Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Indoles for Commercial Scale
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Indoles for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds with high efficiency and minimal environmental impact. A significant breakthrough in this domain is detailed in patent CN107619385B, which discloses a novel method for synthesizing 2-trifluoromethyl indoles via palladium-catalyzed intramolecular amination of aryl enamines. This technology addresses the critical demand for fluorinated indole derivatives, which are pivotal structural motifs in numerous bioactive molecules and drug candidates due to the metabolic stability and lipophilicity conferred by the trifluoromethyl group. By leveraging a catalytic amount of Pd(OAc)2 under mild oxidative conditions, this process offers a streamlined pathway that eliminates the need for harsh reagents or complex instrumentation, positioning it as a superior alternative for the reliable pharmaceutical intermediate supplier seeking to optimize their production lines.
The strategic importance of this synthesis route cannot be overstated for R&D directors and procurement managers alike. Traditional methods for introducing trifluoromethyl groups onto indole cores often suffer from poor atom economy, require expensive electrophilic trifluoromethylating agents, or involve multi-step sequences that erode overall yield. In contrast, the approach outlined in the patent utilizes N-trifluoromethyl alkenyl arylamines as direct precursors, effectively building the indole core and installing the fluorine functionality in a single convergent step. This not only simplifies the synthetic tree but also significantly reduces the accumulation of waste and the consumption of raw materials, aligning perfectly with modern green chemistry principles and cost reduction in pharmaceutical intermediate manufacturing goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl indoles has been plagued by significant operational challenges that hinder scalability and economic viability. Conventional strategies often rely on the electrophilic substitution of trifluoromethyl radicals or the use of specialized trifluoromethyl electrophiles, which are frequently hazardous, unstable, and prohibitively expensive for large-scale applications. Furthermore, earlier cyclization strategies required rigorous exclusion of moisture and oxygen, necessitating specialized glovebox techniques or high-pressure reactors that increase capital expenditure and operational complexity. These legacy methods often result in broad impurity profiles due to non-selective radical pathways, forcing downstream purification teams to engage in laborious chromatographic separations that drastically lower the final isolated yield and extend the production lead time for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology described in patent CN107619385B represents a paradigm shift by employing a palladium-catalyzed C-H activation strategy that operates under remarkably benign conditions. Instead of relying on stoichiometric amounts of toxic oxidants or extreme temperatures, this novel approach utilizes molecular oxygen (1 atm) as the terminal oxidant, which is not only cost-effective but also generates water as the sole byproduct, thereby enhancing the environmental profile of the synthesis. The reaction proceeds smoothly at 100°C in common solvents like DMSO or toluene, utilizing a simple catalyst system of Pd(OAc)2 and zinc acetate additives. This simplicity translates directly into commercial advantages, as it allows for the commercial scale-up of complex polymer additives and fine chemicals without the need for retrofitting existing reactor infrastructure with high-pressure capabilities.
As illustrated in the reaction scheme above, the transformation is highly versatile, accommodating a wide array of substituents on both the aromatic ring attached to the nitrogen and the alkene moiety. The patent data confirms that electron-rich groups such as methyl and methoxy, as well as electron-deficient groups like fluoro and chloro, are well-tolerated, yielding the desired 2-trifluoromethyl indole products in yields ranging up to 84%. This broad substrate scope is crucial for medicinal chemists who require rapid access to diverse analog libraries for structure-activity relationship (SAR) studies. The ability to synthesize these derivatives in a single pot with high selectivity minimizes the formation of regioisomers, ensuring that the final product meets the stringent purity specifications required for clinical trial materials and eventual commercial drug substance production.
Mechanistic Insights into Pd-Catalyzed Intramolecular Amination
Understanding the catalytic cycle is essential for R&D teams aiming to further optimize this process or adapt it to novel substrates. The mechanism initiates with the coordination of the palladium(II) acetate catalyst to the nitrogen atom of the N-trifluoromethyl alkenyl arylamine substrate, accompanied by the loss of one molecule of acetic acid to form a key organopalladium intermediate. This coordination activates the proximal C-H bond on the aromatic ring, facilitating an intramolecular cyclization event that constructs the five-membered indole ring. The resulting cyclopalladated complex then undergoes reductive elimination to release the final 2-trifluoromethyl indole product and generate a zero-valent palladium species, Pd(0). This step is critical as it regenerates the active catalytic species, allowing the cycle to continue with minimal metal loading.
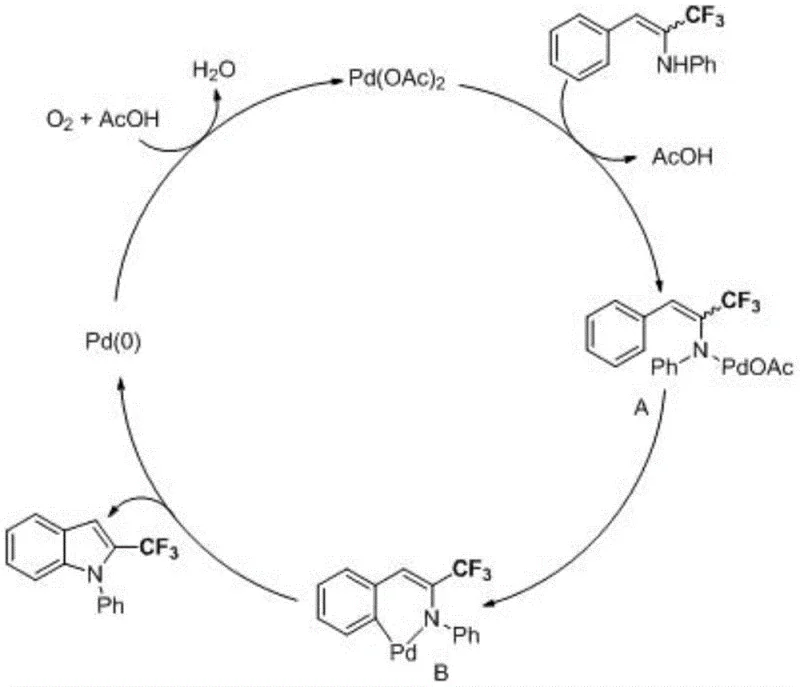
The sustainability of the cycle is maintained by the re-oxidation of Pd(0) back to Pd(II) by molecular oxygen in the presence of acetic acid, completing the catalytic loop without the accumulation of inactive metal black. This mechanistic feature is particularly advantageous for impurity control, as the homogeneous nature of the catalysis ensures consistent reaction kinetics throughout the batch, reducing the risk of hot-spots or incomplete conversions that often lead to difficult-to-remove side products. For quality assurance teams, this predictable mechanism means that critical process parameters (CPPs) such as oxygen flow rate and temperature can be tightly controlled to ensure batch-to-batch consistency, a fundamental requirement for regulatory compliance in the manufacture of high-purity OLED material and pharmaceutical intermediates.
How to Synthesize 2-Trifluoromethyl Indole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the additives and the management of the oxygen atmosphere to maximize turnover numbers. The standard protocol involves charging the reaction vessel with the N-trifluoromethyl alkenyl arylamine substrate, 10 mol % Pd(OAc)2, 2 equivalents of Zn(OAc)2, and activated 4A molecular sieves to scavenge trace water which could inhibit the catalyst. The mixture is suspended in a 2:1 ratio of DMSO to toluene, and the system is purged and filled with oxygen before heating to 100°C for 12 hours. Detailed standardized synthesis steps for this procedure are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Combine N-trifluoromethyl alkenyl arylamine substrate with 10 mol % Pd(OAc)2 catalyst, additives like Zn(OAc)2, and 4A molecular sieves in a DMSO/Toluene solvent mixture.
- Introduce an oxygen atmosphere (1 atm) to the reaction vessel and stir the mixture at 100°C for approximately 12 hours to facilitate cyclization.
- Upon completion, filter the reaction mixture, extract with organic solvents, dry over anhydrous MgSO4, and purify the residue via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this palladium-catalyzed methodology offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials; unlike traditional methods that may require custom-synthesized trifluoromethylating reagents with long lead times, this process utilizes commodity chemicals like palladium acetate and zinc acetate which are readily available from multiple global suppliers. This diversification of the supply base mitigates the risk of production stoppages due to single-source bottlenecks, ensuring enhanced supply chain reliability for critical intermediates. Furthermore, the use of atmospheric oxygen as the oxidant eliminates the need for purchasing, storing, and handling hazardous chemical oxidants, thereby reducing logistics costs and safety compliance burdens.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the use of catalytic rather than stoichiometric amounts of precious metal, combined with the elimination of expensive specialty reagents. By avoiding the need for cryogenic conditions or high-pressure autoclaves, the method significantly lowers energy consumption and capital depreciation costs associated with specialized equipment maintenance. Additionally, the simplified workup procedure, which involves basic filtration and extraction rather than complex distillation or recrystallization sequences, reduces labor hours and solvent usage, leading to substantial cost savings in the overall cost of goods sold (COGS) for the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply security, as the process is less sensitive to minor fluctuations in temperature or reagent quality compared to sensitive organometallic reactions. The tolerance for various functional groups means that a single platform technology can be used to produce a wide family of derivatives, allowing manufacturers to respond quickly to changing market demands without requalifying entirely new synthetic routes. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates, enabling faster time-to-market for downstream drug products and providing a competitive edge in the fast-paced biopharmaceutical sector.
- Scalability and Environmental Compliance: From an environmental perspective, the generation of water as the primary byproduct and the absence of halogenated waste streams simplify wastewater treatment protocols, aligning with increasingly strict global environmental regulations. The process is inherently scalable, having been demonstrated to work efficiently from milligram to gram scales in the patent examples, with a clear path to kilogram and tonne production through standard batch reactor engineering. This scalability ensures that the technology can support the commercial scale-up of complex fine chemicals without encountering the mass transfer or heat dissipation issues that often plague heterogeneous or gas-liquid reactions at larger volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed synthesis route. These insights are derived directly from the experimental data and mechanistic understanding presented in the patent literature, providing a solid foundation for process development teams evaluating this technology for adoption. Understanding these nuances is critical for ensuring successful technology transfer and maintaining high standards of product quality throughout the manufacturing lifecycle.
Q: What are the optimal reaction conditions for this Pd-catalyzed amination?
A: The optimal conditions involve using 10 mol % Pd(OAc)2 as the catalyst with 2 equivalents of Zn(OAc)2 as an additive in a DMSO:Toluene (2:1) solvent system under 1 atmosphere of oxygen at 100°C for 12 hours.
Q: Can this method tolerate diverse substituents on the aryl ring?
A: Yes, the methodology demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as fluoro, chloro, and trifluoromethyl on both the aniline nitrogen and the alkene phenyl ring.
Q: Is the process suitable for large-scale manufacturing?
A: Absolutely. The process utilizes readily available reagents, avoids specialized high-pressure equipment by using balloon oxygen, and employs a simple workup procedure involving filtration and chromatography, making it highly amenable to kilogram-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN107619385B for driving innovation in drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the bench to the plant. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl indole delivered meets the highest industry standards for potency and impurity profiles, thus safeguarding your downstream processing and final product efficacy.
We invite you to collaborate with our technical team to explore how this efficient synthesis route can be tailored to your specific project needs. By engaging with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic feasibility of implementing this route versus your current supply chain. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in delivering high-quality chemical solutions that accelerate your path to market success.
