supply of these vital intermediates while optimizing your manufacturing costs and environmental footprint.
