Scalable Visible Light Nickel Catalysis for High-Purity Aryl Halide Manufacturing
Scalable Visible Light Nickel Catalysis for High-Purity Aryl Halide Manufacturing
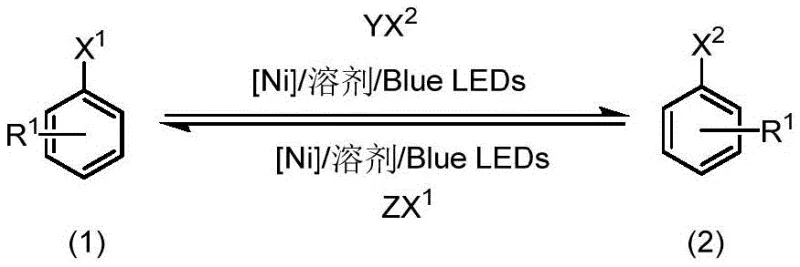
The chemical industry is constantly seeking more efficient pathways to synthesize critical building blocks, and the recent disclosure in patent CN115010563B represents a significant leap forward in the preparation of aryl halides. This intellectual property details a novel methodology utilizing visible light-promoted nickel catalysis to facilitate halogen exchange reactions, effectively transforming less reactive aryl chlorides or bromides into highly valuable aryl iodides or alternative bromides. For R&D directors and procurement specialists alike, this technology addresses the perennial challenge of accessing diverse halogenated scaffolds without resorting to harsh, energy-intensive conditions. By leveraging the unique properties of endogenous nickel-bipyridine complexes under blue LED irradiation, this process achieves high yields while maintaining exceptional operational simplicity. The implications for supply chain stability are profound, as it enables the production of high-purity pharmaceutical intermediates with a drastically reduced environmental footprint compared to legacy thermal methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the interconversion of halogen atoms on aromatic rings has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional protocols often rely on copper or nickel mediators that require stoichiometric quantities of metal, leading to substantial waste generation and difficult purification challenges due to heavy metal residues. Furthermore, these classical reactions typically demand high reaction temperatures and the use of high-boiling, polar aprotic solvents such as DMF or DMSO, which are notoriously difficult to remove and pose serious environmental and safety concerns during disposal. The harsh conditions necessary to drive these transformations frequently result in poor functional group tolerance, causing decomposition of sensitive substrates and lowering the overall purity of the final product. Consequently, manufacturers face inflated costs related to energy consumption, solvent recovery, and extensive downstream processing to meet stringent regulatory limits on impurities.
The Novel Approach
In stark contrast to these archaic techniques, the visible light-promoted nickel-catalyzed method described in the patent data offers a transformative solution that aligns perfectly with modern green chemistry principles. By employing a catalytic amount of nickel complexed with bipyridine ligands and activating the system with blue LEDs, the reaction proceeds smoothly at mild temperatures ranging from 25°C to 70°C. This approach eliminates the need for stoichiometric metal promoters and avoids the use of toxic, high-boiling solvents, favoring instead greener ether-based alternatives like cyclopentyl methyl ether (CPME) or 1,4-dioxane. The mildness of the conditions ensures that sensitive functional groups remain intact, thereby expanding the scope of accessible chemical space for drug discovery teams. Moreover, the ability to drive the reaction to completion through the precipitation of insoluble salts provides a thermodynamic driving force that enhances conversion rates without requiring excessive heat or pressure.
Mechanistic Insights into Visible Light Promoted Nickel-Catalyzed Halogen Exchange
The core of this technological breakthrough lies in the sophisticated interplay between the nickel catalyst, the bipyridine ligand, and the photon energy supplied by the blue LED source. Upon irradiation, the nickel complex enters an excited state that facilitates the oxidative addition of the aryl halide substrate, a step that is traditionally the rate-limiting barrier in thermal processes. The ligand environment, specifically optimized with substituents like tert-butyl groups on the bipyridine ring, stabilizes the active nickel species and prevents catalyst deactivation through aggregation. This photoredox manifold allows for the reversible exchange of halogen atoms, where the equilibrium is strategically shifted towards the desired product by the formation of less soluble salt byproducts. For the R&D director, understanding this mechanism is crucial as it highlights the robustness of the catalytic cycle against common poisons and the ability to fine-tune reactivity through ligand modification. The result is a highly selective transformation that minimizes side reactions such as homocoupling or dehalogenation, ensuring a clean impurity profile.
Furthermore, the control over impurity generation is inherently built into the reaction design through the choice of solvent and halogen source. The use of ether solvents with lower polarity not only aids in the solubility of the organic substrates but also promotes the precipitation of inorganic salts, which drives the equilibrium forward according to Le Chatelier's principle. This physical separation of byproducts simplifies the workup procedure significantly, as filtration can often replace complex aqueous extractions or chromatographic purifications. From a quality control perspective, this means that the final aryl halide products exhibit superior purity levels with minimal residual metal content, a critical parameter for API intermediate manufacturing. The mechanistic pathway also suggests that the reaction is tolerant to a wide array of electronic environments on the aromatic ring, accommodating electron-withdrawing groups like cyano and aldehyde functionalities as well as electron-donating groups like methoxy and alkyl chains without significant loss in efficiency.
How to Synthesize Aryl Halides Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalyst system and the maintenance of an inert atmosphere to prevent oxidation of the low-valent nickel species. The standard protocol involves pre-mixing the nickel source, such as bis(1,5-cyclooctadiene)nickel, with the bipyridine ligand in the chosen solvent before introducing the substrates. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the catalyst system by mixing a nickel source such as Ni(COD)2 with a bipyridine ligand like dtbbpy in an ether solvent under nitrogen protection.
- Add the aryl halide substrate and the halogen exchange salt (e.g., sodium iodide or tetrabutylammonium bromide) to the reaction mixture.
- Seal the vessel and irradiate with blue LEDs (410-480nm) at temperatures between 25-70°C for 12 to 48 hours to achieve high conversion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light nickel-catalyzed technology translates directly into tangible operational efficiencies and risk mitigation strategies. The shift away from high-energy thermal processes to ambient temperature photochemistry results in a substantial reduction in utility costs, as there is no longer a need for extensive heating or cooling infrastructure to maintain extreme reaction conditions. Additionally, the replacement of hazardous high-boiling solvents with safer, lower-boiling ethers simplifies solvent recovery and recycling loops, further driving down the cost of goods sold. The robustness of the catalyst system means that raw material variability has less impact on the final outcome, ensuring consistent batch-to-batch quality which is essential for maintaining reliable supply chains. By minimizing the number of unit operations required for purification, manufacturers can significantly increase throughput capacity without expanding their physical footprint.
- Cost Reduction in Manufacturing: The elimination of stoichiometric metal reagents and the reduction in catalyst loading to merely 5-10 mol% drastically lowers the raw material expenditure per kilogram of product. Furthermore, the avoidance of expensive and difficult-to-remove polar solvents reduces the burden on waste treatment facilities and lowers the overall environmental compliance costs associated with production. The energy savings derived from operating at room temperature or mild heating conditions contribute to a leaner manufacturing budget, allowing for more competitive pricing structures in the global market. These cumulative effects create a compelling economic case for transitioning existing halogen exchange processes to this newer, more efficient methodology.
- Enhanced Supply Chain Reliability: The reagents required for this transformation, including nickel salts, bipyridine ligands, and common halogen sources like sodium iodide, are commodity chemicals with stable and abundant global supply chains. This reduces the risk of production delays caused by the scarcity of specialized or exotic reagents that often plague niche synthetic routes. The simplicity of the reaction setup, which does not require high-pressure vessels or specialized heating mantles, allows for greater flexibility in manufacturing site selection and equipment utilization. Consequently, lead times for high-purity pharmaceutical intermediates can be shortened, enabling faster response to market demands and reducing inventory holding costs for downstream customers.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but the use of LED arrays allows for modular expansion where light intensity can be precisely controlled regardless of reactor volume. The use of greener solvents like CPME aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and worker safety exposure limits. The generation of solid salt byproducts that can be easily filtered off minimizes the volume of liquid waste streams, simplifying effluent treatment and reducing the environmental impact of the manufacturing process. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the brand value of the final product in markets that prioritize green chemistry credentials.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this halogen exchange technology based on the patent specifications. These answers are derived directly from the experimental data and claims to provide clarity on process capabilities and limitations for potential partners.
Q: What are the advantages of visible light promotion in this halogen exchange?
A: Visible light promotion allows the reaction to proceed at significantly lower temperatures (25-70°C) compared to traditional thermal methods, reducing energy consumption and improving functional group tolerance.
Q: Can this method convert aryl chlorides to aryl iodides?
A: Yes, the patent data demonstrates successful conversion of various aryl chlorides and bromides to their corresponding iodides or bromides using specific halogen sources like sodium iodide.
Q: Is the catalyst loading economical for large scale production?
A: The method utilizes low catalyst loadings, typically ranging from 5 mol% to 10 mol%, which significantly reduces the cost of goods compared to stoichiometric metal processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Halides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light nickel catalysis in modernizing the production of critical chemical building blocks. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical clients. We are committed to leveraging technologies like the one described in CN115010563B to deliver superior value through enhanced efficiency and sustainability.
We invite you to engage with our technical procurement team to discuss how this advanced halogen exchange method can be integrated into your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener protocol for your specific targets. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless transition to a more efficient and reliable sourcing strategy.
