Advanced One-Pot Synthesis of Benzo[c,d]indole Imine Derivatives for Pharmaceutical Applications
Advanced One-Pot Synthesis of Benzo[c,d]indole Imine Derivatives for Pharmaceutical Applications
The development of efficient synthetic routes for heterocyclic compounds remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activity. Patent CN114349684B discloses a groundbreaking methodology for the synthesis of polysubstituted benzo[c,d]indole imine derivatives, a class of molecules renowned for their efficacy in inhibiting tumor cell proliferation and treating cardiovascular diseases. Unlike traditional multi-step sequences that rely on hazardous reagents, this invention leverages a palladium-catalyzed one-pot cyclization strategy. By reacting 8-halo-1-naphthylamine compounds with isonitrile derivatives under mild nitrogen atmospheres, the process achieves high yields with exceptional operational simplicity. This technical breakthrough not only streamlines the creation of diverse compound libraries for drug discovery but also addresses critical supply chain concerns regarding safety and scalability, positioning it as a vital asset for reliable pharmaceutical intermediate supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzo[c,d]indole imine core has been fraught with significant chemical and logistical challenges that hinder efficient manufacturing. Conventional pathways typically initiate from benzo[c,d]indol-2-one, requiring a cumbersome sequence of nitrogen atom modification followed by thionation using phosphorus pentasulfide to generate benzo[c,d]indole-2-thione derivatives. The final conversion to the desired imine often necessitates the use of mercuric oxide or mercury acetate as catalysts, introducing severe toxicity issues and complicating waste disposal protocols. Alternatively, methods employing phosphorus oxychloride as a dehydrating agent for condensation with primary amines suffer from similar drawbacks, including harsh reaction conditions and limited structural modifiability. The inherent rigidity of the benzo[c,d]indol-2-one starting material restricts the diversity of accessible derivatives, resulting in a scarcity of reported compounds and limiting the ability of R&D teams to explore comprehensive structure-activity relationships effectively.
The Novel Approach
In stark contrast to these archaic techniques, the novel approach detailed in the patent utilizes a direct, catalytic cyclization that fundamentally reshapes the synthetic landscape for these valuable heterocycles. By employing 8-halo-1-naphthylamine compounds as the primary building blocks, the method bypasses the need for pre-formed indole ketones entirely. The reaction proceeds via a streamlined one-pot protocol where the amine substrate reacts directly with an isonitrile derivative in the presence of a high-efficiency transition metal catalyst and a base additive. 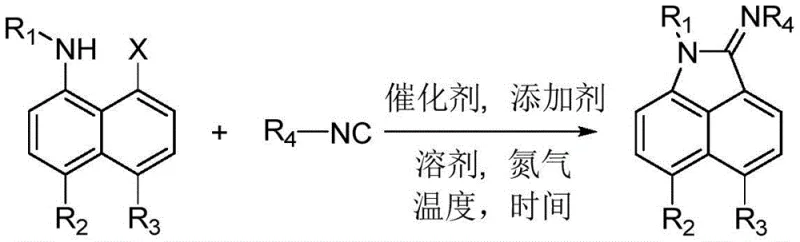 This transformation occurs smoothly in aprotic solvents under a nitrogen atmosphere, typically at temperatures ranging from 25°C to 120°C. The result is a robust, versatile platform that enables the rapid assembly of complex polysubstituted architectures with minimal purification steps, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing while adhering to stricter environmental standards.
This transformation occurs smoothly in aprotic solvents under a nitrogen atmosphere, typically at temperatures ranging from 25°C to 120°C. The result is a robust, versatile platform that enables the rapid assembly of complex polysubstituted architectures with minimal purification steps, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing while adhering to stricter environmental standards.
Mechanistic Insights into Pd-Catalyzed Cyclization
The core of this innovation lies in the sophisticated interplay between the palladium catalyst and the specific electronic properties of the 8-halo-1-naphthylamine substrate. The mechanism likely initiates with the oxidative addition of the palladium species into the carbon-halogen bond at the 8-position of the naphthalene ring, generating a reactive organopalladium intermediate.  Subsequent coordination and insertion of the isonitrile functionality into the palladium-carbon bond facilitate the formation of the new C-C bond necessary for ring closure. The presence of additives such as potassium carbonate (K2CO3) plays a crucial role in neutralizing acidic byproducts and regenerating the active catalytic species, ensuring the cycle continues efficiently. This mechanistic pathway avoids the high-energy barriers associated with traditional dehydration or thionation steps, allowing the reaction to proceed under kinetically favorable conditions that preserve sensitive functional groups on the aromatic rings.
Subsequent coordination and insertion of the isonitrile functionality into the palladium-carbon bond facilitate the formation of the new C-C bond necessary for ring closure. The presence of additives such as potassium carbonate (K2CO3) plays a crucial role in neutralizing acidic byproducts and regenerating the active catalytic species, ensuring the cycle continues efficiently. This mechanistic pathway avoids the high-energy barriers associated with traditional dehydration or thionation steps, allowing the reaction to proceed under kinetically favorable conditions that preserve sensitive functional groups on the aromatic rings.
From an impurity control perspective, this catalytic system offers distinct advantages over stoichiometric heavy metal methods. The use of catalytic amounts of palladium (typically 0.05 to 0.2 equivalents) significantly reduces the metal load in the crude reaction mixture compared to stoichiometric mercury reagents. Furthermore, the mild reaction temperatures and the specificity of the C-H activation/C-N bond formation process minimize the formation of polymeric byproducts or over-reacted species often seen in harsh acidic conditions. The broad substrate tolerance, accommodating electron-withdrawing groups like nitro and electron-donating groups like methoxy on the naphthalene core, indicates a robust catalytic cycle that is less susceptible to deactivation by substrate electronics. This reliability is paramount for maintaining high-purity pharmaceutical intermediate specifications required for downstream biological testing and eventual clinical application.
How to Synthesize Benzo[c,d]indole Imine Efficiently
Executing this synthesis requires precise attention to molar ratios and atmospheric conditions to maximize yield and reproducibility. The patent outlines a generalized procedure where the 8-halo-1-naphthylamine, isonitrile derivative, catalyst, and additive are combined in a specific molar ratio of 1.0:1.5:0.1:2.0. The choice of solvent is critical, with 1,2-dichloroethane (DCE) identified as the optimal medium, although alternatives like acetonitrile, toluene, and DMF are viable depending on substrate solubility. The reaction is conducted under a strict nitrogen atmosphere to prevent oxidation of the catalyst or sensitive intermediates, with heating applied between 25°C and 120°C for durations up to 24 hours. Following the reaction, a straightforward workup involving aqueous quenching and ethyl acetate extraction isolates the crude product, which is then purified via silica gel chromatography.
- Combine 8-halo-1-naphthylamine, isonitrile derivative, Pd catalyst (e.g., Pd(TFA)2), and additive (e.g., K2CO3) in an aprotic solvent.
- Heat the mixture to 25-120°C under a nitrogen atmosphere and stir for 0-24 hours until reaction completion.
- Cool to room temperature, extract with ethyl acetate after water addition, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route represents a strategic opportunity to optimize both cost structures and operational resilience. The elimination of toxic mercury salts and phosphorus pentasulfide removes the need for specialized hazardous waste disposal contracts and expensive heavy metal scavenging resins, leading to substantial cost savings in waste management and regulatory compliance. Furthermore, the one-pot nature of the reaction consolidates multiple unit operations into a single vessel, drastically reducing solvent consumption, energy usage for heating and cooling cycles, and overall processing time. This simplification translates directly into improved throughput and reduced manufacturing lead times, enabling faster response to market demands for key pharmaceutical building blocks without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic sequence and the replacement of expensive, hazardous reagents. By utilizing catalytic amounts of palladium rather than stoichiometric quantities of mercury or phosphorus reagents, the raw material costs are significantly lowered. Additionally, the high atom economy of the cyclization reaction minimizes waste generation, reducing the burden on effluent treatment plants. The ability to use readily available 8-halo-1-naphthylamine precursors, which are commodity chemicals in many regions, further stabilizes the supply chain against price volatility associated with exotic starting materials, ensuring a more predictable and lower cost of goods sold (COGS) for the final active pharmaceutical ingredients.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness and flexibility of the reaction conditions. The method tolerates a wide range of substrates and functional groups, meaning that if a specific isonitrile or naphthylamine variant faces temporary shortages, alternative analogs can often be substituted without re-optimizing the entire process. The use of common aprotic solvents like DCE or acetonitrile ensures that solvent supply chains remain stable and unaffected by niche chemical shortages. Moreover, the mild reaction parameters reduce the risk of batch failures due to thermal runaways or sensitivity to moisture, leading to higher first-pass yields and more reliable delivery schedules for downstream customers relying on just-in-time inventory models.
- Scalability and Environmental Compliance: Scaling this process from laboratory grams to industrial tons is facilitated by the absence of exothermic hazards associated with strong dehydrating agents like POCl3. The reaction can be safely performed in standard stainless steel reactors equipped with standard heating and agitation systems, requiring no specialized corrosion-resistant lining needed for mercury or strong acid processes. From an environmental standpoint, the removal of persistent organic pollutants and heavy metals from the waste stream aligns perfectly with increasingly stringent global environmental regulations. This green chemistry profile not only mitigates regulatory risk but also enhances the corporate sustainability credentials of the manufacturing entity, a factor of growing importance for multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production pipelines or R&D programs.
Q: What are the advantages of this Pd-catalyzed method over traditional mercury-based synthesis?
A: Traditional methods often utilize highly toxic mercury catalysts (HgO or mercury acetate) and phosphorus pentasulfide, posing significant environmental and safety hazards. This novel patent CN114349684B employs a high-efficiency transition metal catalyst (Palladium) in a one-pot process, eliminating toxic heavy metal waste and simplifying the purification workflow significantly.
Q: What is the substrate scope for the R1 and R4 positions in this synthesis?
A: The method demonstrates wide substrate universality. R1 can be hydrogen, aryl, alkyl, sulfonyl, or acyl groups, while R4 accommodates alkyl, phenyl, or naphthyl groups. This flexibility allows for the rapid construction of diverse compound libraries for biological screening without needing complex protecting group strategies.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states suitability for industrialized large-scale production. The reaction uses readily available raw materials, operates under relatively mild temperatures (25-120°C), and utilizes a simple workup procedure involving extraction and distillation, which are standard unit operations in chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzo[c,d]indole Imine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed synthesis for accelerating drug discovery programs focused on oncology and cardiovascular health. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzo[c,d]indole imine derivative delivered meets the highest international standards for pharmaceutical intermediates. We are committed to leveraging our technical expertise to optimize this novel route for your specific needs, ensuring consistent quality and supply continuity.
We invite you to collaborate with us to unlock the full commercial potential of this advanced synthetic methodology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume, demonstrating exactly how this greener, more efficient process can improve your bottom line. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, and let us help you bring your next-generation therapeutics to market faster and more economically.
