Advanced Catalytic Halogenation for High-Purity Pharmaceutical Intermediates
Advanced Catalytic Halogenation for High-Purity Pharmaceutical Intermediates
The development of efficient synthetic routes for aryl halides remains a cornerstone challenge in modern organic chemistry, particularly for the production of complex pharmaceutical intermediates and agrochemical building blocks. Patent CN114292153A introduces a groundbreaking methodology that addresses the longstanding limitations of traditional halogenation strategies by employing a tertiary amine catalyst system. This innovation enables the direct halogenation of aromatic and heterocyclic hydrocarbons using N-halosuccinimides or dihalohydantoins under remarkably mild conditions. By shifting away from hazardous elemental halogens and corrosive hydrogen halide by-products, this technology offers a safer, more sustainable pathway for generating high-purity aryl halides. For R&D directors and process chemists, this represents a significant leap forward in controlling impurity profiles while maintaining high atom economy. The ability to operate at ambient temperatures with inexpensive catalysts further underscores the potential for substantial cost reduction in pharmaceutical intermediate manufacturing, making it an attractive option for large-scale commercial adoption.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl halides has relied heavily on the use of elemental halogens such as chlorine or bromine gas, which pose severe safety hazards due to their high toxicity and corrosivity. These traditional processes often require specialized equipment to handle volatile gases and necessitate rigorous scrubbing systems to manage the evolution of corrosive hydrogen halide by-products. Furthermore, alternative methods utilizing inorganic halides often demand harsh oxidizing conditions and elevated temperatures, leading to poor regioselectivity and the formation of complex impurity profiles that are difficult to separate. The reliance on expensive activating reagents for N-halosuccinimides in previous iterations also added unnecessary cost and complexity to the supply chain. For procurement managers, these factors translate into higher raw material costs, increased waste disposal fees, and potential supply chain disruptions due to the regulated nature of hazardous chemicals. The operational burden of managing such dangerous reagents significantly impacts the overall efficiency and environmental compliance of manufacturing facilities.
The Novel Approach
The methodology disclosed in CN114292153A fundamentally transforms this landscape by introducing a catalytic system that activates mild halogenating agents through simple tertiary amines. This approach eliminates the need for elemental halogens and strong oxidants, thereby drastically simplifying the reaction setup and post-treatment procedures. The use of commercially available and inexpensive catalysts like triethylenediamine ensures that the process remains economically viable even at large scales. Reaction conditions are exceptionally mild, typically proceeding at room temperature (25°C) in common solvents like dichloromethane, which reduces energy consumption and thermal stress on sensitive functional groups. This gentle yet effective protocol allows for the precise installation of chlorine, bromine, or iodine atoms onto diverse aromatic scaffolds with high selectivity. For supply chain heads, this translates to a more robust and reliable aryl halide supplier capability, as the raw materials are stable solids rather than hazardous gases, ensuring consistent quality and easier logistics management.
Mechanistic Insights into Tertiary Amine Catalyzed Halogenation
The core of this technological advancement lies in the unique activation mechanism wherein the tertiary amine catalyst interacts with the N-halosuccinimide reagent to generate a highly reactive electrophilic species in situ. Unlike uncatalyzed reactions where N-halosuccinimides exhibit low reactivity towards electron-rich aromatics, the presence of the amine facilitates the polarization of the nitrogen-halogen bond. This interaction lowers the activation energy barrier for the electrophilic aromatic substitution, allowing the reaction to proceed rapidly at ambient temperatures without the need for external heating or Lewis acid promoters. The catalytic cycle is efficient, with the amine regenerating after the halogen transfer, meaning only catalytic amounts (molar ratios as low as 1:0.01 relative to substrate) are required to drive the transformation to completion. This mechanistic efficiency is critical for minimizing metal contamination and simplifying the purification process, a key concern for producing high-purity OLED material or API intermediates where trace impurities can be detrimental.
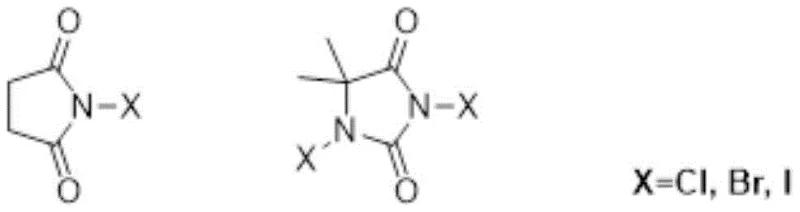
Furthermore, the selectivity of this catalytic system is governed by the electronic properties of the aromatic substrate and the specific nature of the halogenating agent. The mild electrophilic character of the activated species ensures that halogenation occurs preferentially at the most electron-rich positions, such as the para- or ortho-positions relative to activating groups like methoxy or amino substituents. This inherent regioselectivity minimizes the formation of isomeric by-products, which are often the most challenging impurities to remove during downstream processing. The tolerance for various functional groups, including esters, aldehydes, and protected amines, demonstrates the versatility of this method for late-stage functionalization of complex molecules. For R&D teams, understanding this mechanism allows for the rational design of synthetic routes that avoid protecting group manipulations, thereby shortening the overall synthesis timeline. The ability to control the degree of halogenation simply by adjusting the stoichiometry of the reagent provides an additional layer of process control that is essential for commercial scale-up of complex polymer additives or fine chemicals.
How to Synthesize Aryl Halides Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting involves a straightforward protocol that leverages the stability and ease of handling of the reagents. The process begins with the dissolution of the aromatic substrate and the tertiary amine catalyst in a suitable organic solvent, followed by the controlled addition of the N-halosuccinimide reagent. Detailed standardized synthesis steps are provided below to ensure reproducibility and safety during operation. The reaction progress can be easily monitored via TLC or HPLC, and upon completion, the workup involves simple solvent removal and chromatographic purification. This simplicity makes it an ideal candidate for technology transfer and rapid scale-up, reducing the time-to-market for new drug candidates.
- Prepare the reaction mixture by combining the aromatic substrate, N-halosuccinimide (or dihalohydantoin), and a tertiary amine catalyst such as triethylenediamine in a suitable solvent like dichloromethane.
- Stir the reaction mixture at room temperature (approximately 25°C) under an air atmosphere for a duration ranging from 0.5 to 6 hours, depending on the substrate reactivity.
- Upon completion, remove the solvent via concentration and purify the resulting aryl halide product using standard column chromatography techniques to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic halogenation technology offers profound benefits that extend beyond mere chemical efficiency. For procurement managers, the shift from hazardous elemental halogens to stable solid reagents like N-bromosuccinimide significantly reduces the regulatory burden and insurance costs associated with storing and transporting dangerous goods. The elimination of corrosive gas scrubbing systems and specialized containment infrastructure leads to substantial capital expenditure savings for manufacturing facilities. Moreover, the use of inexpensive tertiary amine catalysts, which are commodity chemicals, ensures that the raw material cost base remains low and stable, shielding the supply chain from volatility associated with specialty reagents. This economic stability is crucial for long-term contracting and budget forecasting in the competitive pharmaceutical and agrochemical sectors.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive activation reagents and the reduction of energy consumption due to ambient temperature operation. By avoiding the need for cryogenic cooling or high-temperature heating, utility costs are significantly lowered, contributing to a leaner manufacturing cost structure. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, improving the overall yield and reducing the cost per kilogram of the final active ingredient. The simplified post-treatment process, which avoids complex neutralization steps for acidic by-products, further reduces labor and waste disposal expenses, creating a compelling value proposition for cost-sensitive production runs.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable reagents enhances the resilience of the supply chain against disruptions. Unlike elemental halogens which may face strict transportation regulations and supply constraints, N-halosuccinimides and tertiary amines are readily sourced from multiple global suppliers. This diversification of the supply base reduces the risk of single-source dependency and ensures continuous production capability even during market fluctuations. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, providing a consistent output that meets stringent quality specifications. For supply chain heads, this reliability translates into shorter lead times for high-purity aryl halides and greater confidence in meeting delivery commitments to downstream customers.
- Scalability and Environmental Compliance: The green chemistry attributes of this method align perfectly with increasingly strict environmental regulations and corporate sustainability goals. The absence of heavy metal catalysts and toxic gas emissions simplifies the permitting process for new manufacturing lines and reduces the environmental footprint of the facility. The reaction generates benign succinimide by-products which are easier to handle and dispose of compared to inorganic salt waste from traditional methods. This environmental compatibility facilitates smoother regulatory approvals and enhances the brand reputation of the manufacturer as a responsible partner. The demonstrated scalability from milligram to multi-gram scales in the patent data suggests that the process can be seamlessly transferred to multi-ton production without significant re-engineering, ensuring a smooth path from R&D to commercial manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halogenation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for potential partners and licensees. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of this tertiary amine catalyzed halogenation method over traditional methods?
A: Unlike traditional methods that rely on toxic elemental halogens or harsh oxidizing conditions, this patented approach utilizes mild N-halosuccinimides activated by inexpensive tertiary amines. This results in significantly improved operational safety, reduced corrosive by-products, and excellent regioselectivity for electron-rich aromatic systems.
Q: Is this synthesis method scalable for industrial production of aryl halides?
A: Yes, the patent explicitly demonstrates scalability. The reaction proceeds efficiently at room temperature without the need for cryogenic cooling or high-pressure equipment. The use of common solvents like dichloromethane and stable solid reagents facilitates easy handling and scale-up from gram to multi-kilogram batches.
Q: What types of substrates are compatible with this halogenation protocol?
A: The method exhibits broad substrate scope, successfully halogenating various electron-rich aromatics including substituted benzenes, naphthalenes, indoles, and thiophenes. It effectively introduces chlorine, bromine, or iodine atoms, making it versatile for synthesizing diverse pharmaceutical and agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Halide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN114292153A for the production of high-value chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical clients. We are committed to leveraging such efficient synthetic strategies to deliver cost-effective and high-quality aryl halides that accelerate our partners' drug development timelines.
We invite you to collaborate with us to explore how this technology can optimize your specific supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements. Please contact us to request specific COA data and route feasibility assessments for your target molecules, and let us demonstrate how our expertise can drive value and efficiency in your manufacturing operations.
