Revolutionizing Nicotinamide Derivative Production with Efficient One-Pot Copper Catalysis for Commercial Scale
Introduction to Advanced Nicotinamide Synthesis
The landscape of organic chemical synthesis is continually evolving to meet the rigorous demands of the pharmaceutical and cosmetic industries, particularly for bioactive molecules like nicotinamide derivatives. As detailed in the recent patent CN110483387B, a groundbreaking methodology has been established for the one-pot synthesis of 2,N,4,5,6-pentasubstituted-N'-sulfonyl nicotinimide amide derivatives. This innovation addresses critical bottlenecks in producing Vitamin B3 derivatives, which are renowned not only for their role as coenzymes in redox reactions but also for their potent anti-aging properties in dermatological applications. The disclosed technology leverages a sophisticated copper-catalyzed multicomponent reaction that seamlessly integrates cycloaddition, nucleophilic addition, condensation, and oxidation steps. By streamlining what was previously a fragmented synthetic pathway, this approach offers a robust platform for generating diverse libraries of high-purity intermediates essential for next-generation drug discovery and skincare formulations.
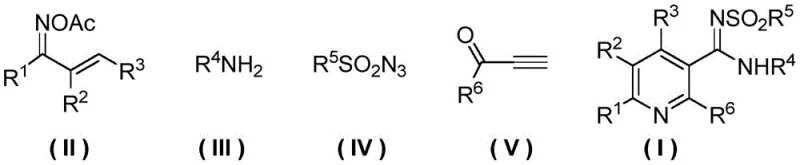
For R&D directors and procurement specialists, the significance of this patent lies in its ability to transform complex molecular architectures into accessible commercial realities. Traditional routes often struggle with functional group compatibility and environmental sustainability, but this new protocol utilizes mild conditions and widely available reagents to achieve exceptional results. The method specifically targets the synthesis of Formula (I) compounds through the convergence of four distinct building blocks: an O-acetyl arylethanone oxime derivative, an arylamine, a sulfonyl azide, and an alpha-carbonyl terminal alkyne. This convergence strategy not only enhances atom economy but also drastically reduces the operational complexity typically associated with constructing the pyridine core, thereby positioning this technology as a cornerstone for reliable pharmaceutical intermediate supplier networks aiming to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted pyridines and nicotinamide derivatives has relied heavily on classical methodologies such as the Hantzsch pyridine synthesis or modifications involving malononitrile derivatives. While these methods laid the foundational chemistry for the field, they are increasingly viewed as obsolete for modern high-throughput manufacturing due to inherent inefficiencies. The Hantzsch synthesis, for instance, typically requires the initial formation of a dihydropyridine intermediate followed by a separate, often harsh oxidation step to aromatize the ring. This multi-step sequence inevitably leads to cumulative yield losses and generates significant chemical waste, complicating the purification process and inflating production costs. Furthermore, existing literature highlights that methods utilizing ketoximes often suffer from low reactivity of beta-carbonyl amide derivatives, necessitating expensive and difficult-to-obtain starting materials that limit the structural diversity of the final products.
Beyond the chemical inefficiencies, conventional routes frequently exhibit poor functional group tolerance, restricting the types of substituents that can be introduced onto the pyridine ring without protecting group strategies. The reliance on strong oxidants or extreme thermal conditions in older protocols can degrade sensitive pharmacophores, rendering the final API intermediates unsuitable for delicate biological applications. Additionally, the environmental footprint of these legacy processes is substantial, involving multiple solvent exchanges and extensive workup procedures that conflict with modern green chemistry principles. For supply chain managers, these factors translate into longer lead times, higher raw material costs, and increased regulatory scrutiny regarding waste disposal, creating a compelling business case for adopting more streamlined and sustainable synthetic alternatives like the one described in the subject patent.
The Novel Approach
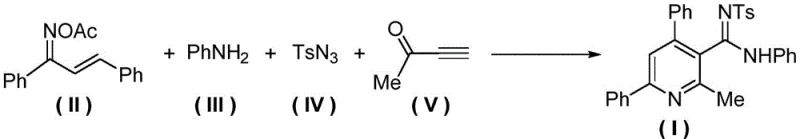
The methodology disclosed in patent CN110483387B represents a paradigm shift by employing a copper-catalyzed one-pot strategy that circumvents the pitfalls of traditional multi-step syntheses. By utilizing an O-acetyl arylethanone oxime derivative as a key synthon, the reaction simultaneously serves as both an amine source and an internal oxidant, effectively driving the aromatization of the dihydropyridine intermediate in situ. This elegant design eliminates the need for external oxidizing agents and separate reaction vessels, collapsing what was once a linear, time-consuming process into a single, efficient operation. The use of a copper compound catalyst, specifically cuprous iodide (CuI), in conjunction with a specialized ligand system facilitates the activation of the terminal alkyne and the subsequent cycloaddition with the oxime and azide components under remarkably mild conditions.
This novel approach not only simplifies the operational workflow but also dramatically expands the chemical space accessible to medicinal chemists. The protocol demonstrates excellent compatibility with a wide range of substituents, including halogens, alkoxy groups, and various alkyl chains, allowing for the rapid generation of diverse analog libraries for structure-activity relationship (SAR) studies. The reaction proceeds smoothly in common organic solvents like acetonitrile at temperatures ranging from 60°C to 120°C, avoiding the thermal degradation issues associated with higher temperature protocols. Consequently, this method delivers target nicotinamide derivatives with exceptional purity levels, often exceeding 98% as confirmed by HPLC analysis, thereby reducing the burden on downstream purification teams and ensuring a consistent supply of high-quality materials for clinical and commercial applications.
Mechanistic Insights into Copper-Catalyzed Multicomponent Cyclization
The success of this one-pot synthesis hinges on the intricate interplay between the copper catalyst, the ligand, and the reactive intermediates generated during the reaction cascade. Mechanistically, the process initiates with the coordination of the copper(I) species to the terminal alkyne of the alpha-carbonyl component, enhancing its nucleophilicity and facilitating the initial attack on the electrophilic centers of the oxime derivative. The presence of the tris[(1-benzyl-1H-1,2,3-triazol-4-yl)methyl]amine (TBTA) ligand is critical, as it stabilizes the copper center in its active +1 oxidation state, preventing disproportionation to inactive copper(II) species which would otherwise stall the catalytic cycle. This stabilization ensures a steady concentration of the active catalytic species throughout the reaction duration, which is essential for maintaining high turnover numbers and consistent product quality across different batches.
Following the initial activation, the reaction proceeds through a series of concerted steps involving nucleophilic addition and condensation that construct the pyridine ring skeleton. The sulfonyl azide component plays a dual role, acting as a nitrogen source for the amidine functionality while also participating in the oxidative aromatization process. The O-acetyl group on the oxime serves as a leaving group that drives the elimination necessary to form the aromatic system, effectively replacing the need for external oxidants. This internal redox balance is a key feature of the mechanism, minimizing side reactions and byproduct formation. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters such as stoichiometry and temperature to maximize yield and minimize impurities, ensuring that the final high-purity pharmaceutical intermediates meet the stringent specifications required for global regulatory compliance.
Impurity control is inherently built into this catalytic system due to the high selectivity of the copper-ligand complex. Unlike non-catalytic thermal cyclizations which often produce a complex mixture of regioisomers and polymeric byproducts, this method directs the reaction pathway towards the desired 2,N,4,5,6-pentasubstituted product with high fidelity. The mild reaction conditions prevent the decomposition of sensitive functional groups such as esters or halides, which might otherwise undergo hydrolysis or dehalogenation under harsher regimes. Furthermore, the use of acetonitrile as the preferred solvent aids in solubilizing the polar intermediates while remaining inert to the reaction conditions, further contributing to the clean reaction profile. This level of control is paramount for R&D teams focused on developing robust manufacturing processes that can withstand the rigors of scale-up without compromising on the purity or identity of the final active ingredient.
How to Synthesize 2,N,4,5,6-Pentasubstituted-N'-Sulfonyl Nicotinimide Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the preparation of the key O-acetyl arylethanone oxime precursor, as its quality directly influences the outcome of the one-pot reaction. The precursor is typically prepared by reacting a chalcone derivative with hydroxylamine hydrochloride in ethanol with a base like pyridine, followed by acetylation with acetic anhydride. Once the precursor is secured, the main transformation involves combining the oxime, arylamine, sulfonyl azide, and terminal alkyne in acetonitrile with CuI and TBTA. The detailed standardized synthesis steps for this process are outlined in the guide below, providing a clear roadmap for reproducibility.
- Prepare the O-acetyl arylethanone oxime derivative (Formula II) by reacting a chalcone derivative with hydroxylamine hydrochloride followed by acetylation with acetic anhydride.
- In an organic solvent such as acetonitrile, combine Formula II, arylamine (Formula III), sulfonyl azide (Formula IV), and alpha-carbonyl terminal alkyne (Formula V).
- Add a copper catalyst (preferably CuI) and a ligand (preferably TBTA), then heat the mixture to 60-120°C for 1-24 hours to complete the cycloaddition and oxidation sequence.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this one-pot copper-catalyzed methodology offers transformative benefits for procurement and supply chain operations within the fine chemical sector. The primary advantage lies in the significant simplification of the manufacturing process, which directly correlates to reduced operational expenditures and enhanced throughput. By consolidating multiple synthetic steps into a single reactor charge, manufacturers can eliminate the need for intermediate isolation, drying, and storage, thereby freeing up valuable tank farm capacity and reducing labor costs associated with material handling. This streamlined workflow also minimizes the exposure of reactive intermediates to the environment, lowering the risk of contamination and ensuring a more consistent product quality profile that aligns with Good Manufacturing Practice (GMP) standards.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the use of inexpensive and commercially abundant starting materials such as chalcones and simple arylamines, which are readily available from bulk chemical suppliers. The elimination of expensive external oxidants and the reduction in solvent consumption due to the one-pot nature of the reaction contribute to a substantially lower cost of goods sold (COGS). Furthermore, the high catalytic efficiency of the CuI/TBTA system means that catalyst loading can be kept low while still achieving excellent conversion, reducing the burden on metal scavenging processes during purification. These factors collectively enable a cost reduction in pharmaceutical intermediate manufacturing that allows companies to remain competitive in price-sensitive markets without sacrificing quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions, which tolerate a wide range of substrate variations without requiring bespoke optimization for each new analog. This flexibility allows manufacturers to respond rapidly to changing market demands or custom synthesis requests, significantly reducing lead time for high-purity pharmaceutical intermediates. The reliance on standard equipment and common solvents like acetonitrile ensures that production can be easily transferred between different facilities or scaled up without the need for specialized hardware, mitigating the risk of supply disruptions caused by equipment bottlenecks or unique resource constraints.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior to traditional methods, aligning with the increasing regulatory pressure for greener chemical processes. The high atom economy and reduced waste generation simplify wastewater treatment and solvent recovery operations, lowering the overall environmental footprint of the facility. The ability to run the reaction at moderate temperatures (60-120°C) also reduces energy consumption compared to high-temperature reflux protocols. These attributes facilitate the commercial scale-up of complex pharmaceutical intermediates while ensuring compliance with stringent environmental regulations, making it an attractive option for companies aiming to enhance their sustainability credentials and secure long-term contracts with eco-conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on catalyst selection, reaction scope, and process viability. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this technology into their existing production pipelines.
Q: What are the primary advantages of this one-pot synthesis over traditional Hantzsch methods?
A: Unlike traditional multi-step Hantzsch syntheses which suffer from harsh conditions and low yields, this copper-catalyzed one-pot method achieves high purity (over 98%) and significantly simplifies the workflow by combining cycloaddition, nucleophilic addition, condensation, and oxidation in a single vessel.
Q: Which catalyst and ligand system provides the optimal yield for these derivatives?
A: Experimental data indicates that Cuprous Iodide (CuI) serves as the most effective catalyst, while tris[(1-benzyl-1H-1,2,3-triazol-4-yl)methyl]amine (TBTA) acts as the superior ligand, consistently delivering yields above 90% under optimized conditions compared to other copper salts or amines.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process utilizes readily available starting materials like chalcones and operates at moderate temperatures (60-120°C) in common solvents like acetonitrile, making it highly amenable to commercial scale-up with straightforward post-processing via silica gel chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nicotinamide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in patent CN110483387B for driving innovation in the pharmaceutical and cosmetic sectors. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of nicotinamide derivatives meets the highest international standards for safety and efficacy. Our state-of-the-art facilities are equipped to handle the specific requirements of copper-catalyzed reactions, including precise temperature control and advanced purification capabilities.
We invite potential partners to engage with our technical procurement team to discuss how this cutting-edge synthesis can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to verify specific COA data for a target molecule, we are ready to provide comprehensive support. By leveraging our expertise in route feasibility assessments, we can help you optimize your development timeline and secure a reliable supply of high-quality intermediates. Contact us today to explore how our capabilities align with your goals for cost reduction in pharmaceutical intermediate manufacturing and accelerate your path to market.
