Advanced Manufacturing of 2-Quaternary Carbon Indole-3-Ketones for Global Pharma Supply Chains
Introduction to Next-Generation Indole-3-One Synthesis
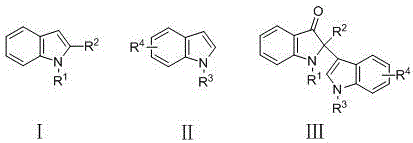
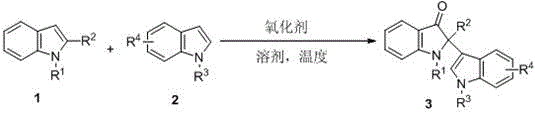
The construction of complex heterocyclic scaffolds containing quaternary carbon centers remains one of the most challenging frontiers in modern organic synthesis, particularly within the realm of pharmaceutical intermediate manufacturing. The indole-3-one core structure is a privileged substructure found extensively in natural alkaloids and biologically active molecules, serving as the critical backbone for numerous therapeutic agents including (-)-isatisine A and strobilanthoside A. Historically, accessing these motifs required convoluted multi-step sequences that often suffered from poor atom economy and harsh reaction conditions. However, the technological landscape has shifted dramatically with the disclosure of patent CN108218762B, which details a highly efficient and operationally simple method for synthesizing 2-position quaternary carbon indol-3-ketone compounds. This breakthrough methodology leverages peroxide oxidants to facilitate a direct intermolecular reaction between 2-substituted indoles and indole compounds, achieving yields as high as 96% under neutral conditions. For R&D directors and procurement specialists alike, this represents a pivotal advancement, offering a pathway to high-purity intermediates that aligns perfectly with the industry's growing demand for green chemistry and cost-effective production strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing 2,2-disubstituted indoline-3-ones have long been plagued by significant structural and economic inefficiencies that hinder large-scale adoption. Prior art typically relies on three main categories of transformation: cyclization of non-indole precursors, unimolecular or bimolecular reactions of indole derivatives, or modifications of existing 3-indolone substrates. These legacy methods frequently necessitate the use of expensive transition metal catalysts, stoichiometric amounts of hazardous reagents, or extreme pH conditions that compromise the integrity of sensitive functional groups. Furthermore, the substrate scope in conventional protocols is often narrowly defined, failing to accommodate diverse electronic environments or steric bulk without drastic drops in selectivity and yield. From a supply chain perspective, the reliance on specialized precursors creates bottlenecks, while the generation of complex waste streams increases the burden on environmental compliance teams. These factors collectively drive up the cost of goods sold (COGS) and extend lead times, making traditional approaches less viable for the rapid development cycles required in modern drug discovery and agrochemical intermediate manufacturing.
The Novel Approach
In stark contrast to these legacy limitations, the innovative strategy outlined in the patent data introduces a direct oxidative coupling mechanism that fundamentally simplifies the synthetic architecture. By utilizing inexpensive peroxide oxidants, such as 70% aqueous tert-butyl hydroperoxide (TBHP), the reaction proceeds efficiently in green solvents like water or water-organic mixtures at moderate temperatures ranging from 60°C to 140°C. This approach eliminates the need for precious metal catalysts and avoids the generation of heavy metal waste, thereby streamlining the downstream purification process significantly. The method demonstrates exceptional robustness, tolerating a wide array of substituents including halogens, alkyls, aryls, and electron-withdrawing groups like nitro and cyano moieties without compromising reaction efficiency. This versatility ensures that a single standardized protocol can be applied to generate a diverse library of analogues, accelerating the optimization phase for medicinal chemists. Moreover, the operational simplicity—merely mixing substrates with oxidant and solvent followed by heating—makes this technology uniquely suited for seamless technology transfer from laboratory benchtop to commercial reactor vessels.
Mechanistic Insights into Peroxide-Mediated Oxidative Coupling
The mechanistic underpinning of this transformation involves a sophisticated radical-mediated pathway that enables the formation of the sterically congested quaternary carbon center at the 2-position of the indole ring. Upon thermal activation, the peroxide oxidant undergoes homolytic cleavage to generate reactive radical species, likely tert-butoxyl radicals, which initiate the catalytic cycle by abstracting a hydrogen atom or adding to the electron-rich indole system. This activation step generates a transient radical intermediate at the C3 position of the indole nucleophile, which subsequently attacks the electrophilic C2 position of the 2-substituted indole partner. The resulting intermediate undergoes further oxidation and rearomatization to yield the final 2-position quaternary carbon indol-3-ketone product. Crucially, the reaction proceeds under neutral conditions, which prevents the acid- or base-catalyzed decomposition of the sensitive ketone functionality and minimizes the formation of polymeric byproducts. This controlled radical environment ensures high chemoselectivity, allowing the reaction to proceed cleanly even in the presence of potentially reactive functional groups on the aromatic rings.
From an impurity control perspective, the choice of oxidant and solvent plays a pivotal role in defining the purity profile of the final API intermediate. The use of aqueous TBHP in water or mixed solvents facilitates a homogeneous reaction environment that promotes consistent heat transfer and mass transport, reducing the likelihood of localized hot spots that could trigger side reactions. The quenching step using saturated sodium bicarbonate effectively neutralizes any residual acidic byproducts or unreacted peroxides, ensuring the stability of the product during isolation. Furthermore, the extraction protocol using ethyl acetate allows for the efficient separation of the organic product from inorganic salts and polar impurities, resulting in a crude material that requires minimal chromatographic purification. This inherent cleanliness of the reaction profile is a major advantage for regulatory filings, as it simplifies the characterization of the impurity spectrum and reduces the risk of genotoxic impurity carryover, thereby enhancing the overall safety and quality of the pharmaceutical supply chain.
How to Synthesize 2-Quaternary Carbon Indole-3-Ketones Efficiently
Implementing this synthetic route in a production setting requires careful attention to reaction parameters to maximize yield and safety while maintaining operational simplicity. The general procedure involves charging a reaction vessel with the oxidant and solvent, followed by the sequential addition of the 2-substituted indole substrate and the indole coupling partner. The molar ratio of the substrates is flexible, typically ranging from 1.0:1.0 to 1.0:10.0, allowing process chemists to optimize based on the relative cost and availability of the starting materials. The reaction mixture is then heated in an oil bath, with temperatures optimized between 60°C and 120°C, preferably around 100°C, for a duration of 1 to 48 hours depending on the specific electronic nature of the substrates. Upon completion, indicated by TLC analysis, the reaction is cooled to room temperature and quenched carefully to ensure safe handling of residual oxidants. The detailed standardized synthesis steps, including specific workup procedures and purification protocols validated for GMP environments, are provided in the technical guide below.
- Prepare the reaction mixture by adding the oxidant (preferably 70% TBHP aqueous solution) and solvent (water or mixtures with dioxane/acetonitrile) to a reaction vessel.
- Introduce the substrate 2-substituted indole and the indole compound into the reaction flask, ensuring a molar ratio between 1.0: 1.0 and 1.0:10.0.
- Heat the mixture in an oil bath at 60-140°C for 1-48 hours, then cool, quench with saturated sodium bicarbonate, and extract to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this peroxide-mediated oxidative coupling technology offers transformative benefits that directly impact the bottom line and operational resilience. The primary driver of value creation lies in the drastic simplification of the raw material portfolio; by replacing expensive transition metal catalysts and specialized precursors with commodity chemicals like TBHP and water, the direct material costs are substantially reduced. This shift not only lowers the entry barrier for sourcing but also mitigates the risk of supply disruptions associated with niche reagents. Furthermore, the use of water as a preferred solvent eliminates the need for large volumes of volatile organic compounds (VOCs), significantly reducing solvent recovery costs and environmental disposal fees. The simplified workup procedure, which relies on standard extraction and concentration techniques rather than complex distillation or crystallization steps, translates into reduced labor hours and faster batch cycle times. These efficiencies collectively contribute to a more agile manufacturing process capable of responding rapidly to fluctuating market demands.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes the necessity for expensive metal scavenging steps and rigorous residual metal testing, which are often costly and time-consuming bottlenecks in API manufacturing. Additionally, the high atom economy of the direct coupling reaction minimizes waste generation, leading to lower waste treatment costs and improved overall process mass intensity (PMI). The use of inexpensive oxidants and green solvents further drives down the variable cost per kilogram, enabling competitive pricing strategies for high-volume contracts without sacrificing margin.
- Enhanced Supply Chain Reliability: By utilizing widely available industrial chemicals such as tert-butyl hydroperoxide and common indole derivatives, the supply chain becomes far more robust against geopolitical or logistical shocks. The broad substrate scope means that alternative starting materials can be sourced easily if a specific vendor faces issues, ensuring continuity of supply for critical pharmaceutical intermediates. The mild reaction conditions also reduce the wear and tear on reactor equipment, extending asset life and minimizing unplanned maintenance downtime, which is crucial for maintaining consistent delivery schedules to global clients.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on simple thermal activation and liquid-phase reactions, avoiding the complexities of gas-liquid interfaces or high-pressure systems. This ease of scale-up facilitates a smooth transition from pilot plant to commercial production, reducing the time-to-market for new drug candidates. Moreover, the alignment with green chemistry principles, specifically the use of water and the avoidance of heavy metals, ensures compliance with increasingly stringent environmental regulations across major markets, future-proofing the manufacturing site against regulatory changes and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative coupling technology, derived directly from the patent specifications and practical manufacturing experience. These insights are designed to clarify the operational feasibility and strategic value of adopting this method for your specific project needs. Understanding these nuances is essential for making informed decisions about process integration and resource allocation.
Q: What are the primary advantages of this oxidative coupling method over traditional cyclization routes?
A: This method operates under neutral conditions using green solvents like water, avoiding the expensive reagents and harsh acidic or basic conditions often required by conventional cyclization strategies, thereby simplifying purification and reducing environmental impact.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process utilizes readily available industrial oxidants like TBHP and simple workup procedures involving extraction and concentration, which are highly amenable to scale-up from kilogram to multi-ton manufacturing without complex equipment requirements.
Q: What is the substrate scope regarding substituents on the indole ring?
A: The patent demonstrates excellent substrate universality, tolerating various groups such as halogens, alkyls, aryls, benzyloxy, nitro, and cyano groups at different positions, allowing for the synthesis of diverse derivatives for medicinal chemistry applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Quaternary Carbon Indole-3-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patented reaction; it demands a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are equipped with stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to guarantee that every batch of 2-quaternary carbon indole-3-ketone meets the highest international standards. Our commitment to quality assurance extends beyond mere compliance; we proactively manage impurity profiles and optimize crystallization processes to deliver materials that facilitate smoother downstream formulation for our clients.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate concretely how our optimized manufacturing processes can enhance your supply chain efficiency and reduce overall project costs. Let us be your trusted ally in navigating the complexities of fine chemical synthesis, delivering reliability and excellence at every step of the value chain.
