Advanced One-Pot Synthesis of Pentasubstituted Nicotinamide Derivatives for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly those based on the privileged nicotinamide structure. Patent CN110483387B discloses a groundbreaking one-pot method for synthesizing 2,N,4,5,6-pentasubstituted-N'-sulfonyl nicotinimide amide derivatives, addressing critical bottlenecks in current manufacturing protocols. This technology leverages a sophisticated copper-catalyzed multicomponent reaction that integrates cycloaddition, nucleophilic addition, condensation, and oxidation steps into a single operational sequence. By utilizing an O-acetyl arylethanone oxime derivative as a key building block, the process eliminates the need for isolating unstable intermediates, thereby streamlining the production workflow for high-value pharmaceutical intermediates. The strategic design of this reaction not only enhances atom economy but also significantly reduces the environmental footprint associated with traditional multi-step syntheses.
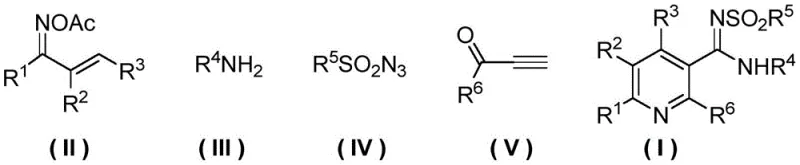
Nicotinamide derivatives are pivotal in diverse applications ranging from cosmetic anti-aging formulations to essential coenzymes in biological systems. However, the existing synthetic landscape has been plagued by inefficiencies that hinder rapid scale-up and cost optimization. The conventional reliance on multi-step sequences introduces cumulative yield losses and complicates purification processes, which is unacceptable for modern commercial scale-up of complex pharmaceutical intermediates. This patent presents a robust solution that transforms the synthesis paradigm, offering a reliable route to structurally diverse nicotinamide analogs with high purity profiles essential for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyridine core found in nicotinamide derivatives has relied heavily on classical methodologies such as the Hantzsch pyridine synthesis or routes involving malononitrile derivatives. The Hantzsch approach typically necessitates the initial formation of a dihydropyridine intermediate, which must subsequently undergo an oxidation step to aromatize the ring system. This multi-step requirement inherently increases operational complexity, as each isolation and purification stage introduces potential yield erosion and material loss. Furthermore, alternative methods utilizing malononitrile often involve harsh hydrolysis conditions that are not only environmentally unfriendly but also exhibit poor functional group tolerance, limiting the structural diversity of the final products. These legacy techniques struggle to meet the rigorous demands of modern API intermediate manufacturing where efficiency and sustainability are paramount.
The Novel Approach
In stark contrast, the methodology described in CN110483387B employs a convergent one-pot strategy that merges four distinct components: an O-acetyl arylethanone oxime derivative, an arylamine, a sulfonyl azide, and an alpha-carbonyl terminal alkyne. This innovative approach capitalizes on the dual functionality of the oxime ester, which serves simultaneously as a nitrogen source and an internal oxidant to drive the aromatization of the dihydropyridine intermediate in situ. By orchestrating these transformations within a single reaction vessel under copper catalysis, the process bypasses the need for external oxidants and separate oxidation steps. This consolidation of reaction steps results in a drastic simplification of the workflow, enabling the rapid generation of complex pentasubstituted nicotinamide scaffolds with exceptional efficiency and reduced waste generation.
Mechanistic Insights into Copper-Catalyzed Multicomponent Cyclization
The success of this transformation hinges on the precise selection of the catalytic system, specifically the synergy between a monovalent copper source and a specialized ligand. Experimental data indicates that monovalent copper compounds, particularly Cuprous Iodide (CuI), exhibit superior catalytic activity compared to their divalent counterparts. The reaction mechanism likely initiates with the activation of the terminal alkyne by the copper catalyst, facilitating a cycloaddition with the oxime derivative to form a metallacycle intermediate. Subsequent nucleophilic attack by the arylamine and insertion of the sulfonyl azide moiety drive the formation of the pyridine ring. The unique role of the O-acetyl group is critical, as it participates in the final oxidative aromatization step, effectively converting the transient dihydropyridine species into the stable aromatic nicotinamide product without requiring additional reagents.
From an impurity control perspective, this mechanism offers significant advantages over traditional routes. The use of the oxime ester as an internal oxidant minimizes the formation of over-oxidation byproducts that are common when using strong external oxidizing agents. Furthermore, the high selectivity of the CuI/TBTA catalyst system ensures that side reactions such as homocoupling of the alkyne or polymerization of the reactive intermediates are suppressed. This inherent selectivity translates directly to higher crude purity, reducing the burden on downstream purification processes like column chromatography or recrystallization. For procurement managers, this means a more predictable supply of high-quality material with consistent specifications, mitigating the risks associated with batch-to-batch variability in complex organic synthesis.
How to Synthesize Pentasubstituted Nicotinamide Derivatives Efficiently
Implementing this synthesis requires careful attention to the preparation of the key O-acetyl arylethanone oxime precursor, followed by the optimized one-pot coupling conditions. The precursor is typically generated by reacting a chalcone derivative with hydroxylamine hydrochloride in the presence of a base like pyridine, followed by acetylation with acetic anhydride. Once prepared, this intermediate enters the main catalytic cycle alongside the amine, azide, and alkyne components.
- Prepare the O-acetyl arylethanone oxime derivative (Formula II) by reacting a chalcone derivative with hydroxylamine hydrochloride followed by acetylation with acetic anhydride.
- In an organic solvent such as acetonitrile, combine the oxime derivative, arylamine, sulfonyl azide, and alpha-carbonyl terminal alkyne with a copper catalyst like CuI and a ligand like TBTA.
- Heat the reaction mixture to 60-120°C for 1-24 hours, then cool, remove solvent, and purify the crude product via silica gel column chromatography to obtain the target nicotinamide derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this one-pot copper-catalyzed technology represents a strategic opportunity to optimize manufacturing costs and enhance supply reliability. The elimination of multiple isolation steps and the use of readily available starting materials fundamentally alter the cost structure of producing these valuable nicotinamide derivatives. By reducing the number of unit operations, manufacturers can achieve substantial reductions in labor, energy consumption, and solvent usage, all of which contribute to a leaner and more competitive production model.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this process eliminates the need for intermediate isolation and purification, which are traditionally the most cost-intensive phases of chemical manufacturing. By avoiding the purchase and handling of external oxidants and reducing solvent volumes through process intensification, the overall cost of goods sold is significantly lowered. Additionally, the high catalytic efficiency of the CuI/TBTA system allows for lower catalyst loading while maintaining high yields, further driving down raw material expenses without compromising product quality.
- Enhanced Supply Chain Reliability: A major bottleneck in pharmaceutical supply chains is the dependency on exotic or difficult-to-source reagents. This method utilizes commodity chemicals such as arylamines, sulfonyl azides, and terminal alkynes, which are widely available from global chemical suppliers. The robustness of the reaction conditions, which tolerate a wide range of functional groups, ensures that supply disruptions due to specific raw material shortages are minimized. This flexibility allows for agile sourcing strategies and ensures continuous production capability even in volatile market conditions.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, particularly when dealing with hazardous oxidants or exothermic multi-step sequences. This one-pot method operates under relatively mild thermal conditions (60-120°C) and avoids the accumulation of unstable intermediates, making it inherently safer for large-scale production. The reduction in waste generation and solvent consumption aligns perfectly with green chemistry principles, facilitating easier regulatory compliance and reducing the costs associated with waste disposal and environmental remediation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this technology into their existing pipelines and for procurement officers assessing the long-term viability of the supply source.
Q: What are the advantages of this one-pot synthesis over traditional Hantzsch pyridine synthesis?
A: Traditional Hantzsch synthesis often requires multi-step procedures involving dihydropyridine intermediates and subsequent oxidation, leading to lower yields and operational difficulties. This patented one-pot method utilizes a copper-catalyzed cascade reaction that combines cycloaddition, nucleophilic addition, condensation, and oxidation in a single vessel, significantly simplifying the process and improving overall yield and purity.
Q: Which copper catalyst and ligand system provides the optimal performance?
A: According to the experimental data, monovalent copper compounds generally outperform divalent ones. Specifically, Cuprous Iodide (CuI) demonstrates the highest catalytic efficiency. When paired with the ligand tris[(1-benzyl-1H-1,2,3-triazol-4-yl)methyl]amine (TBTA) in acetonitrile solvent, the system achieves superior yields compared to other ligands like triethylamine or phenanthroline.
Q: Are the raw materials for this synthesis readily available for industrial scaling?
A: Yes, the key starting materials including arylamines, sulfonyl azides, and alpha-carbonyl terminal alkynes are commercially available. The O-acetyl arylethanone oxime derivative can be easily synthesized from common chalcones and hydroxylamine hydrochloride, ensuring a stable and cost-effective supply chain for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nicotinamide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN110483387B for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of nicotinamide derivative delivered meets the highest standards required for drug substance manufacturing.
We invite you to collaborate with our technical team to explore how this efficient one-pot synthesis can be tailored to your specific project needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient route. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your next-generation therapeutic candidates.
