High-Selectivity Synthesis of Enantiopure 1,4-Dihydropyridines Using Novel Maleimide Auxiliaries
High-Selectivity Synthesis of Enantiopure 1,4-Dihydropyridines Using Novel Maleimide Auxiliaries
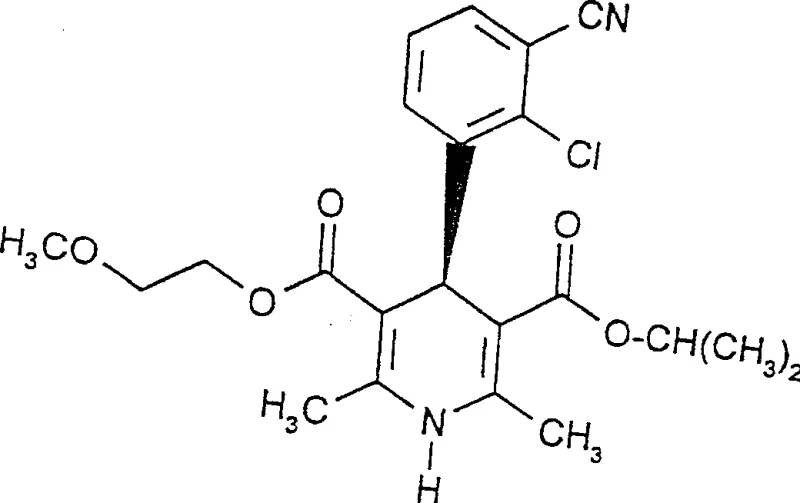
The pharmaceutical industry continuously demands higher purity standards for calcium channel blockers, where the absolute stereochemical structure dictates pharmacological efficacy. Patent CN1105102C discloses a groundbreaking high-selectivity method for preparing enantiomerically pure halogenated phenyl substituted 1,4-dihydropyridine-3,5-dicarboxylates. This technology addresses the critical challenge of introducing chirality without the cumbersome separation processes associated with traditional chiral pool auxiliaries. By utilizing a novel maleimide-based chiral auxiliary, the process achieves exceptional diastereomeric excess and simplifies the downstream purification workflow significantly. This report analyzes the technical merits and commercial viability of this synthetic route for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of enantiopure 1,4-dihydropyridines often relies on separating diastereomeric esters using chiral pool auxiliaries, a process fraught with practical difficulties. The introduction and subsequent elimination of these bulky auxiliary groups frequently require harsh conditions that compromise yield and introduce complex impurity profiles. Furthermore, the removal of large amounts of auxiliary reagents is technologically and chemically complicated, often necessitating multiple chromatographic steps that are untenable for commercial scale-up. These inefficiencies lead to significant material loss and increased production costs, creating a bottleneck for reliable pharmaceutical intermediate supplier operations seeking to optimize their manufacturing footprint.
The Novel Approach
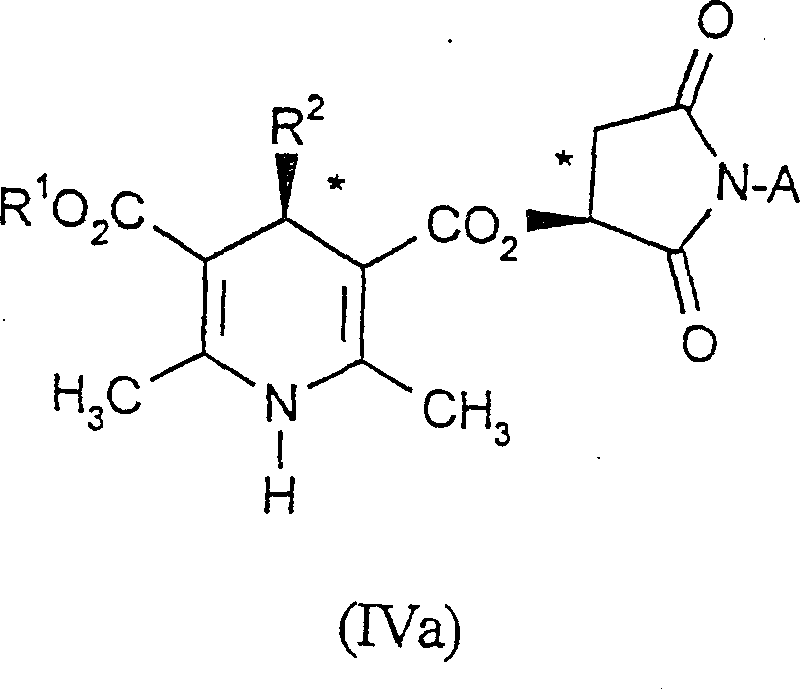
The patented method introduces a highly selective route using a maleimide group as a chiral auxiliary, which can be incorporated into either the benzal compound or the amino-butenate precursor. This approach surprisingly allows for the formation of diastereomerically pure 1,4-dihydropyridines that can be easily purified via crystallization due to the rigid annular structure of the maleimide moiety. The maleimide group can be eliminated under mild basic conditions, optionally isolating the free acid before esterification, ensuring the integrity of the sensitive dihydropyridine ring. This streamlined pathway drastically reduces the number of unit operations and eliminates the need for expensive transition metal catalysts often found in asymmetric hydrogenation routes.
Mechanistic Insights into Maleimide-Assisted Asymmetric Condensation
The core of this technology lies in the condensation reaction between an enantiopure benzal compound and a chiral amino-butenate derivative in an inert solvent. The reaction typically proceeds at temperatures between 60°C and 90°C, preferably in isopropanol or ethyl acetate, facilitating the formation of the 1,4-dihydropyridine ring with high stereocontrol. The rigidity of the maleimide ring restricts conformational freedom during the cyclization, forcing the reaction through a specific transition state that favors one diastereomer over the other. This inherent stereochemical bias is the key to achieving diastereomeric excess values greater than 99.5% as demonstrated in the patent examples, far surpassing typical racemic synthesis outcomes.
Following the cyclization, the removal of the auxiliary is achieved using mild bases such as 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) at room temperature. This step cleaves the maleimide group without racemizing the chiral center at the 4-position of the dihydropyridine ring. The resulting free acid can then be activated using condensing agents like carbonyl diimidazole or dicyclohexylcarbodiimide and esterified with the desired alcohol. This modular approach allows for the systematic variation of the ester groups at the 3 and 5 positions, providing flexibility for generating diverse libraries of calcium antagonist intermediates while maintaining high optical purity throughout the sequence.
How to Synthesize Enantiopure Dihydropyridine Derivatives Efficiently
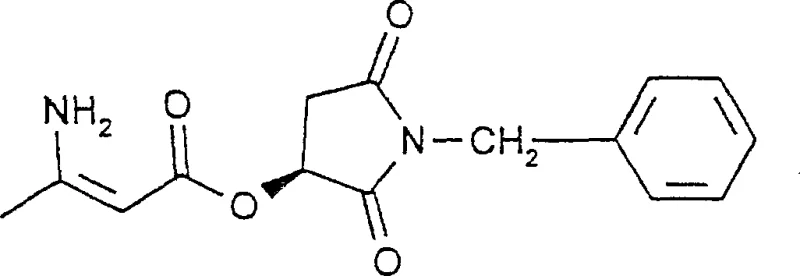
The synthesis protocol outlined in the patent provides a robust framework for producing high-value calcium antagonist intermediates. The process begins with the preparation of the chiral amino-butenate by reacting an enantiopure maleimide with diketene adducts, followed by ammonolysis to install the amine functionality. This intermediate is then condensed with a substituted benzaldehyde derivative under reflux conditions to form the dihydropyridine core. The detailed standardized synthesis steps see the guide below, which covers the specific stoichiometry, solvent choices, and workup procedures required to replicate the high yields and purity reported in the intellectual property.
- Preparation of the chiral amino-butenate intermediate by reacting enantiopure maleimide with diketene or its adducts, followed by ammonolysis.
- Condensation of the chiral amino-butenate with an enantiopure benzal compound in an inert solvent like isopropanol or ethyl acetate at elevated temperatures.
- Removal of the maleimide auxiliary group under mild basic conditions using DBU, followed by activation and esterification to yield the final pure dihydropyridine.
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthetic route offers substantial strategic advantages for procurement managers and supply chain heads focused on cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for precious metal catalysts and complex chiral chromatography, the process significantly lowers the raw material and operational expenditure associated with producing enantiopure compounds. The ability to purify intermediates through simple crystallization rather than column chromatography enhances throughput and reduces solvent consumption, aligning with modern green chemistry initiatives and environmental compliance standards.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent usage due to crystallization-based purification lead to direct cost savings. The high yield of the condensation step minimizes waste generation, further optimizing the cost of goods sold. Additionally, the mild conditions for auxiliary removal reduce energy consumption compared to harsh hydrolysis methods, contributing to a more economical production profile.
- Enhanced Supply Chain Reliability: The starting materials, including substituted benzaldehydes and maleimides, are readily available from established chemical suppliers, reducing the risk of raw material shortages. The robustness of the reaction conditions ensures consistent batch-to-batch quality, minimizing the likelihood of production delays caused by failed runs or out-of-specification results. This reliability is crucial for maintaining continuous supply lines for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed for commercial scale-up, utilizing common solvents like isopropanol and ethyl acetate that are easy to recover and recycle. The absence of heavy metal residues simplifies waste treatment and disposal, ensuring compliance with stringent environmental regulations. The high diastereoselectivity reduces the burden on downstream purification units, allowing for larger batch sizes and improved overall plant efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this maleimide-based synthesis route. These answers are derived directly from the experimental data and process descriptions within the patent documentation, providing clarity on reaction parameters and product specifications. Understanding these details is essential for technical teams evaluating the feasibility of adopting this methodology for their specific production needs.
Q: How does the maleimide auxiliary improve enantioselectivity compared to traditional methods?
A: The rigid cyclic structure of the maleimide auxiliary induces high diastereoselectivity during the Hantzsch-like condensation, allowing for easy separation of diastereomers via crystallization, unlike flexible chiral pool auxiliaries.
Q: What are the typical reaction conditions for removing the chiral auxiliary?
A: The maleimide group can be eliminated under very mild conditions using organic bases such as DBU or triethylamine at temperatures between 0°C and 50°C, preserving the sensitive dihydropyridine core.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process avoids expensive transition metal catalysts and utilizes robust crystallization steps for purification, making it highly scalable and cost-effective for industrial API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Dihydropyridine Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced synthetic technology to deliver high-purity pharmaceutical intermediates to the global market. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our rigorous QC labs and stringent purity specifications guarantee that every batch of enantiopure dihydropyridine meets the highest industry standards for calcium antagonist development.
We invite you to contact our technical procurement team to discuss how this innovative route can optimize your supply chain. Request a Customized Cost-Saving Analysis today to understand the potential economic benefits for your specific project. We are prepared to provide specific COA data and route feasibility assessments to support your R&D and commercialization goals efficiently.