Advanced Ruthenium Catalysis for Efficient Synthesis of Nitrogen-Heterocyclic Ketones
The landscape of fine chemical synthesis is constantly evolving, driven by the urgent need for greener, more efficient catalytic systems that can operate under mild conditions. Patent CN112645984B introduces a groundbreaking advancement in this field with the development of a novel half-sandwich ruthenium complex containing a phenyl benzoxazole structure. This sophisticated organometallic catalyst represents a significant leap forward for the production of nitrogen-containing heterocyclic ketones, which are critical building blocks in the pharmaceutical and agrochemical industries. Unlike conventional catalysts that often demand rigorous exclusion of moisture or elevated temperatures, this new ruthenium species boasts exceptional stability against air and water, fundamentally altering the operational parameters for industrial oxidation reactions. By enabling the use of ambient air as a terminal oxidant at room temperature, this technology not only enhances safety profiles but also drastically reduces the energy footprint associated with heating and pressurization in large-scale manufacturing environments.
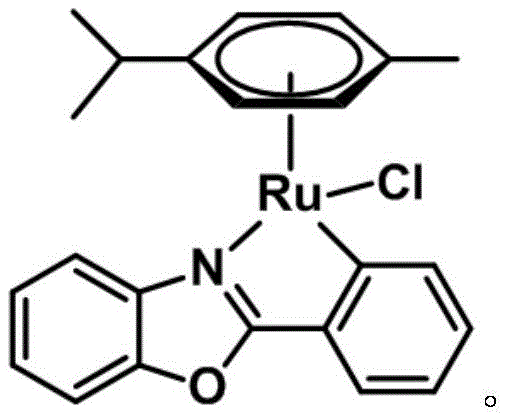
For R&D directors and process chemists, the structural integrity and electronic properties of this catalyst, as depicted in the provided imagery, suggest a robust coordination environment that facilitates efficient C-H activation. The integration of the phenyl benzoxazole ligand creates a unique steric and electronic pocket around the ruthenium center, optimizing it for the selective oxidation of alkyl pyridines. This innovation addresses a long-standing challenge in heterocyclic chemistry where the electron-withdrawing nature of the pyridine ring often deactivates the aromatic system towards traditional electrophilic substitutions. Consequently, this patent offers a reliable pathway for accessing valuable ketone intermediates that were previously difficult or expensive to synthesize, positioning it as a key technology for any organization seeking a reliable pharmaceutical intermediate supplier with cutting-edge capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocyclic ketones has been plagued by significant technical hurdles and safety concerns inherent to classical methodologies. The Friedel-Crafts acylation, a staple in aromatic chemistry, fails miserably when applied to pyridine substrates due to the strong electron-withdrawing effect of the nitrogen atom, which deactivates the ring towards electrophilic attack. To circumvent this, chemists have traditionally resorted to using halogenated pyridines reacting with Grignard reagents; however, this approach imposes severe constraints, requiring strictly anhydrous and oxygen-free conditions to prevent reagent decomposition. Furthermore, alternative oxidative methods reported in literature often rely on toxic solvents like benzonitrile, require pure oxygen gas which presents explosion hazards, or necessitate high temperatures exceeding 130 °C, leading to substantial energy consumption and potential thermal degradation of sensitive functional groups. These limitations collectively result in high production costs, complex waste treatment protocols, and restricted substrate scope, making them ill-suited for modern sustainable manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach detailed in the patent utilizes the aforementioned half-sandwich ruthenium complex to catalyze the oxidation of alkyl pyridines under remarkably benign conditions. The preparation of the catalyst itself is straightforward and green, involving the simple mixing of phenyl benzoxazole, a ruthenium dimer precursor, and sodium acetate in methanol, followed by mild heating. 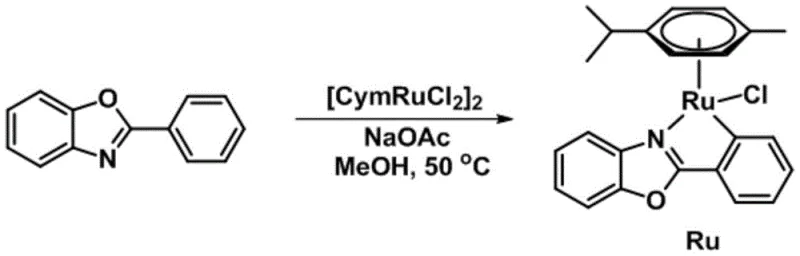 This streamlined synthesis avoids the use of hazardous reagents and produces the active catalyst in high yield with excellent selectivity. Once deployed, the catalyst enables the transformation of alkyl pyridines to their corresponding ketones using nothing more than ambient air as the oxidant in dichloromethane at room temperature. This paradigm shift eliminates the need for expensive oxidants, high-pressure reactors, or energy-intensive heating systems, thereby offering a compelling solution for cost reduction in fine chemical manufacturing while simultaneously expanding the range of accessible chemical space for drug discovery teams.
This streamlined synthesis avoids the use of hazardous reagents and produces the active catalyst in high yield with excellent selectivity. Once deployed, the catalyst enables the transformation of alkyl pyridines to their corresponding ketones using nothing more than ambient air as the oxidant in dichloromethane at room temperature. This paradigm shift eliminates the need for expensive oxidants, high-pressure reactors, or energy-intensive heating systems, thereby offering a compelling solution for cost reduction in fine chemical manufacturing while simultaneously expanding the range of accessible chemical space for drug discovery teams.
Mechanistic Insights into Ru-Catalyzed Aerobic Oxidation
The efficacy of this catalytic system lies in the unique mechanistic pathway facilitated by the phenyl benzoxazole-ligated ruthenium center. While the precise catalytic cycle involves complex organometallic steps, it is understood that the ruthenium species activates the benzylic C-H bond of the alkyl pyridine substrate through a concerted metalation-deprotonation or radical-type mechanism, stabilized by the robust ligand framework. The presence of the cymene ligand provides the necessary half-sandwich geometry that balances stability with reactivity, allowing the metal center to cycle between oxidation states efficiently without decomposing. Crucially, the catalyst's insensitivity to water and oxygen means that the reaction does not require the rigorous exclusion of atmospheric components, which is a common failure point for many transition metal catalysts. This robustness ensures that the active species remains intact throughout the reaction duration, maintaining high turnover numbers and preventing the formation of inactive ruthenium black precipitates that often plague aerobic oxidations.
From an impurity control perspective, this mechanism offers distinct advantages over non-catalytic or harsh oxidative methods. Traditional strong oxidants often lead to over-oxidation, ring cleavage, or the formation of N-oxides, creating a complex impurity profile that is difficult and costly to purge. The mild nature of this ruthenium-catalyzed aerobic oxidation ensures high chemoselectivity, targeting specifically the benzylic position while leaving other sensitive functional groups such as halogens (bromo, chloro) and ethers (methoxy) intact. 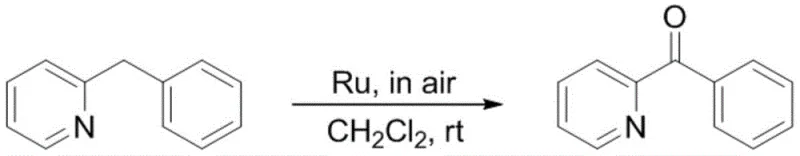 As demonstrated in the substrate scope examples, the system tolerates a wide variety of substituents, yielding high-purity nitrogen-containing heterocyclic ketones with minimal byproduct formation. This high selectivity simplifies downstream purification, often allowing for direct silica gel chromatography without extensive workup procedures, which is a critical factor for maintaining throughput in commercial production settings.
As demonstrated in the substrate scope examples, the system tolerates a wide variety of substituents, yielding high-purity nitrogen-containing heterocyclic ketones with minimal byproduct formation. This high selectivity simplifies downstream purification, often allowing for direct silica gel chromatography without extensive workup procedures, which is a critical factor for maintaining throughput in commercial production settings.
How to Synthesize Half-Sandwich Ruthenium Complex Efficiently
The preparation of this high-performance catalyst is designed for operational simplicity, making it accessible for both laboratory scale optimization and pilot plant production. The process begins with the dissolution of the organic ligand and metal precursor in a common alcohol solvent, followed by the addition of a mild base to facilitate ligand exchange. The reaction proceeds smoothly at moderate temperatures, avoiding the need for cryogenic conditions or specialized high-pressure equipment. Detailed standardized synthetic steps see the guide below, which outlines the precise stoichiometric ratios and purification techniques required to achieve the reported 81% yield and high purity specifications necessary for catalytic applications.
- Dissolve phenyl benzoxazole, [CymRuCl2]2, and sodium acetate in methanol at room temperature with a molar ratio of 1: 0.5:2.
- Heat the reaction system to 50 °C and maintain stirring for 3 to 6 hours to ensure complete complexation.
- Filter the mixture, remove solvent under reduced pressure, and purify the red crude product via column chromatography using petroleum ether and dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits that extend beyond mere technical performance. The shift from hazardous, energy-intensive processes to a mild, air-driven catalytic system fundamentally alters the cost structure and risk profile of producing nitrogen-heterocyclic intermediates. By eliminating the dependency on pure oxygen supplies, anhydrous solvents, and high-temperature reactors, manufacturers can significantly reduce capital expenditure on safety infrastructure and lower operational expenses related to energy consumption and waste disposal. This process intensification allows for faster batch cycles and higher throughput, directly addressing the industry-wide challenge of reducing lead time for high-purity pharmaceutical intermediates while ensuring a more resilient and flexible supply chain.
- Cost Reduction in Manufacturing: The economic implications of switching to this ruthenium-catalyzed protocol are profound, primarily driven by the simplification of reaction conditions and the use of air as a free, abundant oxidant. Traditional methods often incur high costs associated with the purchase and handling of stoichiometric oxidants, the maintenance of inert atmospheres, and the energy required to sustain elevated temperatures for extended periods. In contrast, this novel method operates at room temperature, effectively eliminating heating costs and reducing the cooling load required for exothermic quenches. Furthermore, the high selectivity of the catalyst minimizes the formation of side products, which reduces the burden on purification units and lowers the overall solvent consumption per kilogram of product, leading to substantial cost savings in raw materials and waste treatment.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized reagents that may have volatile market availability or long lead times. This catalytic system utilizes readily available starting materials such as alkyl pyridines and common solvents like dichloromethane and methanol, which are commodity chemicals with stable global supply networks. The catalyst itself is stable to air and moisture, meaning it can be synthesized, stored, and transported without the need for expensive inert packaging or cold chain logistics. This robustness ensures that production schedules are not disrupted by reagent degradation or delivery delays, providing a reliable foundation for long-term manufacturing contracts and just-in-time inventory strategies.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden bottlenecks related to heat transfer and safety, particularly with exothermic oxidations. The mild, room-temperature nature of this reaction makes it inherently safer and easier to scale, as there is no risk of thermal runaway associated with high-temperature operations. Additionally, the use of air as an oxidant generates water as the primary byproduct, aligning perfectly with green chemistry principles and reducing the generation of hazardous heavy metal waste or toxic organic byproducts. This environmental compatibility simplifies regulatory compliance and permitting processes, facilitating the commercial scale-up of complex organometallic catalysts and ensuring sustainable production practices that meet increasingly stringent global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ruthenium catalytic technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on operational parameters and expected outcomes for potential adopters of this methodology.
Q: What are the primary advantages of this ruthenium catalyst over traditional oxidation methods?
A: Unlike traditional methods requiring strong acids, anhydrous conditions, or pure oxygen, this catalyst operates at room temperature using ambient air as the oxidant, significantly simplifying safety protocols and equipment requirements.
Q: Is the catalyst stable during storage and handling?
A: Yes, the half-sandwich ruthenium complex exhibits high physicochemical stability and is insensitive to both air and moisture, allowing for standard storage conditions without specialized inert atmosphere gloveboxes.
Q: What is the substrate scope for this catalytic oxidation?
A: The method demonstrates broad universality, successfully oxidizing various alkyl pyridine derivatives including those with electron-withdrawing groups like bromine and chlorine, as well as electron-donating groups like methyl and methoxy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Half-Sandwich Ruthenium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the phenyl benzoxazole ruthenium complex in driving innovation within the pharmaceutical and fine chemical sectors. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of catalyst or intermediate we produce adheres to the highest quality standards required by global regulatory bodies.
We invite forward-thinking organizations to collaborate with us to leverage this cutting-edge oxidation technology for their specific synthesis needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current production bottlenecks, demonstrating exactly how this mild catalytic route can optimize your bottom line. We encourage you to contact us today to obtain specific COA data for our catalyst offerings and to discuss route feasibility assessments that could accelerate your project timelines and enhance your competitive advantage in the market.
