Revolutionizing 3-Trifluoromethyl-1,2,4-Triazole Production: A Cost-Effective DMF-Based Route for Global Pharma Supply Chains
Introduction: A Paradigm Shift in Heterocycle Manufacturing
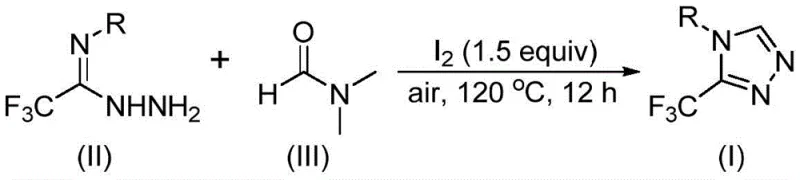
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-rich heterocyclic scaffolds, particularly those bearing fluorinated motifs which are critical for metabolic stability and bioavailability. Patent CN114920707B, published in April 2023, introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds that fundamentally alters the economic and operational landscape of producing these valuable intermediates. This technology leverages the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source, thereby streamlining the synthetic route. For R&D directors and procurement strategists, this represents a significant opportunity to optimize supply chains for key drug candidates like Factor IXa inhibitors and GlyT1 inhibitors, where the 1,2,4-triazole core is a prevalent structural feature. The innovation lies in its operational simplicity, utilizing molecular iodine as a promoter under ambient air conditions, effectively bypassing the stringent requirements for anhydrous and oxygen-free environments that typically plague heterocyclic chemistry.
By integrating this methodology into existing production frameworks, manufacturers can achieve substantial reductions in raw material complexity and energy consumption. The ability to utilize DMF—a low-cost, high-volume commodity chemical—as a reactant eliminates the need for specialized C1 synthons, directly impacting the cost of goods sold (COGS). Furthermore, the tolerance to air and moisture suggests a high degree of process robustness, making it an ideal candidate for large-scale commercial manufacturing where reproducibility and safety are paramount. This report delves deep into the mechanistic nuances, comparative advantages, and commercial implications of this novel synthetic pathway, providing a comprehensive analysis for stakeholders aiming to secure a competitive edge in the global fine chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with significant technical and economic hurdles that hinder efficient commercial scale-up. Conventional routes often rely on the condensation of hydrazines with nitriles or amidines under harsh conditions, frequently requiring strong acids or bases that generate substantial amounts of hazardous waste. Moreover, many established protocols necessitate the use of expensive, specialized fluorinating agents or pre-functionalized building blocks that are not only costly but also suffer from limited availability in the global supply chain. A critical bottleneck in these traditional methods is the stringent requirement for inert atmospheres; the sensitivity of reactive intermediates to oxygen and moisture often mandates the use of nitrogen or argon blankets, specialized Schlenk lines, and rigorously dried solvents. These requirements inflate capital expenditure (CAPEX) for reactor setups and increase operational expenditure (OPEX) through higher utility costs and longer cycle times associated with purging and maintaining inert conditions. Additionally, the purification of products from these complex reaction mixtures often involves tedious chromatographic separations or multiple recrystallization steps, leading to yield losses and increased solvent consumption, which contradicts modern green chemistry principles.
The Novel Approach
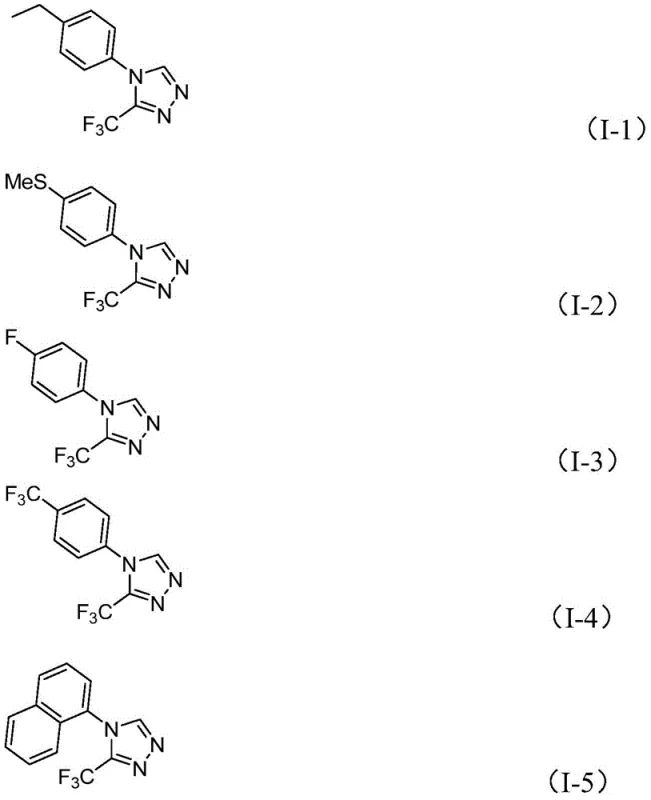
In stark contrast, the method disclosed in patent CN114920707B offers a streamlined, atom-economical alternative that addresses these pain points directly. The core innovation is the utilization of DMF as a dual-function reagent: it serves as the polar aprotic solvent to dissolve reactants and simultaneously provides the carbon atom necessary for ring closure. This dual role drastically simplifies the stoichiometry of the reaction, removing the need for external carbon donors. The reaction is promoted by molecular iodine, a relatively inexpensive and readily available halogen source, which activates the DMF molecule to participate in the cyclization cascade. Perhaps most importantly for industrial application, this transformation proceeds efficiently under an air atmosphere at temperatures between 110°C and 130°C. This elimination of inert gas requirements significantly lowers the barrier to entry for manufacturing, allowing the reaction to be performed in standard stainless steel reactors without specialized modifications. The substrate scope is equally impressive, accommodating a wide range of aromatic substituents including electron-rich and electron-deficient groups, ensuring versatility for diverse medicinal chemistry programs.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific analogues. The reaction initiates with the activation of DMF by molecular iodine. Depending on the specific pathway taken, either the formyl group or the N-methyl group of DMF acts as the carbon source. In the formyl pathway, the carbonyl carbon of DMF undergoes a condensation reaction with the hydrazide moiety of the trifluoroethyliminohydrazide substrate. This forms a hydrazone intermediate, which subsequently undergoes an intramolecular cyclization. The final step in this pathway involves the elimination of a dimethylamine molecule, driving the aromatization of the triazole ring and yielding the desired 3-trifluoromethyl product. Alternatively, when the N-methyl group serves as the carbon source, the mechanism involves the initial formation of an amine salt via the reaction of DMF with iodine. Following the elimination of hydrogen iodide, a nucleophilic addition occurs with the hydrazide, leading to the expulsion of N-methylformamide and the generation of a reactive azadiene intermediate. This intermediate then undergoes intramolecular nucleophilic attack followed by oxidative aromatization to furnish the target heterocycle. The ability of the system to toggle between these pathways depending on substrate electronics provides a level of flexibility that is rare in heterocycle synthesis.
From an impurity control perspective, this mechanism offers distinct advantages. The byproducts generated, such as dimethylamine or N-methylformamide, are highly polar and water-soluble, facilitating their removal during the aqueous workup phase. This contrasts sharply with traditional methods that might generate lipophilic byproducts difficult to separate from the product. Furthermore, the use of iodine as a promoter rather than a stoichiometric oxidant minimizes the formation of iodinated side products, provided the reaction time and temperature are strictly controlled within the 10-15 hour and 110-130°C windows specified in the patent. The robustness of the radical or ionic intermediates involved allows for a broad tolerance of functional groups on the aromatic ring, including halogens and trifluoromethyl groups, without significant degradation or side reactions. This mechanistic resilience ensures that the impurity profile remains manageable even when scaling to multi-kilogram batches, a critical factor for regulatory approval in pharmaceutical applications.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
To implement this synthesis effectively, operators must adhere to the specific parameters outlined in the patent to ensure maximum conversion and yield. The process begins by charging a reaction vessel with the trifluoroethyliminohydrazide starting material and molecular iodine, typically in a molar ratio of 1:1.5, although ratios between 1:1 and 1:2 are acceptable. DMF is then added in excess, serving as the bulk solvent to ensure adequate solubility of the starting materials and to drive the equilibrium forward. The mixture is heated to a temperature range of 110°C to 130°C and maintained for a duration of 10 to 15 hours under ambient air conditions. Monitoring the reaction progress via TLC or HPLC is recommended to determine the precise endpoint, as over-reaction can lead to minor decomposition. Upon completion, the reaction mixture is cooled, and the product is isolated through a straightforward workup involving filtration to remove inorganic salts, followed by silica gel treatment and column chromatography if high purity is required. For large-scale operations, crystallization from appropriate solvent systems may be developed to replace chromatography, further enhancing cost efficiency.
- Combine molecular iodine (1.5 equiv) and trifluoroethyliminohydrazide in a reaction vessel with DMF serving as both solvent and reactant.
- Heat the mixture to 110-130°C under an air atmosphere for 10-15 hours to facilitate the tandem cyclization.
- Upon completion, perform standard post-treatment including filtration, washing, and column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based synthesis strategy offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary driver of value is the drastic simplification of the raw material list. By utilizing DMF as both solvent and reactant, the supply chain is decoupled from the volatility of specialized C1 reagent markets. DMF is a high-volume commodity chemical produced globally with stable pricing and abundant availability, ensuring supply continuity even during market disruptions. This consolidation of materials reduces the number of vendors required, simplifying vendor qualification processes and reducing administrative overhead. Furthermore, the elimination of inert gas requirements translates directly into lower utility costs and reduced dependency on nitrogen or argon supply contracts. In a manufacturing setting, the ability to run reactions open to the air simplifies reactor scheduling and turnover, as the time-consuming purging cycles associated with inert atmospheres are rendered unnecessary. This operational agility allows for faster batch turnover and increased overall equipment effectiveness (OEE).
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the dual role of DMF. By removing the need for expensive, dedicated carbon sources and eliminating the infrastructure costs associated with maintaining anhydrous and anaerobic conditions, the overall production cost is significantly lowered. The use of molecular iodine, a relatively inexpensive promoter, further contributes to a favorable cost structure compared to transition metal-catalyzed alternatives that require costly ligands and metal scavenging steps. Additionally, the simplified workup procedure, which relies on the high polarity of byproducts for easy separation, reduces solvent consumption and waste disposal costs, aligning with sustainability goals while improving the bottom line.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available starting materials. Trifluoroethyliminohydrazide precursors can be synthesized from readily available aromatic amines and trifluoroacetic acid derivatives, commodities that are widely traded. The robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality or minor fluctuations in environmental conditions, reducing the risk of batch failures. This reliability ensures consistent delivery schedules to downstream customers, a critical metric for maintaining long-term contracts with major pharmaceutical companies. The scalability of the method from gram to ton scale without significant re-engineering further secures the supply chain against future demand surges.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this method presents a cleaner profile. The avoidance of strong acids, bases, or pyrophoric reagents reduces the hazard classification of the process, lowering insurance premiums and safety compliance costs. The waste stream is primarily composed of aqueous solutions containing soluble amines and iodide salts, which are easier to treat than organic-heavy waste streams containing heavy metals. The high atom economy of incorporating the solvent into the product structure minimizes the E-factor (mass of waste per mass of product), making this process highly attractive for companies striving to meet rigorous ESG (Environmental, Social, and Governance) targets. The ease of scale-up ensures that pilot plant success can be rapidly translated into commercial production without the typical teething problems associated with changing reaction regimes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within patent CN114920707B, providing a factual basis for decision-making. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing portfolios. The clarity provided here aims to mitigate risks associated with technology transfer and process validation.
Q: What is the primary advantage of using DMF in this triazole synthesis?
A: DMF acts dually as the reaction solvent and the carbon source (providing either the formyl or methyl group), which eliminates the need for separate, expensive C1 building blocks and simplifies the workup process significantly.
Q: Does this reaction require strict anhydrous or oxygen-free conditions?
A: No, unlike many traditional heterocycle syntheses, this iodine-promoted method operates efficiently under a standard air atmosphere, removing the capital and operational costs associated with inert gas manifolds and gloveboxes.
Q: What is the substrate scope for the R-group in this synthesis?
A: The method demonstrates broad compatibility with various substituted aryl groups, including those with electron-donating (methyl, methoxy) and electron-withdrawing (fluoro, chloro, trifluoromethyl) substituents, yielding products with moderate to good efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in CN114920707B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this DMF-based route are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific thermal and handling requirements of iodine-promoted reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the global pharmaceutical industry. We are committed to delivering not just a chemical product, but a reliable supply solution that enhances your project's velocity and cost-efficiency.
We invite you to collaborate with us to leverage this advanced chemistry for your next drug development program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog of triazole derivatives or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in turning innovative patent chemistry into commercial reality.
