Advanced Cobalt-Catalyzed C-H Activation for Scalable 2-Alkoxyindole Manufacturing
Introduction: A Breakthrough in Indole Functionalization
The synthesis of 2-alkoxyindole compounds represents a critical challenge and opportunity in modern medicinal chemistry, particularly given their prevalence in high-value bioactive scaffolds. As detailed in the recent patent CN115772157A, filed in March 2023, a novel preparation method has been disclosed that fundamentally shifts the paradigm for accessing these privileged structures. This technology specifically targets the efficient construction of molecular frameworks found in potent selective 5-HT4 receptor antagonists, such as the well-known candidates GR-125487 and SB-207266.
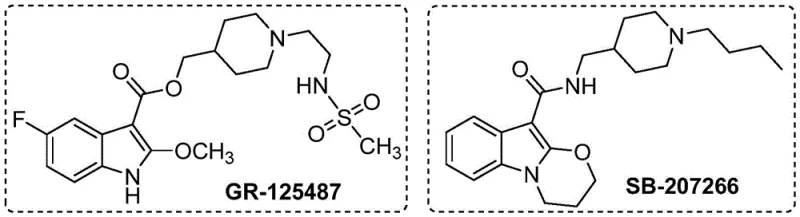
For R&D directors and process chemists, the significance of this patent lies in its departure from convoluted multi-step syntheses. Historically, accessing the 2-alkoxy substitution pattern on an indole ring required laborious protection-deprotection sequences or the use of scarce, expensive transition metals. The disclosed invention offers a direct, atom-economical route via C-H activation, utilizing a cobalt catalytic system that is not only cost-effective but also operationally simple. This development positions the technology as a cornerstone for cost reduction in pharmaceutical intermediate manufacturing, addressing the industry's perpetual demand for more sustainable and economical synthetic pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for synthesizing 2-alkoxyindoles have long been plagued by inefficiencies that hinder rapid drug discovery and commercial scale-up. Conventional routes often rely on noble metal catalysts such as palladium or rhodium, which introduce substantial cost burdens and supply chain vulnerabilities due to the geopolitical scarcity of these elements. Furthermore, many established protocols necessitate the use of pre-functionalized substrates, requiring additional synthetic steps to install leaving groups prior to the alkoxylation event. These extra steps not only increase the overall lead time but also generate significant amounts of chemical waste, complicating environmental compliance and increasing the burden on waste treatment facilities. Additionally, harsh reaction conditions involving strong bases or extreme temperatures are frequently required, which can limit the tolerance of sensitive functional groups often present in complex drug candidates.
The Novel Approach
In stark contrast, the method described in patent CN115772157A introduces a streamlined, cobalt-catalyzed C-H activation strategy that directly functionalizes the indole C2 position. By employing cobalt acetylacetonate (Co(acac)₂) as the catalyst and silver carbonate (Ag₂CO₃) as the oxidant, this process bypasses the need for pre-functionalization entirely. The reaction proceeds smoothly in the alcohol solvent itself, which acts dually as the reaction medium and the alkoxy source. This "solvent-as-reagent" approach drastically simplifies the reaction setup and workup. The mild thermal conditions, typically ranging from 90°C to 110°C, ensure high substrate compatibility, allowing for the preservation of delicate moieties that would otherwise degrade under traditional harsh conditions. This innovation represents a significant leap forward for any organization seeking a reliable pharmaceutical intermediate supplier capable of delivering complex heterocycles with greater efficiency.
Mechanistic Insights into Cobalt-Catalyzed C-H Alkoxylation
Understanding the mechanistic underpinnings of this transformation is crucial for process optimization and impurity control. The reaction initiates with the oxidation of the cobalt(II) catalyst by silver carbonate to generate a reactive cobalt(III) species. This high-valent cobalt center then coordinates with the indole substrate, likely facilitated by the directing group (such as the 8-quinolinyl amide moiety mentioned in the patent examples), positioning the metal in proximity to the C2-H bond. Subsequently, a single-electron transfer (SET) process occurs, leading to the formation of a radical cobalt(II) complex. This radical intermediate is pivotal, as it facilitates the cleavage of the relatively inert C-H bond at the 2-position of the indole ring.
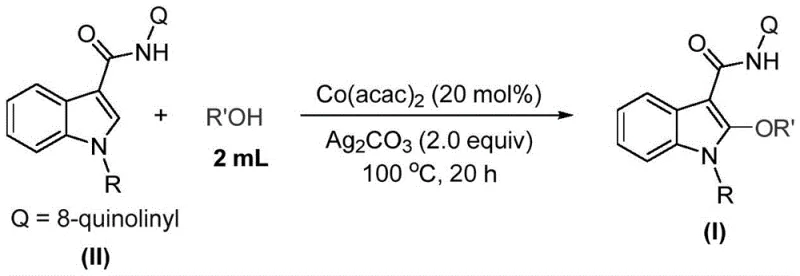
Following C-H activation, the resulting cobalt(III) intermediate undergoes coordination with the alcohol solvent. Through a sequence of migration insertion and reductive elimination steps, the alkoxy group is installed at the C2 position, regenerating the active cobalt species to continue the catalytic cycle. This mechanism explains the high regioselectivity observed in the patent data, where substitution occurs exclusively at the C2 position despite the presence of other reactive sites. For quality assurance teams, this defined mechanistic pathway suggests a predictable impurity profile, primarily limited to unreacted starting materials or over-oxidation byproducts, which can be effectively managed through standard purification techniques like silica gel chromatography. The robustness of this catalytic cycle ensures consistent batch-to-batch reproducibility, a key metric for high-purity pharmaceutical intermediates.
How to Synthesize 2-Alkoxyindole Efficiently
The practical execution of this synthesis is designed for accessibility, requiring standard laboratory equipment and commercially available reagents. The protocol involves charging a reaction vessel with the indole substrate, the cobalt catalyst, and the oxidant, followed by the addition of the desired alcohol. The mixture is then heated to facilitate the transformation. While the patent provides specific molar ratios and conditions, the general workflow is adaptable to various scales. For detailed operational parameters, safety data, and specific stoichiometric adjustments required for your specific substrate, please refer to the standardized synthesis guide below.
- Combine cobalt acetylacetonate (20 mol%), the indole substrate, and silver carbonate (2.0 equiv) in a reaction vessel.
- Add the corresponding alcohol (acting as both solvent and reagent, approx. 2 mL per 0.2 mmol substrate) to the mixture.
- Heat the reaction mixture to 100°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this cobalt-catalyzed methodology offers compelling advantages that extend beyond mere technical feasibility. The shift from noble metals to base metals like cobalt directly addresses the volatility of raw material costs, providing a more stable pricing structure for long-term supply agreements. Moreover, the simplification of the synthetic route reduces the number of unit operations required, which translates to lower energy consumption and reduced labor costs per kilogram of product. This efficiency is critical for maintaining competitiveness in the global market for commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or rhodium catalysts with inexpensive cobalt salts results in a drastic reduction in catalyst loading costs. Additionally, the use of the alcohol as both solvent and reagent eliminates the need for separate solvent recovery steps and reduces the volume of hazardous waste generated. This streamlined process logic inherently lowers the cost of goods sold (COGS), allowing for more aggressive pricing strategies without compromising margin integrity.
- Enhanced Supply Chain Reliability: Cobalt and silver carbonate are commodity chemicals with robust global supply chains, unlike specialized ligands or rare earth metals that may face supply disruptions. The starting indole compounds can be rapidly synthesized from readily available precursors like 8-aminoquinoline and picolinic acid derivatives. This reliance on bulk chemicals ensures that production schedules remain uninterrupted, significantly reducing lead time for high-purity intermediates and securing continuity of supply for downstream API manufacturing.
- Scalability and Environmental Compliance: The reaction conditions (100°C, ambient pressure) are easily transferable from laboratory glassware to industrial reactors without requiring specialized high-pressure equipment. The simplified workup, involving filtration and chromatography, minimizes the use of exotic extraction solvents. This aligns with green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing process, a key consideration for modern ESG-compliant supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within patent CN115772157A, ensuring that decision-makers have accurate information for evaluating this synthetic route.
Q: What are the key advantages of this cobalt-catalyzed method over traditional palladium methods?
A: This method utilizes earth-abundant cobalt catalysts instead of expensive noble metals like palladium or rhodium, significantly reducing raw material costs. Furthermore, it employs a direct C-H activation strategy that eliminates the need for pre-functionalized starting materials, streamlining the synthetic route and reducing waste generation.
Q: What is the substrate scope and functional group tolerance of this reaction?
A: The protocol demonstrates excellent compatibility with various N-substituents including alkyl, aryl, and benzyl groups. It also tolerates different alcohol chain lengths (methyl to pentyl) and functionalized alcohols like trifluoroethanol, making it highly versatile for generating diverse libraries of 2-alkoxyindole intermediates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's scalability. The use of simple Schlenk tube conditions, commercially available reagents, and straightforward workup procedures (filtration and chromatography) indicates strong potential for scale-up from gram to kilogram levels without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkoxyindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this cobalt-catalyzed C-H activation technology for the pharmaceutical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle transition metal catalysis with stringent purity specifications, ensuring that every batch of 2-alkoxyindole intermediate meets the rigorous quality standards required for clinical and commercial applications. Our rigorous QC labs employ advanced analytical techniques to verify structural integrity and impurity profiles, guaranteeing the reliability of our supply.
We invite you to collaborate with us to leverage this innovative synthesis for your drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget.
