Revolutionizing Arylamide Production: A Metal-Free Route for High-Purity Pharmaceutical Intermediates
Revolutionizing Arylamide Production: A Metal-Free Route for High-Purity Pharmaceutical Intermediates
In the rapidly evolving landscape of fine chemical manufacturing, the demand for efficient, environmentally benign, and cost-effective synthetic routes for pharmaceutical intermediates has never been more critical. A groundbreaking development detailed in patent CN111606820B introduces a novel preparation method for N-iodobenzene-N-phenylamide compounds that fundamentally shifts the paradigm away from traditional transition metal catalysis. This technology leverages a metal-free oxidative coupling strategy utilizing meta-chloroperoxybenzoic acid (m-CPBA) as the oxidant and hexafluoroisopropanol (HFIP) as the solvent system. By operating under mild thermal conditions ranging from 40°C to 80°C, this process not only achieves exceptional yields but also addresses the growing regulatory and economic pressures associated with heavy metal contamination in active pharmaceutical ingredients (APIs). For R&D directors and procurement specialists alike, this represents a significant opportunity to streamline supply chains and reduce the total cost of ownership for complex arylamide scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-aryl amides and their iodinated derivatives has relied heavily on transition metal-catalyzed cross-coupling reactions, predominantly employing copper (Cu) or palladium (Pd) complexes. While these methods are chemically robust, they suffer from inherent limitations that pose significant challenges for large-scale industrial application. The reliance on precious metals like palladium introduces substantial volatility in raw material pricing, directly impacting the cost structure of the final intermediate. Furthermore, the presence of residual metal catalysts in the final product is a major regulatory hurdle, necessitating rigorous and often expensive purification steps such as scavenging or recrystallization to meet stringent ppm limits set by health authorities. Additionally, these traditional processes often generate substantial amounts of metal-containing waste streams, complicating environmental compliance and increasing the burden on waste treatment facilities. The reaction conditions can also be harsh, requiring high temperatures or inert atmospheres that increase energy consumption and operational complexity.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN111606820B offers a transformative solution by completely eliminating the need for transition metal catalysts. This metal-free protocol utilizes a direct oxidative coupling between substituted acetanilides and substituted iodobenzenes, driven by the strong oxidizing power of m-CPBA in the unique medium of HFIP. 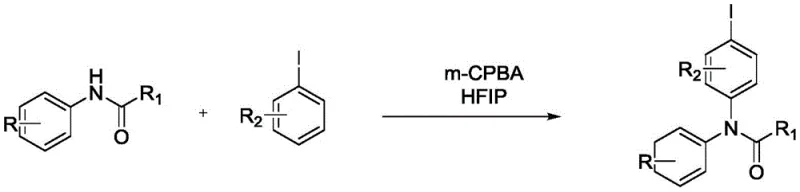 The reaction proceeds smoothly under reflux conditions in an oil bath, typically maintaining temperatures between 40°C and 80°C for a duration of 4 to 10 hours. This approach not only simplifies the reaction setup by removing the need for specialized ligands or inert gas protection but also drastically reduces the impurity profile associated with metal residues. The use of HFIP is particularly ingenious, as its strong hydrogen-bond donating ability activates the oxidant and stabilizes reactive intermediates, leading to high regioselectivity and superior yields often exceeding 90% in optimized examples. This shift towards organocatalytic or reagent-driven oxidation marks a pivotal advancement for reliable pharmaceutical intermediate supplier networks seeking greener alternatives.
The reaction proceeds smoothly under reflux conditions in an oil bath, typically maintaining temperatures between 40°C and 80°C for a duration of 4 to 10 hours. This approach not only simplifies the reaction setup by removing the need for specialized ligands or inert gas protection but also drastically reduces the impurity profile associated with metal residues. The use of HFIP is particularly ingenious, as its strong hydrogen-bond donating ability activates the oxidant and stabilizes reactive intermediates, leading to high regioselectivity and superior yields often exceeding 90% in optimized examples. This shift towards organocatalytic or reagent-driven oxidation marks a pivotal advancement for reliable pharmaceutical intermediate supplier networks seeking greener alternatives.
Mechanistic Insights into Metal-Free Oxidative Coupling
The mechanistic pathway of this transformation is fascinating from a physical organic chemistry perspective, primarily revolving around the activation of the iodine species and the nucleophilic attack by the amide nitrogen. In the presence of HFIP, the m-CPBA oxidant is significantly activated through hydrogen bonding interactions, which enhance its electrophilicity. This activated oxidant likely generates a hypervalent iodine species in situ from the substituted iodobenzene, creating a highly reactive intermediate capable of undergoing ligand coupling. The acetanilide nitrogen, acting as a nucleophile, attacks this activated iodine center, facilitating the formation of the new N-I bond. The unique solvation properties of HFIP play a critical role here; it stabilizes the developing positive charge on the nitrogen and the iodine species, lowering the activation energy barrier for the coupling step. This allows the reaction to proceed efficiently at relatively low temperatures compared to traditional thermal coupling methods, preserving sensitive functional groups on the aromatic rings.
Furthermore, the selectivity of this reaction is a key attribute for ensuring high-purity N-iodobenzene-N-phenylamide outputs. The steric and electronic environment created by the HFIP solvent shell directs the substitution pattern, favoring the formation of the para-iodo product relative to the amide group in many substrates, as evidenced by the specific examples in the patent data. For instance, when 2-iodotoluene is used, the reaction selectively targets the position para to the iodine or ortho to the methyl group depending on the specific electronic activation, minimizing the formation of regioisomers that are difficult to separate. This intrinsic selectivity reduces the burden on downstream purification processes like column chromatography or preparative HPLC. By controlling the stoichiometry carefully—typically using a slight excess of the iodobenzene and oxidant (e.g., a molar ratio of 0.2:0.3:0.3 for acetanilide:iodobenzene:m-CPBA)—the process ensures complete conversion of the limiting reagent while suppressing side reactions such as over-oxidation or polymerization, resulting in a clean crude product profile.
How to Synthesize N-Iodobenzene-N-Phenylamide Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The process begins with the precise charging of reagents into a reactor equipped with a reflux condenser and magnetic stirring capability. The choice of solvent purity is paramount; the patent specifies using hexafluoroisopropanol with a purity of 98% to ensure optimal solvation effects. Following the addition of the substituted acetanilide, substituted iodobenzene, and m-CPBA, the mixture is heated in a silicone oil bath, ensuring the immersion depth is sufficient to maintain uniform thermal distribution throughout the reaction vessel. The detailed standardized synthesis steps, including specific workup procedures and purification protocols, are outlined in the guide below to assist technical teams in replicating these high-yielding results.
- Charge the reactor with substituted acetanilide, substituted iodobenzene, m-chloroperoxybenzoic acid (m-CPBA), and hexafluoroisopropanol (HFIP) solvent.
- Heat the reaction mixture in an oil bath at 40-80°C under reflux conditions with magnetic stirring for 4-10 hours to ensure complete conversion.
- Quench the reaction with distilled water, extract the organic phase three times with an organic solvent, concentrate under reduced pressure, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metal-free synthesis route offers profound advantages for procurement managers and supply chain heads tasked with optimizing costs and ensuring continuity. The most immediate impact is seen in the raw material cost structure; by eliminating palladium or copper catalysts, which are subject to significant market price fluctuations and geopolitical supply risks, manufacturers can achieve substantial cost savings in pharmaceutical intermediate manufacturing. The removal of these metals also means that the expensive and time-consuming steps associated with metal scavenging resins or specialized filtration are no longer required, further reducing the operational expenditure per kilogram of product. Moreover, the simplified waste profile, devoid of heavy metals, lowers the cost of environmental compliance and waste disposal, contributing to a more sustainable and economically viable production model.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts directly translates to a lower bill of materials, as expensive ligands and metal salts are replaced by more commodity-grade oxidants like m-CPBA. Additionally, the simplified downstream processing reduces the consumption of auxiliary materials such as scavengers and specialized filter aids. The high yields reported in the patent examples, often reaching up to 98%, mean that less raw material is wasted, improving the overall atom economy and reducing the cost per unit of the final active ingredient. This efficiency gain is critical for maintaining competitive pricing in the global market for fine chemicals.
- Enhanced Supply Chain Reliability: Relying on transition metals often exposes supply chains to volatility due to mining constraints and export restrictions in key producing regions. By shifting to an organic oxidant-based system, the dependency on these critical minerals is severed, thereby enhancing the resilience of the supply chain. The reagents used, such as substituted iodobenzenes and acetanilides, are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. This diversification of the supply base ensures consistent availability of key starting materials, allowing for more reliable production scheduling and on-time delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions (40-80°C) and the use of standard glass-lined or stainless-steel reactors make this process highly amenable to scale-up from kilogram to multi-ton quantities without the need for specialized high-pressure equipment. The absence of heavy metals simplifies the environmental permitting process and reduces the regulatory burden associated with effluent treatment. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this chemistry into existing production lines.
Q: What are the primary advantages of this metal-free synthesis over traditional copper or palladium catalysis?
A: The primary advantage is the elimination of expensive and toxic transition metal catalysts like Cu or Pd. This significantly reduces raw material costs, removes the need for complex heavy metal removal steps during purification, and minimizes environmental waste, aligning with green chemistry principles.
Q: What is the role of Hexafluoroisopropanol (HFIP) in this reaction mechanism?
A: HFIP acts as a unique solvent that enhances the electrophilicity of the oxidant (m-CPBA) through strong hydrogen bonding interactions. It stabilizes cationic intermediates and improves regioselectivity, allowing the reaction to proceed under mild conditions (40-80°C) with high yields.
Q: Is this process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable. It utilizes standard equipment (oil baths, reflux condensers) and avoids hazardous high-pressure or cryogenic conditions. The simple workup procedure involving extraction and distillation facilitates easy scale-up from laboratory to industrial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Iodobenzene-N-Phenylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in modernizing the production of complex pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity N-iodobenzene-N-phenylamide derivatives that meet the most stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality assurance guarantees that every batch delivered is fully characterized and compliant with international regulatory standards, providing our partners with the confidence they need to advance their drug development pipelines.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free route can optimize your budget without compromising quality. Please contact us today to request specific COA data for our available inventory or to discuss route feasibility assessments for custom synthesis projects. Let us be your partner in driving innovation and efficiency in your supply chain.
