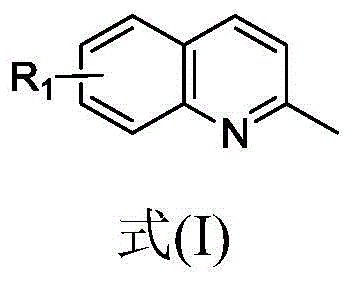
Revolutionizing Imidazoquinoline Production: Metal-Free Electrochemical Synthesis for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective methods for constructing complex heterocyclic scaffolds. Patent CN111910206A introduces a groundbreaking methodology for the synthesis of 3-cyano-substituted imidazo[1,5-a]quinoline compounds, a class of molecules with significant biological activity and utility as key pharmaceutical intermediates. This innovation leverages organic electrosynthesis to achieve a three-component coupling reaction that traditionally requires harsh conditions or expensive metal catalysts. By utilizing an undivided electrolytic cell, the process enables the direct construction of the imidazoquinoline core from 2-methylquinolines, substituted aliphatic amines, and a cyano source. This approach not only streamlines the synthetic pathway but also aligns with modern green chemistry principles by eliminating the need for external chemical oxidants and transition metals, thereby addressing critical purity concerns in drug manufacturing.
For R&D directors and process chemists, the implications of this technology are profound. The ability to generate high-value nitrogen-containing heterocycles through an electrocatalytic manifold offers a distinct advantage over conventional thermal methods. The reaction proceeds under mild conditions, typically between 0°C and 100°C, using common solvents like N,N-dimethylacetamide or acetonitrile. This flexibility allows for the accommodation of diverse functional groups without compromising yield or selectivity. Furthermore, the atom economy of this three-component reaction is exceptionally high, as it combines three distinct molecular fragments into a single complex structure in one pot. This efficiency reduces the overall step count, minimizes waste generation, and accelerates the timeline from laboratory discovery to pilot-scale production, making it an attractive option for the rapid development of new drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of cyano-functionalized imidazo[1,5-a]nitrogen heterocycles has relied heavily on transition metal catalysis. While effective, these conventional methods suffer from significant drawbacks that impact both the economic and environmental viability of large-scale manufacturing. The primary concern is the residual presence of heavy metals in the final product, which necessitates rigorous and often costly purification steps to meet stringent regulatory standards for pharmaceutical ingredients. Additionally, many traditional routes require stoichiometric amounts of chemical oxidants, which generate substantial quantities of hazardous waste and increase the overall E-factor of the process. The multi-step nature of some classical syntheses further exacerbates these issues, leading to lower overall yields and higher production costs due to material loss at each stage. These limitations create a bottleneck for supply chain managers who require consistent, high-purity materials with reliable lead times.
The Novel Approach
In stark contrast, the electrochemical method disclosed in CN111910206A offers a transformative solution by replacing chemical oxidants with electrons. This metal-free approach fundamentally changes the reaction landscape, allowing for the direct oxidative coupling of substrates at the electrode surface. The use of an undivided cell simplifies the reactor design, reducing capital expenditure and operational complexity. By avoiding transition metals entirely, the risk of product contamination is virtually eliminated, simplifying downstream processing and quality control. The reaction demonstrates excellent functional group tolerance, accommodating various substituents on the quinoline and amine components, which is crucial for generating diverse libraries of analogs during drug discovery. This novel pathway represents a paradigm shift towards sustainable manufacturing, offering a cleaner, safer, and potentially more economical route to these valuable intermediates.
![General reaction scheme for the electrochemical synthesis of 3-cyano-substituted imidazo[1,5-a]quinolines from 2-methylquinolines, amines, and cyano sources](/insights/img/imidazoquinoline-synthesis-electrochemical-pharma-supplier-20260305115805-06.webp)
Mechanistic Insights into Electrocatalytic Three-Component Coupling
The core of this innovation lies in the anodic oxidation mechanism that drives the formation of the imidazoquinoline ring system. In this electrocatalytic cycle, the 2-methylquinoline substrate undergoes oxidation at the anode to generate a reactive radical cation intermediate. This species is highly electrophilic and readily reacts with the nucleophilic amine component present in the solution. The subsequent steps involve a series of proton transfers and further oxidation events that facilitate the cyclization process. The cyano source, typically a silyl cyanide or similar reagent, acts as a nucleophile or radical trap to introduce the cyano group at the 3-position of the newly formed heterocycle. The electrolyte plays a critical role in maintaining conductivity and stabilizing charged intermediates, ensuring efficient electron transfer throughout the reaction mixture. Understanding this mechanism is vital for optimizing reaction parameters such as current density, electrode material, and electrolyte concentration to maximize yield and selectivity.
From an impurity control perspective, the electrochemical method offers superior precision compared to thermal radical reactions. The potential applied at the electrode can be finely tuned to selectively oxidize the specific substrate without affecting other sensitive functional groups, thereby minimizing side reactions and byproduct formation. This selectivity is particularly important when dealing with complex molecules containing multiple oxidizable sites. The absence of metal catalysts also removes a major source of variability and potential toxicity associated with metal-ligand complexes decomposing under reaction conditions. Consequently, the resulting crude product typically exhibits a cleaner profile, reducing the burden on purification teams. For procurement and supply chain stakeholders, this translates to more predictable manufacturing outcomes and reduced risk of batch failures due to impurity spikes, ensuring a steady supply of high-quality intermediates.
How to Synthesize 3-Cyano-substituted Imidazo[1,5-a]quinolines Efficiently
Implementing this electrochemical synthesis requires careful attention to reaction setup and parameter optimization to ensure reproducibility and safety. The process begins with the preparation of the electrolytic solution, where precise molar ratios of the 2-methylquinoline, amine, and cyano source are mixed with the appropriate electrolyte and solvent. The choice of electrode material, such as platinum or carbon, can influence the reaction rate and efficiency, so screening may be necessary for specific substrates. Once the cell is assembled and the current is applied, the reaction progress should be monitored using techniques like TLC or HPLC to determine the optimal endpoint. Following the reaction, standard workup procedures involving extraction and chromatography are employed to isolate the pure product. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this advanced methodology.
- Prepare the electrolytic mixture by adding electrolyte, 2-methylquinoline derivative, substituted aliphatic amine, cyano source, and solvent into an undivided cell.
- Install electrodes (Pt, C, Ni, or Cu) into the cell and apply constant current electricity while stirring at temperatures between 0°C and 100°C.
- Upon reaction completion, separate and purify the solution via column chromatography or recrystallization to isolate the target 3-cyano-substituted compound.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this electrochemical technology offers substantial strategic benefits for organizations focused on cost reduction and supply chain resilience in pharmaceutical intermediate manufacturing. The elimination of expensive transition metal catalysts directly lowers raw material costs and removes the need for specialized metal scavenging resins or complex filtration steps. This simplification of the downstream process significantly reduces processing time and solvent consumption, leading to a leaner and more cost-effective operation. Furthermore, the use of electricity as a traceless reagent enhances the sustainability profile of the manufacturing process, which is increasingly important for meeting corporate environmental goals and regulatory compliance. The robustness of the method across a wide range of substrates ensures that supply chains remain flexible and responsive to changing project requirements without the need for extensive process redevelopment.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts and stoichiometric chemical oxidants results in significant savings on raw material expenditures. Additionally, the simplified workup procedure reduces labor and utility costs associated with purification, contributing to a lower overall cost of goods sold. The high atom economy of the three-component reaction minimizes waste disposal fees, further enhancing the economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: By relying on readily available starting materials and standard electrical equipment, the risk of supply disruptions caused by specialized reagent shortages is mitigated. The robustness of the electrochemical protocol ensures consistent batch-to-batch quality, reducing the likelihood of production delays due to failed batches. This reliability is crucial for maintaining uninterrupted supply to downstream API manufacturers and meeting tight project deadlines.
- Scalability and Environmental Compliance: The use of undivided cells and common solvents facilitates easy scale-up from laboratory to pilot and commercial scales. The green nature of the process, characterized by reduced hazardous waste generation and energy efficiency, aligns well with strict environmental regulations. This compliance reduces the regulatory burden and potential liabilities associated with waste management, making the process more sustainable in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of imidazoquinolines. These answers are derived from the detailed experimental data and scope analysis provided in the patent literature. They are intended to clarify the practical aspects of implementing this technology in a production environment and to highlight its advantages over traditional methods. Understanding these details helps stakeholders make informed decisions about integrating this innovative pathway into their existing manufacturing portfolios.
Q: Does this electrochemical method eliminate heavy metal residues?
A: Yes, the process described in patent CN111910206A operates without transition metal catalysts or chemical oxidants, significantly reducing the risk of heavy metal contamination in the final pharmaceutical intermediate.
Q: What is the substrate scope for the amine component?
A: The method demonstrates high tolerance for various substituted aliphatic amines, including benzylamines with electron-donating or electron-withdrawing groups such as methyl, chloro, fluoro, and trifluoromethyl substituents.
Q: Is this process scalable for industrial production?
A: The use of undivided cells and commercially available electrode materials like platinum or carbon suggests strong potential for scale-up, offering a greener alternative to traditional multi-step metal-catalyzed routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyano-substituted Imidazo[1,5-a]quinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN111910206A can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We are committed to delivering high-quality intermediates that empower our clients to accelerate their drug development programs with confidence.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule and volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can optimize your supply chain, reduce costs, and bring life-saving medicines to market faster through the power of advanced organic electrosynthesis.
