Advanced Synthesis of Thiostrepton Derivatives for Commercial Scale-up of Complex Antibiotic Intermediates
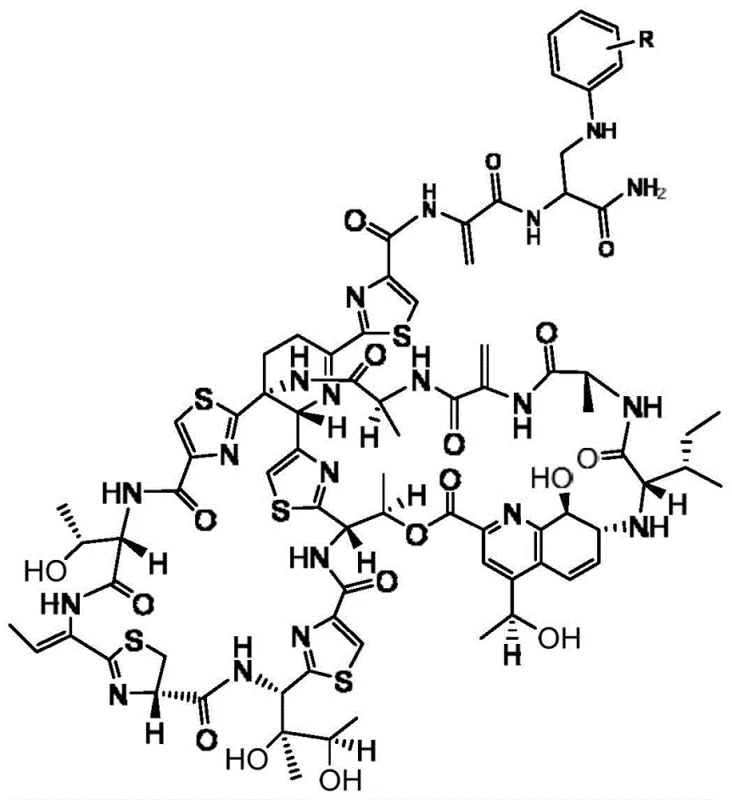
The pharmaceutical industry continuously seeks robust synthetic pathways to modify natural products for enhanced therapeutic efficacy, particularly in the realm of combating multi-drug resistant bacteria. Patent CN111196839B introduces a groundbreaking methodology for the preparation of thiostrepton derivatives, addressing the critical limitations of the parent compound regarding water solubility and bioavailability. This innovation utilizes a triethylamine-catalyzed reaction in chloroform, enabling the efficient coupling of thiostrepton with various aniline derivatives to generate novel antibacterial agents. The structural versatility of this approach allows for the introduction of diverse functional groups, such as halogens and trifluoromethyl moieties, which are essential for optimizing pharmacokinetic profiles. By leveraging this specific chemical transformation, manufacturers can access a library of high-potency intermediates that serve as the foundation for next-generation antibiotics targeting Gram-positive pathogens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for modifying complex thiopeptide antibiotics like thiostrepton often suffer from harsh reaction conditions that compromise the integrity of the sensitive peptide backbone. Conventional protocols may require elevated temperatures or strong acidic/basic environments, leading to significant degradation of the starting material and the formation of complex impurity profiles that are difficult to separate. Furthermore, the poor solubility of thiostrepton in common organic solvents frequently results in heterogeneous reaction mixtures, causing inconsistent conversion rates and low overall yields. These inefficiencies not only increase the cost of goods but also extend the development timeline for new drug candidates, creating a bottleneck in the supply chain for critical antibiotic intermediates. The lack of regioselectivity in older methods further complicates the purification process, necessitating extensive chromatographic steps that reduce the final throughput.
The Novel Approach
In stark contrast, the method disclosed in CN111196839B employs a mild, homogeneous reaction system using chloroform as the solvent and triethylamine as a catalyst, which dramatically improves substrate solubility and reaction controllability. This novel approach operates effectively at room temperature, specifically around 25 ℃, preserving the delicate stereochemistry of the thiostrepton core while facilitating high-yield coupling with aniline nucleophiles. Experimental examples within the patent demonstrate isolated yields reaching up to 90% for unsubstituted aniline and consistently exceeding 70% for various substituted derivatives, indicating a highly efficient process suitable for scale-up. The use of readily available aniline derivatives allows for rapid diversification of the chemical space, enabling medicinal chemists to explore structure-activity relationships without the burden of complex synthetic routes. This streamlined protocol minimizes side reactions and simplifies downstream processing, making it an ideal candidate for industrial adoption.
Mechanistic Insights into Triethylamine-Catalyzed Condensation
The core of this synthetic breakthrough lies in the activation of the thiostrepton molecule towards nucleophilic attack by the aniline derivative, facilitated by the basic environment provided by triethylamine. Mechanistically, the dehydroalanine moiety within the thiostrepton structure acts as a Michael acceptor, undergoing conjugate addition with the amino group of the aniline substrate. The triethylamine serves to deprotonate the aniline slightly or stabilize the transition state, thereby lowering the activation energy required for the bond formation without inducing elimination or hydrolysis side reactions. This specific interaction ensures that the modification occurs selectively at the desired site, maintaining the integrity of the surrounding thiazole rings and peptide bonds which are crucial for biological activity. The reaction proceeds smoothly in chloroform due to the excellent solvation of both the lipophilic thiostrepton and the aromatic amine reactants, creating a unified phase that maximizes molecular collisions.
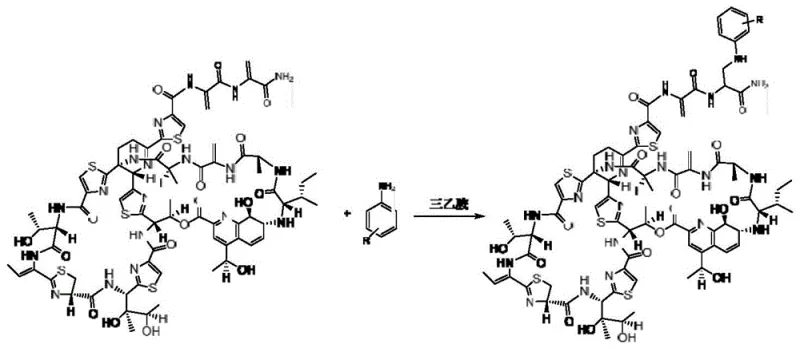
Controlling the impurity profile in such complex macrocyclic systems is paramount for regulatory compliance and patient safety, and this method excels by minimizing thermal stress. By maintaining the reaction temperature between 15-30 ℃, the process avoids the thermal degradation pathways that typically generate open-ring byproducts or epimerized species. The stoichiometry is carefully optimized, with a preferred molar ratio of thiostrepton to aniline of 1:20, ensuring that the expensive antibiotic core is fully consumed while driving the equilibrium towards the product. Subsequent purification via column chromatography using a methanol and dichloromethane gradient effectively removes unreacted aniline and catalyst residues, while the final semi-preparative HPLC step guarantees the high-purity specifications required for pharmaceutical applications. This rigorous control over the reaction parameters results in a clean crude extract, significantly reducing the load on purification columns and extending their operational life.
How to Synthesize Thiostrepton Derivative Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and environmental controls to replicate the high yields reported in the patent literature. The process begins with the dissolution of the reactants in chloroform, followed by a controlled incubation period that allows the condensation to reach completion without rushing the kinetics. Detailed standard operating procedures regarding the specific grades of solvents and the order of addition are critical to ensuring batch-to-batch consistency and maximizing the recovery of the valuable product. For a comprehensive breakdown of the exact experimental conditions and workup procedures, please refer to the standardized synthesis guide below.
- Dissolve thiostrepton, aniline derivatives, and triethylamine in chloroform at a molar ratio of 1: 20:10.
- Maintain the reaction mixture at 25 ℃ for 24 hours to ensure complete conversion.
- Purify the crude extract using column chromatography followed by semi-preparative HPLC to obtain the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial strategic benefits by utilizing commodity chemicals that are widely available in the global market, thereby mitigating supply chain risks associated with exotic reagents. The reliance on simple aniline derivatives and triethylamine means that sourcing is straightforward and cost-effective, allowing purchasing managers to negotiate better terms due to the high volume availability of these raw materials. Furthermore, the elimination of expensive transition metal catalysts or specialized ligands removes a significant cost center from the bill of materials, directly contributing to a lower cost of goods sold for the final active pharmaceutical ingredient. The mild reaction conditions also translate to reduced energy consumption, as there is no need for cryogenic cooling or high-temperature heating, aligning with sustainability goals and lowering utility overheads.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by eliminating the need for precious metal catalysts and complex protecting group strategies that are often required in peptide modification. By utilizing a direct condensation approach with high atom economy, the method reduces waste generation and solvent usage, which lowers the environmental disposal costs associated with chemical manufacturing. The high yields observed across a broad range of substrates mean that less starting material is required to produce the same amount of product, effectively stretching the budget for raw materials. Additionally, the simplified purification workflow reduces the consumption of chromatography media and HPLC columns, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: Utilizing widely sourced aniline derivatives ensures that production schedules are not disrupted by the scarcity of niche reagents, providing a stable foundation for long-term manufacturing planning. The robustness of the reaction conditions allows for flexibility in production timing, as the process is not overly sensitive to minor fluctuations in ambient temperature or humidity, reducing the risk of batch failures. This reliability enables supply chain heads to maintain leaner inventory levels of finished goods, knowing that production lead times are predictable and consistent. The scalability of the chloroform-based system also means that technology transfer from lab to plant is seamless, preventing delays during the commercialization phase.
- Scalability and Environmental Compliance: The use of chloroform, while requiring careful handling, is a well-understood solvent in industrial settings with established recovery and recycling protocols that minimize environmental impact. The reaction generates minimal hazardous byproducts, simplifying the waste treatment process and ensuring compliance with stringent environmental regulations regarding effluent discharge. The ability to run the reaction at near-room temperature reduces the carbon footprint of the manufacturing process, supporting corporate sustainability initiatives. Moreover, the high purity of the crude product reduces the solvent load in downstream purification, contributing to a greener overall process profile that appeals to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this thiostrepton derivative synthesis, drawing directly from the experimental data and scope defined in the patent documentation. Understanding these nuances is essential for R&D teams looking to adapt this chemistry for their specific drug discovery programs or process development workflows. The answers provided reflect the optimal conditions identified during the patent examples to ensure the highest quality outcomes.
Q: What is the primary advantage of the triethylamine-catalyzed method for thiostrepton derivatives?
A: The method described in CN111196839B offers superior substrate solubility and controllability, achieving yields up to 90% under mild conditions compared to traditional methods.
Q: Which aniline derivatives are compatible with this synthesis route?
A: The process supports a wide range of substrates including halogenated anilines (F, Cl, Br, I) and trifluoromethyl-substituted anilines, allowing for diverse structure-activity relationship studies.
Q: How is the purity of the final thiostrepton derivative ensured?
A: High purity is achieved through a two-stage purification process involving initial column chromatography to remove bulk impurities, followed by semi-preparative HPLC for final isolation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiostrepton Derivative Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to bring this advanced synthetic route from the laboratory bench to full-scale commercial production. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency and quality. We employ stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and potency of every batch of thiostrepton derivative we produce. Our commitment to excellence means that we can handle the complex purification requirements of these macrocyclic compounds, delivering material that meets the exacting standards of the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this innovative chemistry can be integrated into your supply chain for cost-effective antibiotic development. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this triethylamine-catalyzed method for your specific project needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a smooth and successful partnership.
