Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Indoles for Commercial Pharmaceutical Manufacturing
Introduction to Novel Trifluoromethyl Indole Synthesis
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in patent CN115385895A, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted indole compounds. This technology represents a paradigm shift from conventional synthetic routes by employing a transition metal palladium-catalyzed double carbon-hydrogen activation strategy. The core innovation lies in the utilization of inexpensive and readily accessible starting materials, specifically trifluoroethylimidoyl chloride and unactivated olefins, to directly construct the indole core. This approach not only simplifies the operational complexity but also significantly broadens the substrate scope, allowing for the introduction of diverse functional groups at various positions on the indole ring. For research and development teams focused on drug discovery, this method offers a versatile platform for generating libraries of fluorinated indole derivatives, which are critical motifs in numerous bioactive molecules and commercial drugs such as indomethacin.
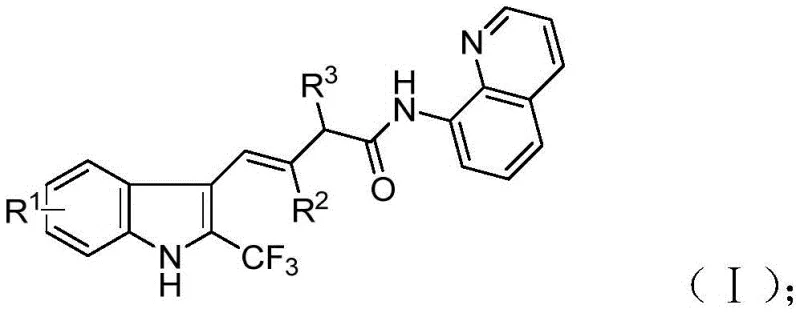
The structural versatility of the resulting compounds, as depicted in the general formula, allows for precise tuning of physicochemical properties. By varying the substituents R1, R2, and R3, chemists can optimize lipophilicity, electronegativity, and conformational preferences, which are essential parameters in lead optimization phases. The ability to synthesize these complex architectures directly from simple precursors without the need for pre-functionalized substrates marks a substantial advancement in synthetic efficiency. Furthermore, the method's compatibility with various functional groups ensures that late-stage functionalization is feasible, thereby accelerating the timeline from bench-scale synthesis to potential clinical candidate selection. This patent underscores the growing trend towards atom-economical and step-economical processes in modern organic synthesis, addressing the critical need for sustainable and cost-effective manufacturing pathways in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted indoles has been fraught with significant challenges that hinder their widespread application in industrial settings. Traditional literature reports primarily rely on three main strategies, each carrying distinct disadvantages that impact cost and scalability. The first common approach involves the coupling of pre-synthesized indole molecules with electrophilic trifluoromethylating agents, such as Togni's or Umemoto's reagents. While effective in small-scale laboratory settings, these reagents are notoriously expensive, sensitive to moisture, and often require strict anhydrous conditions, driving up the overall production cost. The second strategy employs Fischer indole synthesis using substrates that already bear a trifluoromethyl group; however, this limits the diversity of the final products and often necessitates multi-step sequences to install the fluorine motif prior to cyclization. The third category involves tandem cyclization reactions, which frequently suffer from harsh reaction conditions, low yields, and narrow substrate tolerance. These conventional methods collectively impose a heavy burden on process chemistry teams, who must navigate complex purification protocols and deal with significant amounts of hazardous waste generated by stoichiometric oxidants or activating agents.
The Novel Approach
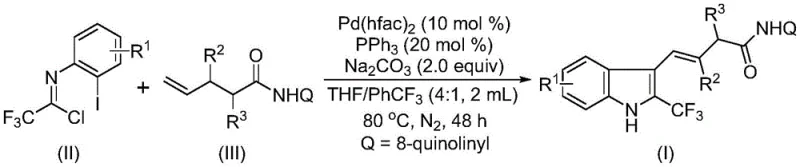
In stark contrast to these legacy methods, the technology described in patent CN115385895A introduces a streamlined catalytic cycle that bypasses the need for expensive reagents and harsh conditions. The novel approach leverages a palladium-catalyzed double C-H activation mechanism, utilizing trifluoroethylimidoyl chloride and unactivated alkenes as the primary building blocks. This strategy is fundamentally more economical because the starting materials are cheap, commercially available, and stable under ambient conditions. The reaction proceeds through a sophisticated cascade involving oxidative addition, migratory insertion, and beta-hydride elimination, ultimately forging the indole ring and installing the trifluoromethyl group in a single operational step. The use of an 8-aminoquinoline directing group on the alkene substrate facilitates the regioselective C-H activation, ensuring high fidelity in the formation of the desired product structure. This method eliminates the requirement for pre-activated substrates, thereby reducing the number of synthetic steps and minimizing waste generation. The operational simplicity is further enhanced by the use of a mixed solvent system that effectively dissolves all reactants, promoting high conversion rates without the need for exotic additives or extreme temperatures.
Mechanistic Insights into Pd-Catalyzed Double C-H Activation
The mechanistic pathway proposed for this transformation offers a fascinating glimpse into the power of modern organometallic catalysis. The reaction is initiated by the oxidative addition of a zero-valent palladium species into the aryl carbon-iodine bond of the trifluoroacetimide chloride precursor. This step generates a reactive divalent palladium intermediate, which is crucial for the subsequent catalytic turnover. The presence of the 8-aminoquinoline moiety on the alkene substrate plays a pivotal role as a bidentate directing group, coordinating to the palladium center and facilitating the migratory insertion of the olefin into the palladium-carbon bond. This coordination creates a stable six-membered palladacycle intermediate, which lowers the activation energy for the C-H cleavage event. Following the insertion, a beta-hydride elimination occurs, releasing the initial olefinic product and regenerating a palladium-hydride species. However, the process does not stop there; the system undergoes a second cycle of activation where the zero-valent palladium inserts into the carbon-chlorine bond of the imine chloride. This triggers a similar sequence of migratory insertion and beta-hydride elimination, coupled with an isomerization of the alkene chain. This tandem cascade effectively constructs the indole nucleus while simultaneously positioning the trifluoromethyl group at the 2-position with high regioselectivity. Understanding this intricate dance of bond-breaking and bond-forming events is essential for process chemists aiming to optimize reaction parameters and troubleshoot potential impurities during scale-up.
From an impurity control perspective, the mechanism suggests that side reactions could arise from incomplete oxidative addition or competing beta-hydride elimination pathways that lead to non-cyclized byproducts. However, the specific choice of ligands, such as triphenylphosphine, and the optimized solvent mixture of THF and trifluorotoluene, helps to stabilize the active catalytic species and suppress these off-cycle pathways. The use of sodium carbonate as a base is also critical, as it neutralizes the hydrochloric acid generated during the reaction, preventing the decomposition of acid-sensitive intermediates. The patent data indicates that maintaining the reaction at 80°C for 48 hours provides the necessary thermal energy to overcome the kinetic barriers of the C-H activation steps without causing significant degradation of the starting materials. This balance between reaction rate and stability is a key factor in achieving the reported yields, which range from moderate to high depending on the electronic nature of the substituents. For R&D directors, this mechanistic clarity provides confidence in the robustness of the process, allowing for rational design of analogues and prediction of potential scale-up challenges related to heat transfer and mixing efficiency in larger reactors.
How to Synthesize 2-Trifluoromethyl Indoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to maximize yield and purity. The protocol begins with the precise weighing of the palladium catalyst, specifically palladium hexafluoroacetylacetonate, along with the triphenylphosphine ligand and sodium carbonate base. These components are introduced into a reaction vessel, typically a Schlenk tube for small-scale optimization, along with the trifluoroethylimidoyl chloride and the alkene substrate. The choice of solvent is paramount; a mixture of tetrahydrofuran and trifluorotoluene in a volume ratio of approximately 4:1 is preferred to ensure complete solubility of all organic and inorganic components. Once the mixture is homogenized, the vessel is sealed and heated to 80°C under an inert nitrogen atmosphere to prevent oxidation of the palladium catalyst. The reaction is allowed to proceed for 48 hours, a duration determined to be optimal for balancing conversion efficiency with raw material stability. Post-reaction, the mixture is cooled, filtered to remove inorganic salts, and the filtrate is concentrated. The crude residue is then subjected to silica gel chromatography to isolate the pure 2-trifluoromethyl substituted indole compound. Detailed standardized synthesis steps follow below.
- Combine palladium catalyst (Pd(hfac)2), triphenylphosphine ligand, sodium carbonate additive, trifluoroethylimidoyl chloride, and the alkene substrate in an organic solvent mixture of THF and trifluorotoluene.
- Heat the reaction mixture to 80°C under a nitrogen atmosphere and maintain stirring for 48 hours to ensure complete conversion via double C-H activation.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product using column chromatography to isolate the high-purity 2-trifluoromethyl substituted indole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs. By replacing expensive, specialized fluorinating reagents with commodity chemicals like trifluoroethylimidoyl chloride and simple alkenes, the direct material cost of goods sold (COGS) is significantly lowered. This shift away from proprietary reagents also mitigates supply chain risks associated with single-source suppliers, as the new starting materials are widely available from multiple global vendors. Furthermore, the simplified operational protocol, which avoids cryogenic conditions or high-pressure equipment, reduces the capital expenditure required for manufacturing infrastructure. The process operates at atmospheric pressure and moderate temperatures, meaning existing standard glass-lined or stainless steel reactors can be utilized without major modifications. This compatibility with standard equipment accelerates the technology transfer from R&D to commercial production, shortening the time-to-market for new drug candidates containing this scaffold.
- Cost Reduction in Manufacturing: The elimination of costly electrophilic trifluoromethylating agents and the reduction in synthetic steps directly translate to substantial cost savings. The use of a catalytic amount of palladium, rather than stoichiometric amounts of expensive reagents, further optimizes the cost structure. Additionally, the simplified workup procedure, involving basic filtration and chromatography, reduces labor hours and solvent consumption compared to multi-step purification processes required by older methods. These factors collectively contribute to a more lean and efficient manufacturing process, enhancing the overall profit margin for the final API or intermediate.
- Enhanced Supply Chain Reliability: Relying on broadly available starting materials ensures a stable and resilient supply chain. Unlike specialized reagents that may face availability bottlenecks or long lead times, the precursors for this reaction are produced at scale by the bulk chemical industry. This abundance guarantees consistent supply continuity, which is critical for meeting the rigorous delivery schedules of pharmaceutical clients. The robustness of the reaction conditions also means that production is less susceptible to disruptions caused by minor variations in utility supplies or environmental conditions, providing a reliable source of high-quality intermediates for downstream synthesis.
- Scalability and Environmental Compliance: The method is explicitly designed with scalability in mind, having been demonstrated to work effectively at the gram level with potential for ton-scale production. The use of a defined solvent system and the absence of highly toxic or explosive reagents simplify waste management and regulatory compliance. The process generates less hazardous waste compared to traditional methods that utilize strong oxidants or generate heavy metal byproducts. This alignment with green chemistry principles not only reduces disposal costs but also supports the sustainability goals of modern pharmaceutical companies, making the supply of these intermediates more attractive to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing a reliable basis for decision-making. Understanding the nuances of substrate compatibility, catalyst loading, and purification is essential for successfully integrating this technology into existing manufacturing workflows. The responses highlight the flexibility of the method and its suitability for producing a wide array of structurally diverse indole derivatives.
Q: What are the key advantages of this palladium-catalyzed method over traditional trifluoromethylation?
A: Unlike traditional methods requiring expensive electrophilic reagents like Togni's or Umemoto's reagents, this process utilizes cheap and readily available trifluoroethylimidoyl chloride and unactivated alkenes. It operates under milder conditions with broader substrate compatibility and avoids harsh pre-activation steps.
Q: What is the optimal solvent system and temperature for this reaction?
A: The patent specifies that a mixed solvent system of tetrahydrofuran (THF) and trifluorotoluene (PhCF3) in a volume ratio of roughly 4:1 provides the highest conversion rates. The reaction is optimally conducted at 80°C for 48 hours to balance efficiency and原料 stability.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the methodology is explicitly designed for scalability. The patent notes that the process can be expanded to the gram level and potentially beyond, utilizing commercially available catalysts and simple post-treatment procedures like filtration and column chromatography, making it suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in patent CN115385895A for the future of pharmaceutical manufacturing. As a leading CDMO partner, we possess the technical expertise and infrastructure to seamlessly adopt and scale such innovative pathways. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is smooth and efficient. We are committed to delivering high-purity 2-trifluoromethyl indole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex organometallic chemistry, including palladium-catalyzed C-H activations, positions us as an ideal partner for developing cost-effective and scalable supply chains for next-generation therapeutics.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your drug development programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs, helping you identify opportunities for efficiency and budget optimization. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Let us help you accelerate your timeline to market with reliable, high-quality chemical solutions.
