Revolutionizing Alpha-Arylation: Advanced Phosphonium Salt Ligands for Commercial Scale Pharmaceutical Intermediates
Revolutionizing Alpha-Arylation: Advanced Phosphonium Salt Ligands for Commercial Scale Pharmaceutical Intermediates
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the relentless demand for more efficient, cost-effective, and environmentally sustainable manufacturing processes. A pivotal advancement in this domain is documented in patent CN101717408B, which discloses a novel class of phosphonium salt compounds containing a bis-phosphorus ylide cyclopentadienyl carbene precursor. This innovation addresses critical bottlenecks in the synthesis of alpha-aryl derivatives of carbonyl compounds, which are ubiquitous structural motifs in active pharmaceutical ingredients (APIs) and agrochemicals. By replacing traditional nitrogen-based ligands with this unique phosphorus-containing architecture, the technology unlocks unprecedented catalytic activity, enabling the direct coupling of carbonyl compounds with electrophilic aryl reagents in a single step. For global procurement and R&D teams, this represents a strategic opportunity to optimize supply chains for high-value fine chemicals.
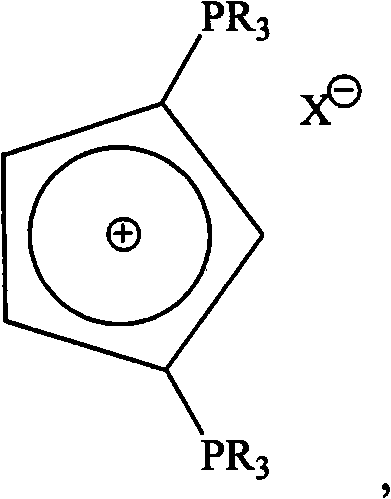
The core of this technological breakthrough lies in the specific molecular design of the ligand, which features a cyclopentadienyl ring substituted with two phosphine groups that form a stable phosphonium salt structure. Unlike conventional ligands that often suffer from instability or limited electronic tunability, this bis-phosphorus ylide system offers exceptional electron-donating capabilities. The patent details a robust preparation method involving the reaction of 1,1'-ferrocene dimercuty compounds with tertiary phosphines and palladium chloride under nitrogen protection. This synthesis pathway is not only chemically elegant but also practically viable for industrial replication, utilizing standard organic solvents like DMF and straightforward purification techniques such as recrystallization. The resulting ligands exhibit remarkable versatility, accommodating a wide range of counterions including halides and complex anions like PF6- and BF4-, thereby allowing chemists to fine-tune solubility and reactivity profiles for specific manufacturing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-aryl carbonyl derivatives has been plagued by significant inefficiencies that hinder large-scale production. Traditional methodologies often necessitate multi-step reaction sequences, each requiring distinct reagents and purification stages, which cumulatively drive up operational costs and extend lead times. Furthermore, classical approaches frequently rely on expensive and highly reactive aryl iodides or bromides as electrophilic partners, excluding the use of more abundant and economical aryl chlorides. The reliance on traditional N-heterocyclic carbene (NHC) ligands or bulky alkylphosphines often demands high catalyst loadings to achieve acceptable conversion rates, leading to substantial metal contamination in the final product. This contamination necessitates rigorous and costly downstream purification processes to meet the stringent purity specifications required by regulatory bodies for pharmaceutical applications. Additionally, many conventional catalytic systems exhibit poor functional group tolerance, limiting their applicability to complex molecular scaffolds common in modern drug discovery.
The Novel Approach
In stark contrast, the novel approach utilizing the bis-phosphorus ylide phosphonium salt ligands offers a paradigm shift towards atom economy and process intensification. By leveraging the superior electron-donating induction of the phosphorus ylide moiety, which surpasses that of traditional amino groups, the catalytic system achieves activation of substrates under significantly milder conditions. This allows for the direct use of less reactive and lower-cost aryl chlorides and aromatic sulfonic acid esters, dramatically reducing raw material expenditures. The one-step catalytic synthesis eliminates the need for tedious multi-step protections and deprotections, streamlining the workflow from kilograms to tons. Moreover, the high catalytic activity permits the use of reduced metal salt loadings, minimizing heavy metal residues and simplifying the workup procedure. This efficiency translates directly into enhanced throughput and reduced waste generation, aligning perfectly with the principles of green chemistry and sustainable manufacturing.
Mechanistic Insights into Phosphonium Salt-Catalyzed Alpha-Arylation
The exceptional performance of this catalytic system can be attributed to the unique electronic and steric environment created by the bis-phosphorus ylide cyclopentadienyl framework. Mechanistically, the phosphonium salt acts as a precursor to a highly reactive carbene species upon interaction with the metal center and base. The phosphorus ylide groups exert a powerful inductive effect, enriching the electron density at the carbene carbon more effectively than nitrogen analogues. This increased electron density facilitates the oxidative addition of the aryl halide to the metal center, which is typically the rate-determining step in cross-coupling reactions. Consequently, the energy barrier for this critical transformation is lowered, enabling the reaction to proceed rapidly even with sterically hindered or electronically deactivated substrates. The stability of the ferrocene backbone further ensures that the ligand remains intact throughout the catalytic cycle, preventing premature decomposition that often plagues other carbene precursors.
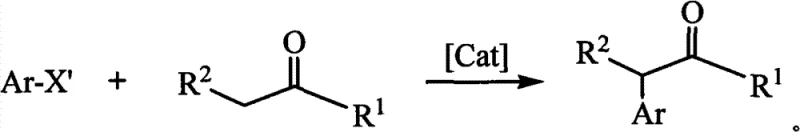
From an impurity control perspective, the mechanism offers distinct advantages for producing high-purity pharmaceutical intermediates. The high selectivity of the catalyst minimizes side reactions such as homocoupling of the aryl halide or over-arylation of the carbonyl substrate. The use of mild bases like potassium carbonate or sodium tert-butoxide, as opposed to harsh organolithium reagents, reduces the formation of degradation byproducts. Furthermore, the compatibility of the ligand with various metal salts, including palladium, copper, nickel, and iron, provides flexibility in choosing the most cost-effective and least toxic metal for a specific application. For instance, the ability to utilize iron or copper catalysts instead of precious palladium in certain contexts could offer substantial cost benefits. The robust nature of the catalytic cycle ensures consistent performance across different batches, a critical factor for maintaining quality control in commercial manufacturing environments where reproducibility is paramount.
How to Synthesize Alpha-Aryl Carbonyl Derivatives Efficiently
The implementation of this technology into a production setting involves a straightforward protocol that balances reaction efficiency with operational safety. The process begins with the precise weighing of the phosphonium salt ligand and the chosen metal salt catalyst, which are then introduced into a reaction vessel along with the carbonyl substrate and the aryl electrophile. A suitable base is added to activate the catalyst system, and the mixture is suspended in a solvent such as tetrahydrofuran, dioxane, or toluene. The reaction is conducted under an inert nitrogen atmosphere to prevent oxidation of sensitive intermediates, with heating applied to maintain reflux temperatures optimized for the specific substrate pair.
- Prepare the reaction mixture by combining the phosphonium salt ligand, a metal salt catalyst (such as palladium or copper), a base, the carbonyl substrate, and the aryl halide electrophile in a suitable solvent.
- Maintain the reaction under an inert nitrogen atmosphere and heat the mixture to reflux temperatures ranging between 20°C and 130°C depending on substrate reactivity.
- Upon completion, cool the mixture, extract the product with organic solvents like dichloromethane, and purify via chromatography to isolate the high-purity alpha-aryl derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phosphonium salt technology presents a compelling value proposition centered on cost optimization and supply security. The ability to utilize cheaper aryl chlorides instead of premium aryl iodides directly impacts the bill of materials, offering significant cost reduction in API manufacturing without compromising yield. The streamlined one-step process reduces the number of unit operations required, lowering labor costs and energy consumption associated with heating, cooling, and intermediate isolations. Furthermore, the high isolated yields reported in the patent examples, often exceeding ninety percent, mean that less raw material is wasted, improving the overall mass balance of the production line. This efficiency allows manufacturers to produce larger quantities of intermediates within the same timeframe, effectively increasing plant capacity without capital expenditure on new equipment.
- Cost Reduction in Manufacturing: The elimination of expensive N-heterocyclic carbene ligands and the reduction in precious metal catalyst loading contribute to a leaner cost structure. By enabling the use of base metals like iron or copper in certain variations, the dependency on volatile precious metal markets is reduced. The simplified purification process due to fewer byproducts also lowers the cost of solvents and chromatography media. These cumulative savings enhance the profit margin for high-volume pharmaceutical intermediates, making the final drug product more competitive in the global market.
- Enhanced Supply Chain Reliability: The synthetic route for the ligand itself relies on readily available starting materials such as ferrocene derivatives and common tertiary phosphines, ensuring a stable supply of the catalyst. The robustness of the catalytic reaction against moisture and air, relative to other sensitive organometallic systems, reduces the risk of batch failures due to environmental factors. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream customers. The versatility of the ligand across different metal salts also provides a backup strategy; if one metal becomes scarce, the process can potentially be adapted to another without redesigning the entire synthetic route.
- Scalability and Environmental Compliance: The reaction conditions described are amenable to scale-up from laboratory glassware to industrial reactors, with temperature and pressure parameters well within standard operating limits. The use of common organic solvents facilitates recycling and recovery, aligning with increasingly strict environmental regulations regarding volatile organic compound emissions. The reduction in heavy metal waste simplifies effluent treatment processes, lowering the environmental footprint of the manufacturing site. This compliance not only avoids regulatory fines but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this novel catalytic system. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of its practical utility.
Q: What represents the primary advantage of this phosphonium salt over traditional N-heterocyclic carbene ligands?
A: The phosphorus ylide moiety provides stronger electron-donating capacity compared to the amino groups in traditional NHC ligands, resulting in superior catalytic activity and allowing for milder reaction conditions.
Q: Can this catalytic system utilize less reactive aryl chlorides?
A: Yes, the enhanced electronic properties of the ligand enable the activation of less expensive and less reactive aryl chlorides and aromatic sulfonic acid esters, significantly reducing raw material costs.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: The methodology employs robust reaction conditions with readily available bases and solvents, demonstrating high isolated yields up to 96% in laboratory settings, which indicates strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphonium Salt Ligand Supplier
As the pharmaceutical industry continues to demand more sophisticated and efficient synthetic solutions, NINGBO INNO PHARMCHEM stands ready to support your transition to this advanced technology. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in your actual supply chain. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications for both the ligand and the resulting intermediates. We understand that consistency is key in API manufacturing, and our dedicated process development team works closely with clients to optimize reaction parameters for their specific molecular targets.
We invite you to engage with our technical procurement team to discuss how this phosphonium salt technology can be integrated into your current portfolio. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this more efficient catalytic system. We encourage you to contact us today to obtain specific COA data for our catalog of ligands and to schedule a consultation for route feasibility assessments tailored to your project needs. Together, we can drive innovation and efficiency in the synthesis of next-generation pharmaceutical intermediates.
