Optimizing 4-Boron-L-Phenylalanine Production for BNCT Applications via Advanced Halogen-Magnesium Exchange
Introduction to Advanced Boronated Amino Acid Synthesis
The escalating demand for Boron Neutron Capture Therapy (BNCT) agents has necessitated a paradigm shift in the manufacturing of key pharmaceutical intermediates, specifically 4-boron-L-phenylalanine (L-BPA). As detailed in Chinese Patent CN116113636A, a novel synthetic methodology has been established that fundamentally alters the strategic placement of the boron moiety within the molecular architecture. Unlike conventional approaches that introduce boron at the inception of the synthetic route, this innovation employs a late-stage halogen-magnesium exchange strategy. This technical breakthrough addresses critical inefficiencies inherent in legacy processes, particularly regarding the conservation of expensive boron-10 isotopes and the mitigation of by-product formation. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding this mechanistic evolution is paramount for securing a sustainable supply chain for next-generation oncology therapeutics.
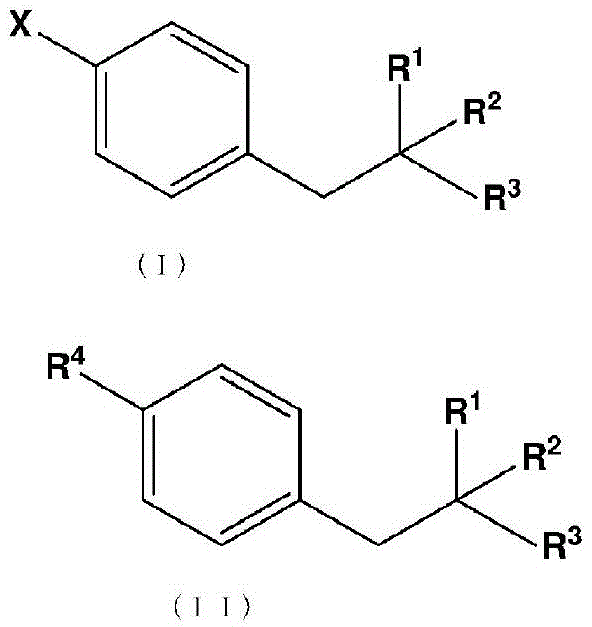
The core of this invention lies in the conversion of a halo-substituted phenylalanine derivative, represented by formula (I), into the corresponding boronic acid intermediate, formula (II). The process utilizes a highly reactive metal halide-alkylmagnesium halide complex, specifically leveraging the synergistic effects of lithium chloride and isopropylmagnesium chloride. This combination facilitates a rapid and clean exchange reaction at cryogenic temperatures, typically ranging from -78°C to 0°C, with optimal results observed between -50°C and -10°C. By deferring the introduction of the boron atom until the penultimate stage of the synthesis, manufacturers can drastically reduce the number of processing steps that the valuable boron moiety must endure, thereby preserving isotopic integrity and maximizing overall process yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 4-boron-L-phenylalanine has relied heavily on routes originating from boronated toluene derivatives. In these traditional pathways, the boron atom is incorporated at the very beginning of the synthetic sequence. While conceptually straightforward, this approach suffers from severe economic and technical drawbacks, especially when utilizing the boron-10 isotope required for effective BNCT. The multi-step nature of extending a simple boronated toluene into a complex amino acid structure inevitably leads to cumulative yield losses. More critically, every purification and transformation step post-borylation risks the loss of the expensive 10B label, rendering the final API cost-prohibitive. Furthermore, prior art methods often involve photo-bromination reactions which are plagued by solubility issues and the generation of difficult-to-remove impurities, complicating the purification landscape and reducing the overall throughput of the manufacturing facility.
The Novel Approach
The methodology disclosed in CN116113636A circumvents these bottlenecks by reversing the synthetic logic. Instead of building the amino acid backbone onto a boronated scaffold, the process constructs the protected amino acid framework first, terminating with a halogen substituent (such as iodine or bromine) at the para-position. This halo-intermediate is then subjected to the innovative exchange-borylation sequence. This strategic inversion ensures that the costly boron reagent is only introduced in the final chemical transformation. Consequently, the risk of isotopic dilution or loss is minimized to a single step. Additionally, the use of soluble organomagnesium intermediates avoids the heterogeneous reaction conditions typical of photo-bromination, leading to cleaner reaction profiles and simplified downstream processing. This represents a significant advancement in cost reduction in pharmaceutical intermediates manufacturing, aligning perfectly with the needs of modern, efficiency-driven production environments.
Mechanistic Insights into LiCl-Mediated Halogen-Magnesium Exchange
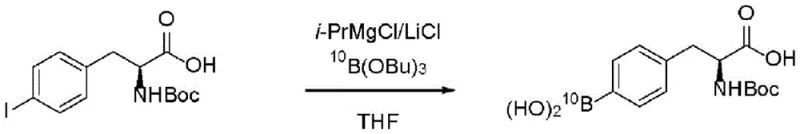
The efficacy of this novel route is underpinned by the specific catalytic environment created by the combination of alkylmagnesium halides and lithium salts. The reaction mechanism initiates with the formation of a "Turbo Grignard" species, where the presence of lithium chloride significantly enhances the reactivity and solubility of the organomagnesium complex. When the halo-substituted precursor is treated with i-PrMgCl/LiCl in anhydrous tetrahydrofuran (THF) or toluene, a rapid halogen-magnesium exchange occurs. This transmetallation is highly selective for the aryl halide bond, leaving sensitive functional groups such as protected amines (Boc, Acetyl) and esters intact, provided the temperature is strictly controlled within the -20°C to -30°C window. The resulting arylmagnesium species is a potent nucleophile, poised for immediate capture by the electrophilic boron source.
Following the exchange, the reaction mixture is quenched with a trialkyl borate, such as triethyl borate or tributyl borate. The nucleophilic attack of the arylmagnesium species on the boron center forms a stable borate complex. Subsequent acidic hydrolysis, typically using dilute hydrochloric acid to adjust the pH to approximately 1-3, liberates the free boronic acid functionality. A critical mechanistic advantage highlighted in the patent data is the role of LiCl in preventing precipitation. Comparative examples demonstrate that in the absence of lithium salts, the reaction mixture can solidify due to the formation of insoluble magnesium salts, rendering mechanical stirring impossible and halting the reaction. The inclusion of LiCl maintains a homogeneous solution phase throughout the exchange, ensuring efficient heat dissipation and mass transfer, which is indispensable for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 4-Boron-L-Phenylalanine Intermediates Efficiently
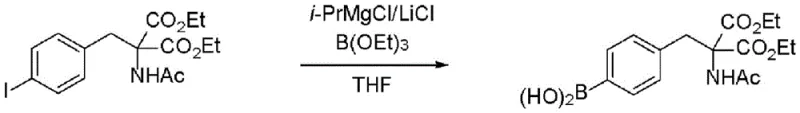
Implementing this synthesis requires precise control over stoichiometry and thermal conditions to maximize yield and isotopic retention. The process generally begins with the dissolution of the protected halo-amino acid precursor in a dry ether or hydrocarbon solvent. The Turbo Grignard reagent is added dropwise to maintain the exotherm within safe limits, followed by the addition of the borate ester. The reaction is allowed to proceed for several hours to ensure complete conversion before quenching. Detailed standard operating procedures regarding specific molar ratios, agitation speeds, and workup protocols are essential for reproducibility. For a comprehensive guide on executing this transformation with high fidelity, please refer to the standardized synthesis steps outlined below.
- Prepare a solution of the halo-substituted phenylalanine derivative in anhydrous THF or toluene under inert atmosphere.
- Add i-PrMgCl/LiCl complex at low temperature (-20°C to -30°C) to effect halogen-magnesium exchange without precipitation.
- Quench the resulting organomagnesium species with a borate ester (e.g., tributyl borate) and hydrolyze to obtain the boronic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this late-stage borylation protocol offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the substantial cost savings associated with raw material utilization, particularly when dealing with enriched boron-10 isotopes. By minimizing the number of unit operations that the boron moiety undergoes, the process inherently reduces the cumulative loss of this high-value input. This efficiency translates directly into a more favorable cost of goods sold (COGS) profile for the final active pharmaceutical ingredient. Furthermore, the elimination of problematic unit operations like photo-bromination removes a significant bottleneck from the production schedule, enhancing overall facility throughput and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The new methodology eliminates the need for specialized photochemical reactors and the associated safety infrastructure required for handling radical bromination processes. By shifting to a standard low-temperature organometallic reaction, facilities can utilize existing glass-lined or stainless-steel reactors without modification. Moreover, the suppression of 10B loss means that less expensive isotopic starting material is required per kilogram of final product, driving down the variable cost of production significantly without compromising on quality or yield.
- Enhanced Supply Chain Reliability: The starting materials for this route, such as protected halo-phenylalanines, are derived from commodity amino acids and are readily available from multiple global sources. This diversifies the supply base and reduces dependency on single-source vendors for specialized boronated scaffolds. The robustness of the reaction, evidenced by its tolerance to various protecting groups and solvents, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining continuous supply lines to downstream drug product manufacturers who cannot afford interruptions in their clinical or commercial production schedules.
- Scalability and Environmental Compliance: The homogeneous nature of the reaction mixture, ensured by the LiCl additive, makes this process highly scalable from laboratory benchtop to multi-ton commercial production. Unlike heterogeneous reactions that suffer from mixing limitations at large scales, this liquid-phase exchange allows for precise temperature control and uniform reaction kinetics. Additionally, the process avoids the generation of heavy metal waste associated with some transition-metal catalyzed borylations, simplifying waste stream management and aligning with increasingly stringent environmental regulations governing pharmaceutical manufacturing effluents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the technology's capabilities and limitations. Understanding these nuances helps stakeholders make informed decisions regarding technology transfer and process validation.
Q: Why is the late-stage borylation method superior for 10B-labeled compounds?
A: Traditional methods introduce boron at the beginning of the synthesis, leading to significant loss of expensive 10B isotopes during subsequent multi-step transformations. The patented late-stage exchange method minimizes these losses by introducing boron in the final steps.
Q: How does the LiCl additive improve the reaction scalability?
A: Comparative examples in the patent demonstrate that without LiCl, solid precipitation occurs during the exchange reaction, making stirring impossible. The LiCl additive maintains solubility, ensuring efficient mixing and heat transfer essential for commercial scale-up.
Q: What protecting groups are compatible with this borylation protocol?
A: The method is robust across various protecting groups, including Boc (tert-butoxycarbonyl), Acetyl, and Benzyl esters, allowing flexibility in downstream deprotection strategies for final API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Boron-L-Phenylalanine Supplier
As the global oncology landscape shifts towards targeted therapies like BNCT, the need for high-quality, isotopically pure intermediates has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the one described in CN116113636A to deliver superior value to our partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 4-boron-L-phenylalanine meets the exacting standards required for clinical applications.
We invite you to collaborate with us to optimize your supply chain for boronated amino acids. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and isotopic specifications. Contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a reliable, cost-effective, and scalable source for your critical BNCT intermediates, ensuring your projects move forward without delay.
