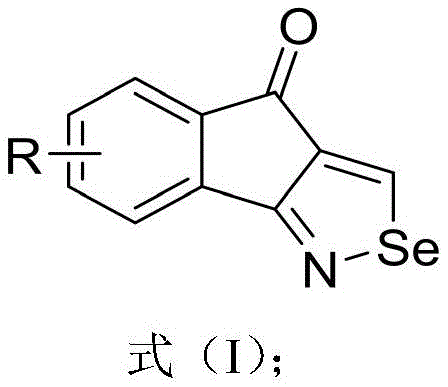
Advanced Silver-Promoted Synthesis of Tetrahydroindenone-Isoselenazole Derivatives for Commercial Scale-Up
Advanced Silver-Promoted Synthesis of Tetrahydroindenone-Isoselenazole Derivatives for Commercial Scale-Up
The landscape of organoselenium chemistry is undergoing a significant transformation, driven by the urgent need for safer, more efficient synthetic routes to bioactive heterocycles. Patent CN114773293A introduces a groundbreaking methodology for the preparation of tetrahydroindenone-isoselenazole derivatives, a class of compounds renowned for their potent biological activities including anti-inflammatory, neuroprotective, and cytoprotective properties. This innovation addresses critical bottlenecks in the production of high-purity pharmaceutical intermediates by leveraging a silver-promoted cyclization strategy that utilizes elemental selenium. For R&D directors and procurement specialists alike, this technology represents a pivotal shift towards sustainable manufacturing, offering a robust pathway to access complex scaffolds that are essential for next-generation drug discovery programs targeting conditions such as Alzheimer's disease and rheumatoid arthritis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoselenazole derivatives has been plagued by substantial technical and economic hurdles that hinder their widespread adoption in commercial pharmaceutical manufacturing. Traditional protocols frequently rely on hazardous selenium reagents that pose significant safety risks and environmental disposal challenges, complicating regulatory compliance for large-scale operations. Furthermore, existing literature often describes multi-step sequences that suffer from poor atom economy and low overall yields, necessitating extensive purification processes that drive up production costs. The difficulty in obtaining specialized starting materials further exacerbates supply chain vulnerabilities, creating lead time uncertainties for downstream API manufacturers who require consistent, high-volume supplies of these critical intermediates for clinical and commercial applications.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN114773293A presents a streamlined, one-pot synthesis that dramatically simplifies the production workflow while enhancing safety profiles. By employing inexpensive and readily available elemental selenium as the selenium source, this novel approach eliminates the need for toxic precursors, thereby reducing raw material costs and waste treatment burdens. The reaction proceeds under mild conditions, typically at 40°C in dimethyl sulfoxide (DMSO), utilizing a synergistic catalytic system of silver acetate and cobalt acetate. This method not only achieves impressive isolated yields ranging from 45% to 88% across various substrates but also demonstrates exceptional functional group tolerance, accommodating both electron-withdrawing and electron-donating substituents with high efficiency.

Mechanistic Insights into Silver-Promoted Cyclization
The core of this technological advancement lies in the intricate interplay between the silver promoter and the cobalt additive, which facilitates a unique cyclization cascade involving o-cyano alkynone precursors. The silver species likely activates the alkyne moiety towards nucleophilic attack by the in situ generated selenide species, initiating the ring closure that forms the isoselenazole core. Simultaneously, the cobalt additive appears to play a crucial role in stabilizing reaction intermediates and promoting the subsequent intramolecular cyclization with the nitrile group to form the fused indanone system. This dual-catalyst system ensures high selectivity and minimizes side reactions, which is paramount for maintaining the stringent purity specifications required for pharmaceutical grade intermediates used in sensitive biological assays and therapeutic applications.
From an impurity control perspective, the mild thermal conditions (25-100°C, optimally 40°C) are instrumental in preventing thermal degradation of the sensitive selenium-containing products. The use of 4Å molecular sieves in the reaction mixture further enhances product quality by sequestering trace moisture that could otherwise hydrolyze reactive intermediates or deactivate the catalyst. This meticulous control over the reaction environment results in a cleaner crude product profile, significantly reducing the burden on downstream purification units. For process chemists, understanding these mechanistic nuances is vital for troubleshooting and optimizing the process during technology transfer, ensuring that the high yields observed in the laboratory can be faithfully reproduced in pilot and commercial plant settings.
How to Synthesize Tetrahydroindenone-Isoselenazole Efficiently
Implementing this synthesis route requires precise adherence to the optimized stoichiometry and reaction parameters established in the patent data to ensure maximum efficiency and reproducibility. The process begins with the careful selection of high-quality o-cyano alkynone starting materials, which can be easily functionalized with various groups such as halogens or alkoxy chains to tailor the final biological activity. The reaction is conducted under an inert nitrogen atmosphere to prevent oxidation of the selenium species, utilizing DMSO as the preferred solvent due to its ability to dissolve both organic substrates and inorganic salts effectively. Detailed standard operating procedures regarding reagent addition rates, temperature ramping, and workup protocols are essential for scaling this chemistry from gram to kilogram quantities without compromising safety or yield.
- Prepare the reaction mixture by combining o-cyano alkynone compounds with elemental selenium in a polar aprotic solvent such as DMSO.
- Add the silver promoter (AgOAc) and cobalt additive (Co(OAc)2) along with molecular sieves to facilitate the cyclization.
- Maintain the reaction at 40°C under nitrogen protection for approximately 2 hours to achieve high yields of the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this silver-promoted synthesis offers tangible strategic benefits that extend beyond mere technical feasibility. The shift towards using elemental selenium as a primary reagent represents a significant cost optimization opportunity, as it replaces expensive and regulated toxic selenium sources with a commodity chemical that is abundant and stable. This substitution not only lowers the direct material costs but also simplifies the logistics of raw material sourcing, reducing the risk of supply disruptions caused by regulatory restrictions on hazardous chemicals. Furthermore, the simplified one-pot nature of the reaction reduces the number of unit operations required, leading to lower utility consumption and reduced labor costs associated with complex multi-step processing.
- Cost Reduction in Manufacturing: The elimination of toxic selenium reagents and the reduction of synthetic steps directly translate to substantial cost savings in the production of these pharmaceutical intermediates. By avoiding the need for specialized containment equipment required for hazardous reagents, capital expenditure for manufacturing facilities can be significantly minimized. Additionally, the high atom utilization rate of this process means less waste generation, which lowers the operational expenses related to waste disposal and environmental compliance, ultimately improving the overall profit margin for the final API product.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials such as o-cyano alkynones and elemental selenium ensures a robust and resilient supply chain. Unlike proprietary or hard-to-source reagents that can create bottlenecks, these commodities are widely produced by multiple global suppliers, mitigating the risk of single-source dependency. This availability allows for flexible procurement strategies and better inventory management, ensuring that production schedules for critical drug candidates remain uninterrupted even in volatile market conditions.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of environmentally friendlier reagents make this process highly amenable to commercial scale-up. Operating at lower temperatures reduces energy consumption and thermal stress on equipment, facilitating safer operation in large reactors. Moreover, the reduced toxicity profile of the reagents aligns with increasingly stringent global environmental regulations, positioning manufacturers as responsible corporate citizens and easing the path for regulatory approvals in key markets like the US and Europe.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these tetrahydroindenone-isoselenazole derivatives. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities. Understanding these aspects is crucial for stakeholders evaluating the feasibility of integrating this new synthetic route into their existing manufacturing portfolios or drug development pipelines.
Q: What are the primary advantages of this silver-promoted method over traditional isoselenazole synthesis?
A: Unlike traditional methods that often require toxic selenium sources and multi-step procedures with low atom utilization, this silver-promoted approach utilizes inexpensive elemental selenium and achieves direct cyclization in a single pot with good yields.
Q: Can this synthesis method accommodate various substituents on the benzene ring?
A: Yes, the method demonstrates excellent universality, successfully tolerating electron-withdrawing groups like halogens (F, Cl, Br) and electron-donating groups such as methyl and methoxy substituents.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Absolutely. The reaction operates at mild temperatures (40°C) and uses readily available raw materials, making it highly scalable and environmentally friendly for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroindenone-Isoselenazole Supplier
As the demand for advanced organoselenium intermediates continues to grow within the pharmaceutical sector, partnering with an experienced CDMO becomes a strategic imperative for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of tetrahydroindenone-isoselenazole derivatives meets the highest quality standards required for clinical and commercial applications.
We invite you to engage with our technical procurement team to discuss how this innovative silver-promoted synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this route for your specific API program. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that accelerate your drug development timelines while optimizing your manufacturing budget.
