Scalable Synthesis of Thiostrepton Derivatives for Advanced Antibiotic Development and Commercial Supply
Scalable Synthesis of Thiostrepton Derivatives for Advanced Antibiotic Development and Commercial Supply
The escalating crisis of antimicrobial resistance demands continuous innovation in the discovery and development of next-generation antibiotics, particularly those targeting Gram-positive bacteria. Patent CN111196839B introduces a groundbreaking technical solution for the preparation of thiostrepton derivatives, a class of compounds derived from the natural product thiostrepton (TSR). This patent details a highly efficient, triethylamine-catalyzed modification strategy that addresses the historical limitations of TSR, such as poor water solubility and low bioavailability, while maintaining its potent antibacterial activity. By enabling the systematic introduction of diverse aniline moieties onto the thiostrepton scaffold, this technology opens new avenues for Structure-Activity Relationship (SAR) studies and the creation of novel therapeutic agents. The method is characterized by its operational simplicity, utilizing mild reaction conditions that are inherently safer and more cost-effective for large-scale manufacturing.
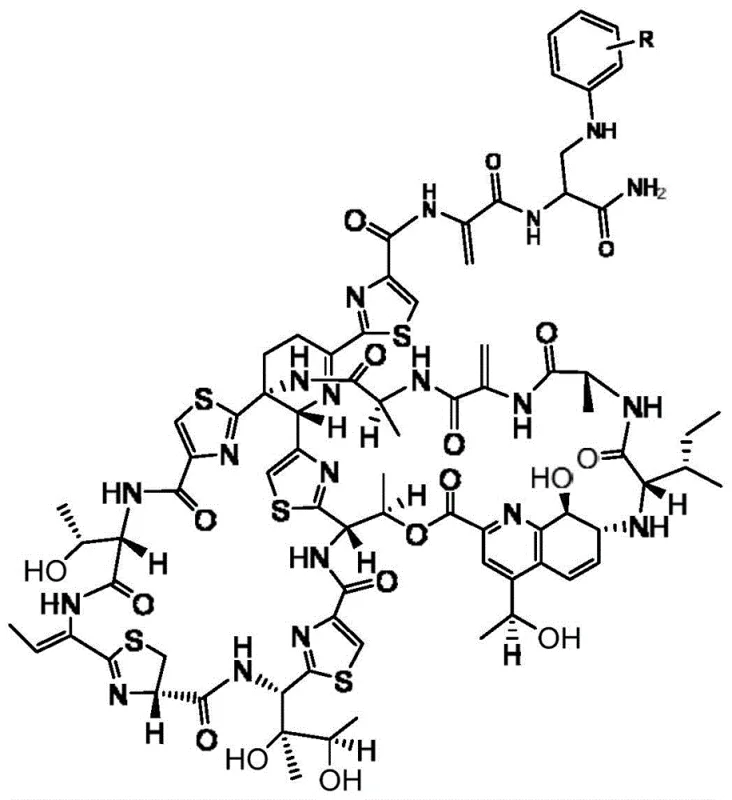
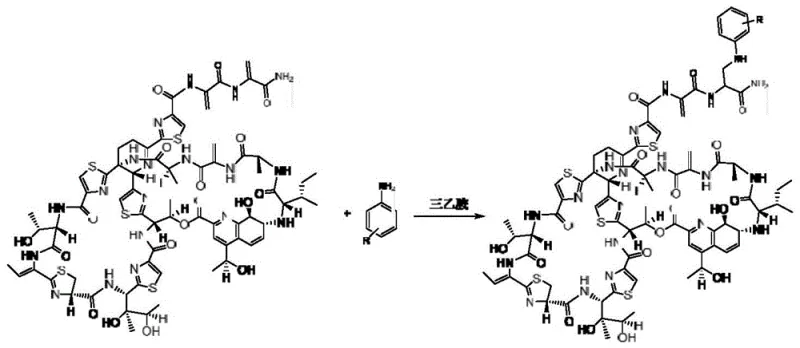
The core innovation lies in the specific chemical transformation where thiostrepton reacts with aniline and its derivatives in a chloroform solvent system under the catalysis of triethylamine. This approach is not merely a incremental improvement but represents a significant leap in process chemistry for complex thiopeptide antibiotics. The resulting derivatives, where the R group can be hydrogen, halogens (F, Cl, Br, I), or various alkyl and alkoxy groups, exhibit enhanced biological profiles. For pharmaceutical R&D directors, this means access to a versatile library of intermediates that can be rapidly synthesized and screened. The robustness of the reaction conditions ensures that the integrity of the complex thiostrepton macrocycle is preserved during modification, a critical factor in maintaining the desired pharmacological properties while improving physicochemical characteristics like solubility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical modification of complex natural products like thiostrepton has been fraught with challenges that hinder both research progress and commercial viability. Traditional methods often suffer from harsh reaction conditions that can degrade the sensitive peptide backbone or the thiazole rings inherent to the thiostrepton structure. These aggressive conditions frequently lead to low reaction yields, complex impurity profiles that are difficult to separate, and poor reproducibility across different batches. Furthermore, many conventional solvents and catalysts used in peptide modification are toxic or environmentally hazardous, creating significant disposal costs and regulatory hurdles for manufacturing facilities. The lack of substrate generality in older methods also restricts the ability to explore diverse chemical space, limiting the potential for discovering derivatives with superior efficacy or reduced toxicity. Consequently, the supply of high-quality thiostrepton analogues for preclinical and clinical development has often been bottlenecked by inefficient synthetic routes.
The Novel Approach
In stark contrast, the method disclosed in CN111196839B utilizes a mild, triethylamine-catalyzed system in chloroform that operates effectively at ambient temperatures ranging from 15°C to 30°C. This gentle approach minimizes the risk of thermal degradation and side reactions, ensuring high fidelity in the final product structure. The patent data demonstrates exceptional yields, with specific examples achieving up to 90% conversion efficiency, which is a remarkable metric for modifying such a complex molecule. The use of chloroform provides excellent solubility for both the bulky thiostrepton substrate and the various aniline derivatives, facilitating homogeneous reaction kinetics. This homogeneity is crucial for consistent quality control and simplifies the downstream purification process. By shifting to this milder protocol, manufacturers can significantly reduce energy consumption associated with heating or cooling, aligning with modern green chemistry principles while simultaneously lowering operational expenditures.
Mechanistic Insights into Triethylamine-Catalyzed Modification
The mechanistic elegance of this synthesis relies on the nucleophilic activation facilitated by triethylamine within the chloroform medium. Triethylamine acts as a base to deprotonate or activate the reactive sites on the thiostrepton molecule, likely targeting the dehydroalanine moiety or specific electrophilic centers susceptible to nucleophilic attack by the aniline nitrogen. This catalytic cycle proceeds efficiently without the need for transition metal catalysts, which eliminates the risk of heavy metal contamination—a critical quality attribute for pharmaceutical intermediates intended for human use. The absence of metals simplifies the purification workflow, as there is no need for expensive scavenging resins or complex chelation steps to meet strict residual metal specifications. The reaction kinetics are optimized by the specific molar ratios defined in the patent, such as a thiostrepton-to-aniline ratio of 1:20 and a thiostrepton-to-triethylamine ratio of 1:10, ensuring that the equilibrium is driven strongly towards product formation.
Impurity control is inherently built into this process due to the high selectivity of the triethylamine catalysis. The mild conditions prevent the formation of polymeric byproducts or degradation fragments that often plague peptide chemistry. The patent highlights that the method produces few side reaction products, which translates to a cleaner crude reaction mixture. This cleanliness is vital for the subsequent semi-preparative HPLC purification step, allowing for higher recovery rates of the target compound and extending the lifespan of chromatography columns. For quality assurance teams, this means a more predictable impurity profile that is easier to characterize and control, reducing the regulatory burden during the drug approval process. The ability to tolerate a wide range of functional groups on the aniline ring—from electron-withdrawing halogens to electron-donating methyl groups—further underscores the robustness of the mechanistic pathway.
How to Synthesize Thiostrepton Derivatives Efficiently
Implementing this synthesis route requires precise adherence to the stoichiometric ratios and environmental controls outlined in the patent to maximize yield and purity. The process begins with the dissolution of the starting materials in chloroform, followed by a controlled reaction period that balances conversion completeness with throughput time. Detailed standard operating procedures for scaling this reaction from milligram to kilogram quantities involve careful monitoring of reaction progress, typically via HPLC, to determine the exact endpoint before workup. The following guide summarizes the critical operational parameters derived from the patent examples to assist process chemists in replicating this high-efficiency transformation.
- Dissolve thiostrepton, aniline derivatives, and triethylamine in chloroform solution at a molar ratio of 1: 10-30 for aniline and 1:5-15 for triethylamine.
- Maintain the reaction mixture at ambient temperature (15-30°C, preferably 25°C) for 12 to 36 hours to ensure complete conversion.
- Purify the collected reaction liquid via column chromatography followed by semi-preparative HPLC to isolate the high-purity thiostrepton derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis method offers tangible strategic benefits that extend beyond simple chemical yield. The elimination of transition metal catalysts removes a significant cost center associated with precious metal procurement and waste management. Furthermore, the use of commodity chemicals like triethylamine and chloroform ensures a stable and reliable supply chain for raw materials, mitigating the risk of shortages that can occur with specialized reagents. The mild temperature requirements mean that existing reactor infrastructure can be utilized without the need for costly retrofits for extreme heating or cryogenic cooling capabilities. This compatibility with standard equipment accelerates the timeline from process development to commercial production, allowing companies to bring new antibiotic candidates to market faster.
- Cost Reduction in Manufacturing: The high reaction yields observed in the patent examples, reaching up to 90%, directly correlate to improved material efficiency and reduced waste generation. By maximizing the conversion of the expensive thiostrepton starting material into the valuable derivative, the cost per gram of the active intermediate is significantly lowered. Additionally, the simplified purification process, necessitated by the clean reaction profile, reduces the consumption of solvents and chromatography media, which are often major cost drivers in fine chemical manufacturing. The avoidance of heavy metals also eliminates the expense of metal testing and removal protocols, further streamlining the cost structure.
- Enhanced Supply Chain Reliability: The broad substrate scope of this reaction allows for flexibility in sourcing aniline derivatives. If a specific substituted aniline becomes unavailable or cost-prohibitive, the robust nature of the chemistry suggests that alternative analogues could potentially be synthesized with minimal process re-optimization. This flexibility provides a buffer against supply chain volatility. Moreover, the short reaction time of approximately 24 hours at ambient temperature facilitates rapid batch turnover, increasing overall production capacity and ensuring consistent delivery schedules to downstream clients.
- Scalability and Environmental Compliance: The green chemistry attributes of this method, specifically the low energy input and reduced hazardous waste, align perfectly with increasingly stringent environmental regulations. Scaling this process from laboratory to industrial scale does not introduce new safety hazards related to high pressure or temperature, making it inherently safer for plant operators. The reduced environmental footprint enhances the corporate sustainability profile of the manufacturing entity, which is becoming a key criterion for partnerships with major pharmaceutical companies committed to ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these thiostrepton derivatives. The answers are derived directly from the experimental data and claims presented in patent CN111196839B, providing a reliable foundation for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this technology into their existing pipelines.
Q: What are the optimal reaction conditions for synthesizing thiostrepton derivatives?
A: According to patent CN111196839B, the optimal conditions involve using chloroform as the solvent with triethylamine as a catalyst at 25°C for 24 hours, achieving yields up to 90%.
Q: Which aniline derivatives are compatible with this synthesis method?
A: The method demonstrates wide substrate applicability, successfully reacting with various substituted anilines including fluoro-, chloro-, bromo-, iodo-, methyl-, and trifluoromethyl-anilines.
Q: How does this method improve upon traditional thiostrepton modification techniques?
A: This novel approach offers superior solubility, strong controllability, and environmentally friendly characteristics with fewer side reaction products compared to conventional harsh modification methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiostrepton Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the race against antibiotic resistance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of thiostrepton derivative meets the exacting standards required for pharmaceutical development. Our commitment to technical excellence ensures that our clients receive materials that are not only chemically pure but also fully documented for regulatory submissions.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for your antibiotic programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and supply chain planning. Let us be your trusted partner in delivering the next generation of life-saving antimicrobial therapies.
