Advanced Ruthenium Catalysis for Efficient Synthesis of Nitrogen-Heterocyclic Ketones
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more sustainable and efficient methodologies to access critical building blocks. Patent CN112645984B introduces a significant breakthrough in this domain with the development of a novel half-sandwich ruthenium complex containing a phenyl benzoxazole structure. This innovative catalyst addresses long-standing challenges in the preparation of nitrogen-containing heterocyclic ketones, which are indispensable intermediates in the synthesis of active pharmaceutical ingredients (APIs) and agrochemicals. The structural integrity and unique electronic properties of this complex allow for catalytic oxidation reactions to proceed under remarkably mild conditions, utilizing atmospheric air as the terminal oxidant. This represents a paradigm shift from traditional stoichiometric oxidation methods that often generate substantial waste. For R&D directors and process chemists, this technology offers a robust platform for developing cleaner synthetic routes with improved atom economy.
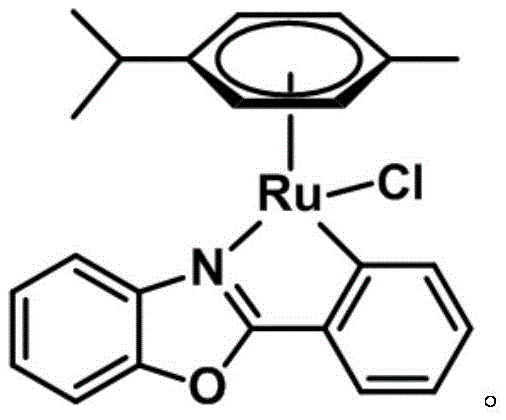
Furthermore, the preparation of the catalyst itself is designed with green chemistry principles in mind, avoiding toxic solvents and extreme temperatures. The resulting complex is not only highly active but also exhibits exceptional stability against moisture and oxygen, a rare trait for many organometallic catalysts. This stability translates directly into practical benefits for supply chain managers, as the catalyst can be stored and handled without the need for inert atmosphere gloveboxes, reducing logistical complexity and cost. As we delve deeper into the technical specifics, it becomes clear that this invention provides a reliable solution for the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and supply continuity for downstream manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocyclic ketones has relied heavily on classical methodologies that are fraught with operational hazards and inefficiencies. The Friedel-Crafts acylation, while a staple in aromatic chemistry, is notoriously ineffective for pyridine substrates due to the strong electron-withdrawing nature of the nitrogen atom, which deactivates the ring towards electrophilic attack. Consequently, chemists have often resorted to the use of halogenated pyridines reacting with Grignard reagents. However, this approach imposes severe constraints on the reaction environment, necessitating strictly anhydrous and oxygen-free conditions to prevent the decomposition of the highly reactive organometallic reagents. Such requirements demand specialized equipment, rigorous solvent drying protocols, and inert gas protection, all of which escalate production costs and increase the risk of batch failures. Moreover, the generation of stoichiometric amounts of magnesium salts as byproducts creates significant waste disposal challenges, conflicting with modern environmental regulations and sustainability goals.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112645984B leverages the unique reactivity of the phenyl benzoxazole-based ruthenium complex to overcome these inherent limitations. This novel approach facilitates the direct oxidation of alkyl pyridine compounds to their corresponding ketones using molecular oxygen from the air, eliminating the need for hazardous stoichiometric oxidants or sensitive organometallic reagents. The reaction proceeds efficiently at room temperature in common solvents like dichloromethane, drastically simplifying the operational protocol. By bypassing the need for cryogenic conditions or high-pressure reactors, this method significantly lowers the energy footprint of the synthesis. The catalyst's ability to activate C-H bonds selectively ensures high yields with minimal byproduct formation, streamlining the purification process. This transition from harsh, waste-generating protocols to a mild, catalytic aerobic oxidation system marks a substantial advancement in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Ru-Catalyzed Aerobic Oxidation
The efficacy of this system lies in the sophisticated design of the half-sandwich ruthenium center coordinated with the phenyl benzoxazole ligand. The "piano-stool" geometry of the complex, featuring a cymene ligand and the chelating benzoxazole moiety, creates a stable electronic environment that protects the metal center from deactivation. Unlike many other transition metal catalysts that are prone to rapid oxidation or hydrolysis, this specific ruthenium complex maintains its structural integrity even when exposed to ambient air and moisture. This robustness is critical for maintaining catalytic turnover numbers over extended periods. The mechanism likely involves the coordination of the pyridine nitrogen to the ruthenium center, facilitating the activation of the benzylic C-H bond. Subsequent interaction with molecular oxygen allows for the insertion of the oxygen atom, regenerating the active catalyst species in a closed loop. This cycle avoids the accumulation of reduced metal species that typically plague aerobic oxidations, ensuring consistent reaction rates throughout the process.
From an impurity control perspective, the mild reaction conditions play a pivotal role in ensuring high product purity. Traditional high-temperature oxidations often lead to over-oxidation or degradation of sensitive functional groups on the substrate. However, operating at room temperature minimizes thermal stress on the molecule, preserving delicate substituents such as halogens or ethers. The high selectivity of the catalyst means that side reactions are suppressed, resulting in a cleaner crude reaction mixture. This reduces the burden on downstream purification steps, such as column chromatography or recrystallization, which are often the most resource-intensive parts of API intermediate production. For quality control teams, this translates to a more predictable impurity profile and easier validation of the manufacturing process, ultimately accelerating the timeline from laboratory discovery to commercial production.
How to Synthesize Half-Sandwich Ruthenium Complex Efficiently
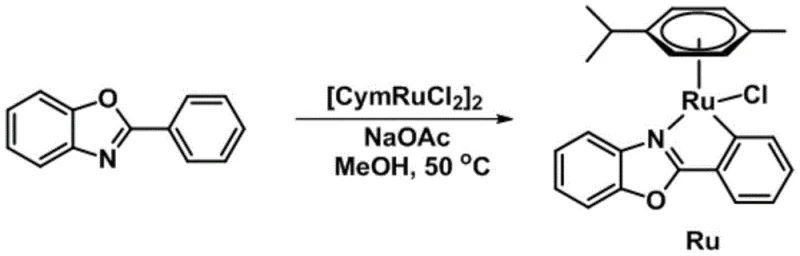
The preparation of the catalyst is designed to be straightforward and scalable, utilizing readily available starting materials and standard laboratory equipment. The process involves the reaction of phenylbenzoxazole with a ruthenium dimer precursor in the presence of a base, followed by a simple workup procedure. This accessibility ensures that the catalyst can be produced in-house or sourced reliably without complex supply chains. The detailed standardized synthesis steps below outline the precise conditions required to achieve optimal yield and purity, serving as a guide for process engineers looking to implement this technology.
- Prepare the catalyst by reacting phenylbenzoxazole, [CymRuCl2]2, and sodium acetate in methanol at 50°C, followed by purification via column chromatography.
- Dissolve the alkyl pyridine substrate in dichloromethane and add the ruthenium complex catalyst (0.01-0.05 molar ratio).
- Stir the reaction mixture at room temperature under an air atmosphere until completion, then concentrate and purify the product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed technology offers tangible strategic advantages beyond mere technical performance. The shift towards using air as the primary oxidant eliminates the recurring cost of purchasing specialized chemical oxidants, which can be expensive and subject to volatile market pricing. Furthermore, the simplified reaction setup reduces the capital expenditure required for specialized reactor vessels capable of handling high pressures or extreme temperatures. The robustness of the catalyst also implies a longer shelf life and reduced waste due to spoilage, contributing to substantial cost savings in inventory management. By integrating this method, companies can achieve a more resilient supply chain that is less dependent on fragile reagent supplies and more aligned with green chemistry mandates.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants and the use of ambient air significantly lower the raw material costs per kilogram of product. Additionally, the mild reaction conditions reduce energy consumption associated with heating or cooling, leading to lower utility bills. The simplified workup procedure minimizes solvent usage and waste disposal fees, further enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The stability of the catalyst against air and moisture ensures that it can be transported and stored under standard conditions, removing the need for costly cold chain logistics or inert atmosphere packaging. This reliability mitigates the risk of supply disruptions caused by shipping delays or improper handling. Moreover, the use of common solvents like dichloromethane and methanol ensures that raw material availability remains high, preventing bottlenecks in production scheduling.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production without significant re-engineering. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, reducing the compliance burden and potential liability for the manufacturer. This eco-friendly profile enhances the corporate sustainability image, which is becoming a key differentiator in the global fine chemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic system. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: What are the primary advantages of this ruthenium catalyst over traditional Grignard methods?
A: Unlike Grignard reactions which require strict anhydrous and oxygen-free conditions, this ruthenium-catalyzed oxidation operates at room temperature using air as the oxidant, significantly simplifying operational safety and equipment requirements.
Q: Is the half-sandwich ruthenium complex stable for long-term storage?
A: Yes, the catalyst exhibits high physicochemical stability and is insensitive to both air and water, allowing for stable existence in ambient environments without special preservation measures.
Q: What types of substrates are compatible with this oxidation process?
A: The method demonstrates broad substrate universality, effectively oxidizing various alkyl pyridine compounds including those with electron-withdrawing or electron-donating groups such as halogens, methyl, and methoxy substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Half-Sandwich Ruthenium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the phenyl benzoxazole ruthenium complex in driving innovation within the pharmaceutical and agrochemical sectors. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of catalyst or intermediate delivered meets the highest international standards. We are committed to supporting your R&D efforts with high-purity materials that enable reproducible and robust synthetic outcomes.
We invite you to collaborate with our technical team to explore how this cutting-edge oxidation methodology can optimize your specific production lines. By leveraging our expertise, you can unlock significant efficiencies and reduce your time-to-market for critical drug candidates. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate the viability of this superior catalytic solution for your business.
