Advanced Synthetic Route for 2-Quaternary Carbon Indole-3-Ketones: Scalable Manufacturing and Process Optimization
Advanced Synthetic Route for 2-Quaternary Carbon Indole-3-Ketones: Scalable Manufacturing and Process Optimization
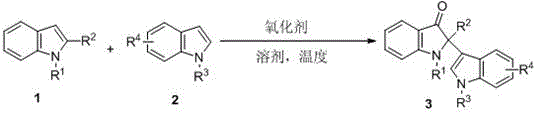
The development of efficient synthetic methodologies for complex heterocyclic scaffolds remains a cornerstone of modern pharmaceutical process research. Specifically, the construction of 2-position quaternary carbon indol-3-one compounds represents a significant challenge due to the steric hindrance associated with forming quaternary centers at the C2 position. A groundbreaking approach detailed in Chinese Patent CN108218762B introduces a highly efficient and operationally simple method for synthesizing these valuable structures. This technology utilizes peroxides as oxidants to facilitate an intermolecular reaction between 2-substituted indoles and indole compounds, achieving yields as high as 96% under neutral conditions. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate supplier partnerships, this patent offers a compelling alternative to traditional, resource-intensive pathways. The ability to utilize water as a primary solvent not only aligns with green chemistry principles but also drastically reduces the cost burden associated with organic solvent disposal and recovery. As we analyze the technical depth of this innovation, it becomes clear that this methodology provides a robust platform for the commercial scale-up of complex pharmaceutical intermediates, ensuring supply chain continuity for critical bioactive alkaloid precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-indolinone compounds possessing a quaternary carbon chiral center at the 2-position has been fraught with significant technical and economic hurdles. Conventional strategies typically fall into three categories: cyclization from non-indole precursors, unimolecular or bimolecular reactions of indole compounds, or derivatization of existing 3-indolinone substrates. However, these established methods frequently suffer from severe limitations that impede their industrial applicability. Many traditional routes require the use of expensive transition metal catalysts or stoichiometric amounts of hazardous reagents, which complicates the purification process and raises concerns regarding heavy metal residues in the final active pharmaceutical ingredient. Furthermore, the substrate scope in older methodologies is often narrowly defined, failing to accommodate diverse functional groups such as halogens or electron-withdrawing nitro groups without significant loss in yield or selectivity. The reliance on harsh acidic or basic conditions in some prior art also necessitates specialized corrosion-resistant equipment and generates substantial volumes of chemical waste, thereby increasing the overall cost reduction in API manufacturing challenges. These factors collectively create bottlenecks in the supply chain, making it difficult to secure high-purity intermediates in a timely manner.
The Novel Approach
In stark contrast to the limitations of legacy technologies, the novel approach disclosed in the patent data leverages a direct oxidative coupling strategy that is both atom-economical and environmentally benign. By employing peroxides such as tert-butyl hydroperoxide (TBHP) as the terminal oxidant, the reaction proceeds smoothly under neutral conditions, thereby preserving sensitive functional groups that might otherwise degrade. The core transformation involves the direct conversion of readily available indole derivatives into the target 2-quaternary carbon indol-3-ones without the need for pre-functionalization or protecting group manipulations. This streamlined process significantly shortens the synthetic sequence, reducing both material costs and processing time. The versatility of this method is further highlighted by its compatibility with a wide range of solvents, with water being the preferred medium, which underscores its potential for green manufacturing. The reaction conditions are mild yet effective, typically operating between 60-140°C, allowing for flexibility in process optimization depending on the specific thermal stability of the substrates involved. This robustness ensures that the method can be adapted for the commercial scale-up of complex polymer additives or pharmaceutical intermediates with minimal re-engineering of existing reactor infrastructure.
Mechanistic Insights into Peroxide-Mediated Oxidative Coupling
The mechanistic pathway of this transformation is believed to involve a radical-mediated oxidation process initiated by the decomposition of the peroxide oxidant. Upon heating, the peroxide generates reactive oxygen species that activate the C3 position of the indole nucleophile, facilitating an electrophilic attack on the C2 position of the 2-substituted indole substrate. This intermolecular coupling is critical for establishing the sterically congested quaternary carbon center. The use of neutral conditions is particularly advantageous as it prevents the protonation of the indole nitrogen, which could otherwise deactivate the nucleophile or lead to polymerization side reactions. The reaction kinetics are influenced by the electronic nature of the substituents on the indole rings; electron-rich indoles tend to react more rapidly, while electron-deficient variants may require extended reaction times or slightly elevated temperatures to reach completion. Understanding these electronic effects is crucial for R&D teams aiming to optimize the process for specific analogues. The mechanism avoids the formation of stable metal-complex intermediates, which simplifies the reaction profile and minimizes the risk of catalyst poisoning by sulfur or nitrogen-containing impurities often found in bulk chemical feedstocks.
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this oxidative coupling method offers inherent advantages in this regard. The primary byproducts of the reaction are typically tert-butanol or water, depending on the specific peroxide used, both of which are easily removed during the aqueous workup. The absence of heavy metal catalysts eliminates the need for costly and time-consuming scavenging steps to reduce metal levels to ppm specifications required by regulatory agencies. Furthermore, the high chemoselectivity of the peroxide oxidant ensures that other oxidizable functional groups on the substrate, such as sulfides or amines, remain intact, thereby maintaining the integrity of the molecular scaffold. The reaction mixture can be quenched simply with saturated sodium bicarbonate, which neutralizes any acidic byproducts and facilitates phase separation. This straightforward workup procedure contributes to a cleaner crude product profile, reducing the load on downstream chromatography or crystallization units. For supply chain heads, this translates to reduced lead time for high-purity pharmaceutical intermediates, as fewer purification cycles are required to meet stringent quality standards.
How to Synthesize 2-Quaternary Carbon Indole-3-Ketone Efficiently
Implementing this synthetic route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and thermal management to maximize yield and safety. The protocol is designed to be scalable, utilizing common laboratory glassware or standard stainless steel reactors capable of handling mild oxidative conditions. The key to success lies in the controlled addition of the oxidant and the maintenance of the optimal temperature window to prevent runaway exotherms while ensuring complete conversion. Operators should be familiar with handling peroxide solutions, adhering to strict safety guidelines regarding storage and compatibility with organic materials. The following guide outlines the generalized operational parameters derived from the patent examples, providing a foundation for process validation. Detailed standardized synthesis steps are provided in the section below to ensure reproducibility and compliance with Good Manufacturing Practices (GMP).
- Charge a reaction vessel with peroxide oxidant (e.g., 70% TBHP) and a green solvent such as water.
- Add the substrate 2-substituted indole and the indole coupling partner in a molar ratio ranging from 1: 1 to 1:10.
- Heat the mixture to 60-140°C for 1-48 hours, then quench with saturated sodium bicarbonate and extract the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this peroxide-mediated synthesis offers transformative benefits for procurement managers and supply chain executives focused on cost efficiency and reliability. The shift towards water-based solvent systems represents a significant departure from traditional organic-heavy processes, resulting in substantial cost savings related to solvent purchase, recovery, and waste disposal. By eliminating the dependency on precious metal catalysts, the process removes a major variable cost driver and mitigates the supply risk associated with fluctuating prices of rare earth elements. This economic advantage is compounded by the high reaction yields reported, which maximize the throughput of raw materials and minimize the generation of waste streams that require treatment. For organizations aiming for cost reduction in pharmaceutical intermediate manufacturing, this technology provides a clear pathway to improving gross margins without compromising on product quality. The simplicity of the operation also reduces the training burden on production staff and lowers the likelihood of operator error, further enhancing process robustness.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive peroxide oxidants like TBHP drastically lower the bill of materials for each batch. Additionally, the ability to use water as a solvent removes the need for complex solvent recovery distillation columns, significantly reducing energy consumption and capital expenditure on equipment. The high atom economy of the coupling reaction ensures that a greater proportion of the starting material ends up in the final product, reducing the effective cost per kilogram of the active intermediate. These factors combine to create a highly competitive cost structure that allows for aggressive pricing strategies in the global market while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including substituted indoles and common peroxides, are commodity chemicals with well-established global supply chains, reducing the risk of shortages. The operational simplicity of the reaction means that it can be easily transferred between different manufacturing sites or contract manufacturing organizations without extensive re-validation. This flexibility ensures business continuity even in the face of regional disruptions or logistical challenges. Furthermore, the mild reaction conditions reduce wear and tear on reactor vessels, extending the lifespan of capital assets and minimizing unplanned downtime for maintenance. This reliability is critical for meeting tight delivery schedules and maintaining trust with downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from milligram to gram scales in the patent examples, with clear parameters for expansion to multi-kilogram production. The use of green solvents and the generation of benign byproducts align perfectly with increasingly stringent environmental regulations and corporate sustainability goals. This compliance reduces the regulatory burden and the risk of fines or shutdowns due to environmental violations. The simplified waste stream, primarily consisting of aqueous layers with low organic load, is easier and cheaper to treat, further enhancing the environmental profile of the manufacturing site. This makes the technology attractive for companies looking to improve their ESG (Environmental, Social, and Governance) ratings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing accurate guidance for technical teams evaluating this route. Understanding these nuances is essential for making informed decisions about process adoption and integration into existing manufacturing portfolios. We encourage stakeholders to review these points carefully to assess the fit for their specific project requirements.
Q: What are the primary advantages of this peroxide-mediated synthesis over traditional methods?
A: This method operates under neutral conditions using water as a preferred solvent, eliminating the need for expensive transition metal catalysts and harsh acidic or basic environments, which significantly simplifies downstream purification and reduces environmental impact.
Q: What is the substrate scope for the indole coupling partners in this reaction?
A: The process demonstrates excellent universality, accommodating various substituents on the indole ring including alkyl, aryl, halogen, benzyloxy, nitro, and cyano groups, allowing for the synthesis of diverse bioactive scaffolds.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the use of inexpensive oxidants like TBHP, simple aqueous workup procedures, and high reaction yields up to 96% makes this protocol highly amenable to kilogram-to-ton scale manufacturing for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Quaternary Carbon Indole-3-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing advanced synthetic technologies that drive innovation in drug discovery and development. Our team of expert chemists has thoroughly analyzed the potential of this peroxide-mediated oxidative coupling route and is fully prepared to support your projects from early-stage process development through to commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of 2-quaternary carbon indole-3-ketone meets the highest industry standards. We are committed to being a long-term partner in your success, offering technical expertise that goes beyond simple manufacturing.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific molecule requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient process. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your timelines and reduce your overall development costs. Contact us today to explore how NINGBO INNO PHARMCHEM can serve as your trusted partner for high-quality pharmaceutical intermediates.
