Advanced Metal-Free Production of N-Iodobenzene-N-Phenylamide Compounds for Global Pharma Supply Chains
Advanced Metal-Free Production of N-Iodobenzene-N-Phenylamide Compounds for Global Pharma Supply Chains
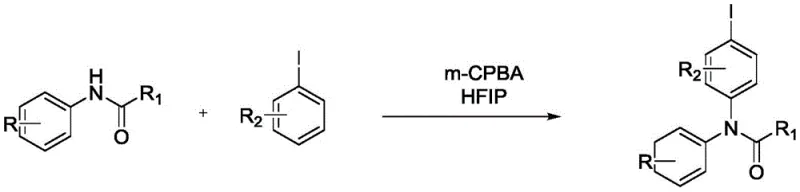
The landscape of fine chemical manufacturing is undergoing a significant paradigm shift towards greener, more sustainable synthetic methodologies, particularly for high-value intermediates used in drug discovery and development. A pivotal advancement in this domain is detailed in Chinese Patent CN111606820B, which discloses a novel, metal-free preparation method for N-iodobenzene-N-phenylamide compounds. This technology addresses critical bottlenecks in traditional aryl amide synthesis by replacing hazardous transition metal catalysts with a robust organic oxidative system. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate supplier partnerships, this patent represents a strategic opportunity to optimize production costs while adhering to stringent environmental regulations. The process utilizes m-chloroperoxybenzoic acid (m-CPBA) as a stoichiometric oxidant in hexafluoroisopropanol (HFIP), enabling the direct coupling of substituted acetanilides with substituted iodobenzenes under remarkably mild conditions. By leveraging this innovative approach, manufacturers can achieve high selectivity and yield without the complications associated with heavy metal residues, thereby streamlining the path from laboratory scale to commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-aryl amides and related biaryl amine structures has relied heavily on transition metal-catalyzed cross-coupling reactions, such as the Buchwald-Hartwig amination. These traditional protocols typically necessitate the use of palladium or copper catalysts, which introduce substantial economic and environmental liabilities to the manufacturing process. The reliance on precious metals like palladium creates significant cost volatility, as market prices for these commodities can fluctuate wildly, impacting the overall cost reduction in API manufacturing strategies. Furthermore, the presence of metal catalysts mandates rigorous downstream purification steps to meet strict regulatory limits for residual metals in active pharmaceutical ingredients, often requiring specialized scavenger resins or complex crystallization sequences. Beyond the financial burden, the disposal of metal-containing waste streams poses a severe environmental challenge, conflicting with modern green chemistry principles and increasing the ecological footprint of chemical production facilities. Additionally, these metal-catalyzed reactions often require harsh conditions, including high temperatures and strong bases, which can compromise the integrity of sensitive functional groups present in complex molecular scaffolds. The cumulative effect of these factors is a process that is not only expensive and operationally complex but also increasingly difficult to justify in an era of heightened sustainability scrutiny.
The Novel Approach
In stark contrast to legacy methods, the technology outlined in CN111606820B offers a transformative metal-free alternative that fundamentally reshapes the synthetic landscape for these valuable intermediates. This innovative route employs a combination of m-chloroperoxybenzoic acid (m-CPBA) and hexafluoroisopropanol (HFIP) to facilitate the oxidative coupling of acetanilides with iodobenzenes, completely bypassing the need for transition metal catalysts. The reaction proceeds efficiently at moderate temperatures between 40°C and 80°C, significantly reducing energy consumption compared to high-thermal demand processes. The use of HFIP as a solvent is particularly advantageous due to its unique ability to stabilize reactive intermediates through hydrogen bonding networks, thereby enhancing reaction rates and selectivity without the need for exotic ligands. This methodology not only simplifies the reaction setup by eliminating air-sensitive catalysts but also drastically reduces the generation of hazardous metal waste, aligning perfectly with global trends towards environmentally friendly chemical synthesis. By adopting this metal-free strategy, producers can achieve superior purity profiles directly from the reactor, minimizing the need for extensive post-reaction remediation and accelerating the overall production timeline.
Mechanistic Insights into Metal-Free Oxidative Coupling
The mechanistic underpinning of this transformation relies on the unique oxidative properties of m-CPBA in conjunction with the specialized solvation effects of HFIP to activate the amide nitrogen for nucleophilic attack or radical coupling. In this system, m-CPBA acts as a potent oxygen transfer agent that likely generates an N-centered radical or a highly electrophilic nitrogen species from the substituted acetanilide precursor. The hexafluoroisopropanol solvent plays a dual critical role: firstly, it acts as a hydrogen-bond donor that stabilizes the developing charge in the transition state, lowering the activation energy barrier for the C-N bond formation. Secondly, HFIP enhances the electrophilicity of the oxidant and helps to solubilize the organic substrates effectively, ensuring a homogeneous reaction environment that promotes consistent kinetics. This synergistic interaction allows for the direct functionalization of the iodobenzene ring by the activated amide nitrogen, forging the crucial C-N bond without the necessity of a metal center to mediate electron transfer. The result is a highly selective process that tolerates a variety of substituents on both the amide and the aryl iodide components, demonstrating broad substrate scope as evidenced by the successful synthesis of diverse derivatives including methyl, ethyl, and chloro-substituted analogs.
From an impurity control perspective, the absence of metal catalysts inherently eliminates a major class of potential contaminants that are notoriously difficult to remove from final products. In traditional metal-catalyzed routes, side reactions such as homocoupling of the aryl halide or dehalogenation can occur, often exacerbated by the presence of metal species that promote unproductive radical pathways. The metal-free oxidative protocol described here minimizes these competing pathways by relying on a controlled organic oxidation mechanism that is specific to the N-H activation of the amide. Furthermore, the mild reaction conditions (40-80°C) prevent thermal degradation of sensitive functional groups, ensuring that the structural integrity of complex molecules is maintained throughout the synthesis. The high yields reported, reaching up to 98% in optimized examples, suggest that the reaction equilibrium strongly favors product formation, reducing the accumulation of unreacted starting materials that would otherwise complicate purification. This level of control over the reaction trajectory is essential for producing high-purity OLED material or pharmaceutical intermediates where trace impurities can have detrimental effects on downstream performance or biological activity.
How to Synthesize N-Iodobenzene-N-Phenylamide Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, making it highly attractive for both laboratory optimization and large-scale manufacturing environments. The process begins with the straightforward charging of reagents into a standard reactor, followed by a controlled heating phase that requires minimal supervision due to the robustness of the chemical system. Detailed standard operating procedures for scaling this reaction from gram to kilogram quantities are critical for ensuring reproducibility and safety, particularly when handling oxidants like m-CPBA. The following guide outlines the fundamental steps derived from the patent data to assist technical teams in replicating this efficient methodology.
- Charge the reactor with substituted acetanilide, substituted iodobenzene, m-chloroperoxybenzoic acid (m-CPBA), and hexafluoroisopropanol (HFIP) solvent.
- Heat the reaction mixture in an oil bath at 40-80°C with magnetic stirring and condense reflux for 4-10 hours to ensure complete conversion.
- Quench the reaction with water, extract the organic phase three times with an organic solvent, concentrate under reduced pressure, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free technology translates into tangible strategic benefits that extend far beyond simple reaction chemistry. The elimination of expensive transition metals like palladium removes a significant variable cost driver, leading to substantial cost savings in raw material expenditure over the lifecycle of the product. Moreover, the simplified workup procedure, which avoids the need for specialized metal scavenging resins or complex filtration steps, reduces the consumption of auxiliary materials and shortens the batch cycle time. This efficiency gain allows for higher throughput in existing manufacturing assets, effectively increasing capacity without the need for capital-intensive infrastructure upgrades. The supply chain for the requisite reagents, such as m-CPBA and HFIP, is well-established and stable, mitigating the risks associated with sourcing scarce precious metals that are often subject to geopolitical tensions and supply disruptions.
- Cost Reduction in Manufacturing: The most immediate financial impact of this technology is the drastic reduction in catalyst costs, as the process completely obviates the need for palladium or copper complexes which are among the most expensive reagents in organic synthesis. By removing these costly inputs, the overall bill of materials is significantly lowered, improving the gross margin profile for the final intermediate. Additionally, the waste treatment costs are markedly reduced because the effluent streams do not contain heavy metals that require specialized and expensive remediation processes before discharge. The combination of lower raw material costs and reduced waste disposal fees creates a compelling economic case for switching to this metal-free methodology, especially for high-volume production runs where even small per-unit savings aggregate into significant total value.
- Enhanced Supply Chain Reliability: Relying on transition metals introduces inherent supply chain vulnerabilities due to the concentrated geographic sources of these elements and their susceptibility to market volatility. In contrast, the organic reagents used in this patented process, such as substituted acetanilides and iodobenzenes, are commodity chemicals with robust and diversified global supply networks. This diversification ensures a steady flow of materials, reducing the risk of production stoppages caused by raw material shortages. Furthermore, the stability of the reagents allows for longer shelf life and easier storage conditions compared to air-sensitive metal catalysts, simplifying inventory management and logistics planning for the procurement team.
- Scalability and Environmental Compliance: Scaling chemical processes that involve heavy metals often triggers complex regulatory hurdles regarding environmental impact assessments and worker safety protocols. This metal-free approach sidesteps many of these regulatory burdens, facilitating a smoother path to commercial scale-up of complex pharmaceutical intermediates. The mild reaction conditions also enhance process safety by reducing the risk of thermal runaways associated with highly exothermic metal-catalyzed reactions. From an environmental standpoint, the reduction in hazardous waste generation aligns with corporate sustainability goals and helps manufacturers maintain compliance with increasingly stringent environmental protection laws, thereby safeguarding the company's social license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal-free synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers evaluating this route for their specific applications. Understanding these nuances is crucial for assessing the feasibility of integrating this method into existing production workflows.
Q: Does this synthesis method require transition metal catalysts like Palladium or Copper?
A: No, the patented process described in CN111606820B is explicitly metal-free. It utilizes m-chloroperoxybenzoic acid (m-CPBA) as an oxidant in hexafluoroisopropanol (HFIP), eliminating the need for expensive and toxic Pd or Cu catalysts traditionally used in aryl amination.
Q: What are the typical reaction conditions and yields for this transformation?
A: The reaction operates under mild thermal conditions ranging from 40°C to 80°C. Experimental data indicates high efficiency, with specific examples achieving isolated yields up to 98% after standard aqueous workup and chromatographic purification.
Q: How does this method impact the impurity profile compared to conventional cross-coupling?
A: By avoiding transition metals, the process inherently prevents heavy metal contamination, significantly simplifying the purification workflow. This results in a cleaner impurity profile, reducing the burden on downstream metal scavenging steps often required for API compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Iodobenzene-N-Phenylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis technology described in CN111606820B for the production of high-value aryl amide intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to leveraging advanced green chemistry techniques to deliver superior products that align with our customers' sustainability and efficiency goals.
We invite you to collaborate with us to explore how this innovative metal-free route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can support your next breakthrough in drug development.
