Advanced Synthesis of 2-Aminobenzimidazole Derivatives for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced Synthesis of 2-Aminobenzimidazole Derivatives for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries rely heavily on heterocyclic compounds, with the 2-aminobenzimidazole scaffold serving as a critical pharmacophore in numerous therapeutic agents ranging from antiparasitics to anticancer drugs. As detailed in patent CN102060780A, a novel preparation method has been developed that addresses the longstanding inefficiencies associated with traditional synthesis routes. This technology leverages a sophisticated dehydration cyclization strategy using phosphorus-based reagents to construct the benzimidazole ring with exceptional precision. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic nuances and commercial implications of this patent is essential. The method not only promises higher purity profiles by avoiding heavy metal contaminants but also offers a streamlined pathway that is inherently more suitable for the rigorous demands of modern Good Manufacturing Practice (GMP) environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the most prevalent method for preparing 2-aminobenzimidazole derivatives involved the condensation of 2-(2-aminoaryl)thiourea with disubstituted carbodiimides followed by desulfurization cyclization. This legacy approach is fraught with significant operational and environmental drawbacks that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Specifically, the desulfurization step necessitates the use of highly toxic reagents such as red mercury, cuprous oxide, chlorine, or methyl iodide. These substances pose severe safety risks to personnel and create substantial waste disposal challenges, leading to increased regulatory compliance costs. Furthermore, the operational complexity of removing trace heavy metals from the final product to meet stringent purity specifications often results in lower overall yields and extended production cycles. Another alternative method involving the condensation of 2-chlorobenzimidazole with anilines requires harsh reaction conditions, typically between 140°C and 150°C, which often leads to low yields and a proliferation of difficult-to-separate byproducts, thereby compromising the economic viability of the process.
The Novel Approach
In stark contrast to these archaic techniques, the novel approach outlined in the patent utilizes a mild and efficient intramolecular dehydration cyclization of urea derivatives. This method begins with the reaction of a protected o-phenylenediamine with an N-phenoxy carbonyl amine to form a urea intermediate, which is then cyclized using dichlorotriphenylphosphorus or dibromotriphenylphosphorus. 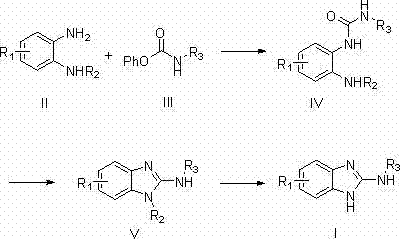 This strategic shift eliminates the need for toxic sulfur-removal agents entirely, replacing them with phosphorus-based dehydrating agents that can be generated in situ from safe and readily available precursors like triphenylphosphine oxide and oxalyl chloride. The reaction proceeds under significantly milder conditions, typically ranging from -30°C to 50°C, which minimizes thermal degradation and side reactions. This results in a much cleaner impurity profile and substantially higher yields, often exceeding 80% in the reported examples, making it a superior choice for cost reduction in API manufacturing where yield optimization is directly correlated to profitability.
This strategic shift eliminates the need for toxic sulfur-removal agents entirely, replacing them with phosphorus-based dehydrating agents that can be generated in situ from safe and readily available precursors like triphenylphosphine oxide and oxalyl chloride. The reaction proceeds under significantly milder conditions, typically ranging from -30°C to 50°C, which minimizes thermal degradation and side reactions. This results in a much cleaner impurity profile and substantially higher yields, often exceeding 80% in the reported examples, making it a superior choice for cost reduction in API manufacturing where yield optimization is directly correlated to profitability.
Mechanistic Insights into Phosphorus-Mediated Dehydration Cyclization
The core innovation of this synthesis lies in the activation of the urea functionality to facilitate ring closure. The mechanism involves the interaction of the urea oxygen with the electrophilic phosphorus species, such as dichlorotriphenylphosphorus, which acts as a potent dehydrating agent. This activation converts the hydroxyl group into a good leaving group, enabling the nucleophilic attack by the adjacent amino nitrogen on the carbonyl carbon. 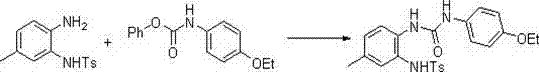 This intramolecular cyclization is driven forward by the formation of the stable aromatic benzimidazole system and the precipitation of phosphorus byproducts. The use of organic bases, such as triethylamine or pyridine, is critical in this step to neutralize the acid generated during the reaction and to maintain the nucleophilicity of the amine. The patent specifies that the phosphorus reagents can be prepared fresh in the reaction vessel, ensuring high reactivity and minimizing the decomposition issues often associated with storing sensitive halogenated phosphorus compounds. This in-situ generation capability is a key factor in enhancing the robustness of the process for industrial applications.
This intramolecular cyclization is driven forward by the formation of the stable aromatic benzimidazole system and the precipitation of phosphorus byproducts. The use of organic bases, such as triethylamine or pyridine, is critical in this step to neutralize the acid generated during the reaction and to maintain the nucleophilicity of the amine. The patent specifies that the phosphorus reagents can be prepared fresh in the reaction vessel, ensuring high reactivity and minimizing the decomposition issues often associated with storing sensitive halogenated phosphorus compounds. This in-situ generation capability is a key factor in enhancing the robustness of the process for industrial applications.
From an impurity control perspective, this mechanism offers distinct advantages over thiourea-based routes. Since no sulfur atoms are introduced into the reaction matrix, there is zero risk of sulfur-containing impurities or residual heavy metals from desulfurization catalysts contaminating the final active pharmaceutical ingredient (API). The byproducts of the cyclization, primarily triphenylphosphine oxide and amine salts, are generally easier to remove via standard aqueous workups and crystallization techniques compared to heavy metal residues. This inherent cleanliness simplifies the downstream purification process, reducing the number of unit operations required to achieve high-purity pharmaceutical intermediates. Consequently, the final product exhibits a superior quality profile that aligns perfectly with the stringent regulatory requirements of global health authorities, thereby reducing the risk of batch rejection and ensuring consistent supply chain reliability.
How to Synthesize 2-Aminobenzimidazole Derivatives Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing these valuable heterocycles with high efficiency and reproducibility. The process is divided into three logical stages: urea formation, cyclization, and deprotection. Each stage is optimized to maximize yield while minimizing waste and operational hazards. The initial urea formation is conducted at room temperature in polar aprotic solvents like dimethyl sulfoxide, ensuring complete conversion of the starting diamines. The subsequent cyclization step utilizes the aforementioned phosphorus chemistry under controlled low-temperature conditions to prevent exothermic runaways, followed by a straightforward deprotection using alkaline hydrolysis. This modular approach allows for easy adaptation to various substrates, as demonstrated by the wide range of R groups successfully tested in the patent examples, including alkyl, aryl, and halogenated substituents.
- React a 2-(N-protected)-o-phenylenediamine compound with an N-phenoxy carbonyl mono-substituted amine to form a 2-(N-protected)-aminophenyl urea intermediate.
- Perform dehydration cyclization on the urea intermediate using dichlorotriphenylphosphorus or dibromotriphenylphosphorus in the presence of an organic base to form the protected benzimidazole ring.
- Execute a deprotection reaction under alkaline conditions to remove the protecting group, yielding the final 2-(N-substituted)-aminobenzimidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis methodology translates into tangible strategic benefits that extend beyond mere technical feasibility. The elimination of toxic heavy metals and the use of commodity chemicals like triphenylphosphine oxide and oxalyl chloride significantly de-risk the supply chain. Sourcing hazardous reagents like red mercury is becoming increasingly difficult and expensive due to tightening global environmental regulations, whereas the reagents used in this novel process are standard industrial chemicals with stable pricing and abundant availability. This shift ensures enhanced supply chain reliability and reduces the lead time for high-purity pharmaceutical intermediates by avoiding bottlenecks associated with special chemical permits and hazardous waste logistics.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the simplification of the purification workflow. By avoiding the use of heavy metal catalysts, manufacturers save substantially on the costs associated with metal scavenging resins, specialized filtration equipment, and hazardous waste disposal fees. Furthermore, the high yields reported in the patent examples, often ranging from 80% to 90%, mean that less raw material is required to produce the same amount of finished product, directly lowering the cost of goods sold (COGS). The mild reaction conditions also reduce energy consumption compared to the high-temperature methods previously employed, contributing to overall operational expenditure savings without compromising on output quality.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous starting materials ensures a more resilient supply chain. Traditional methods often face disruptions due to the restricted transport and storage of toxic desulfurization agents. In contrast, the reagents for this phosphorus-mediated cyclization are widely produced and easily stockpiled, mitigating the risk of production stoppages due to raw material shortages. Additionally, the robustness of the reaction conditions allows for greater flexibility in manufacturing scheduling, as the process is less sensitive to minor fluctuations in temperature or reagent quality, ensuring consistent delivery performance to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden complexities, particularly regarding heat management and waste treatment. This novel method is inherently scalable because the exothermic nature of the cyclization can be easily managed within the specified temperature range of -30°C to 50°C using standard industrial cooling systems. Moreover, the environmental footprint is drastically reduced by eliminating sulfur and heavy metal waste streams. This alignment with green chemistry principles not only simplifies environmental compliance reporting but also enhances the corporate sustainability profile of the manufacturer, which is increasingly a key criterion for selection by major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the phosphorus-mediated dehydration approach over legacy thiourea methods, focusing on yield, purity, and operational safety.
Q: What are the primary advantages of this synthesis method over conventional thiourea desulfurization?
A: Unlike conventional methods that require toxic heavy metals like red mercury or copper oxide for desulfurization, this novel approach utilizes phosphorus-based dehydration agents. This eliminates the need for complex heavy metal removal processes, significantly reducing environmental pollution and operational complexity while maintaining high yields.
Q: How does the use of in-situ generated phosphorus reagents impact production costs?
A: The method allows for the in-situ generation of dichlorotriphenylphosphorus or dibromotriphenylphosphorus from readily available precursors like triphenylphosphine oxide and oxalyl chloride or bromine. This avoids the procurement and storage hazards of pre-formed hazardous reagents, streamlining the supply chain and lowering raw material handling costs.
Q: Is this process suitable for large-scale industrial production of API intermediates?
A: Yes, the process operates under mild conditions, typically between -30°C and 50°C, and uses common organic solvents like acetonitrile or toluene. The simplicity of the unit operations, combined with high reported yields in the patent examples, makes it highly scalable for commercial manufacturing of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminobenzimidazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of 2-aminobenzimidazole derivative meets the exacting standards required by the global pharmaceutical industry. Our commitment to technological excellence means we can leverage advanced synthesis methods like the one described in CN102060780A to deliver superior products that outperform market alternatives in both quality and consistency.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will optimize your supply chain and accelerate your time to market for next-generation therapeutic agents.
