Scalable Synthesis of Polymorphic 1,4-Dihydropyridine Derivatives for Advanced Optoelectronic Applications
Scalable Synthesis of Polymorphic 1,4-Dihydropyridine Derivatives for Advanced Optoelectronic Applications
The rapid evolution of smart materials has necessitated the development of robust organic fluorophores capable of functioning efficiently in solid-state environments. Patent CN107382978B introduces a groundbreaking 1,4-dihydropyridine derivative that overcomes the traditional limitations of fluorescence quenching in aggregated states. This innovative compound exhibits remarkable aggregation-induced emission (AIE) characteristics alongside polymorphic piezoluminescence, making it an ideal candidate for next-generation pressure sensors and anti-counterfeiting technologies. By leveraging a streamlined three-step synthetic pathway, manufacturers can now access high-purity functional materials that offer tunable optical properties through simple recrystallization techniques. This technical insight explores the mechanistic advantages and commercial viability of this novel synthetic route for global supply chains.
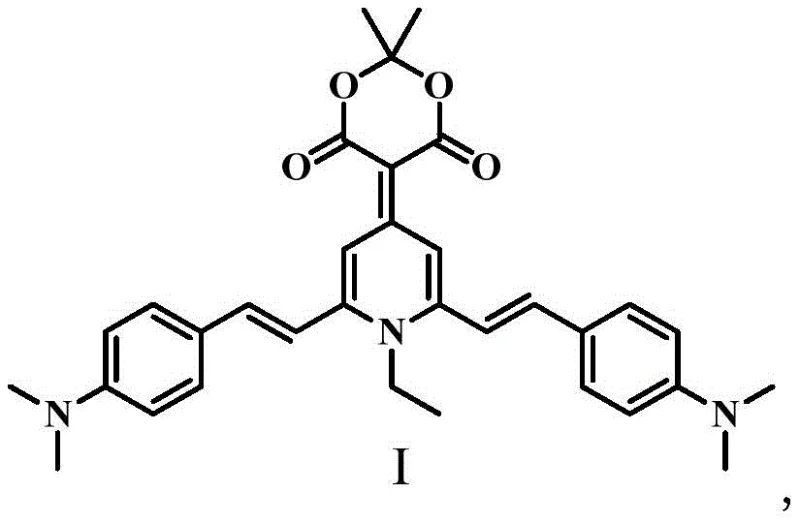
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional 1,4-dihydropyridine derivatives have long served as fundamental building blocks in medicinal chemistry and material science, yet their application in solid-state lighting has been severely restricted by the aggregation-caused quenching (ACQ) effect. In conventional systems, planar molecular structures tend to stack tightly via π-π interactions in the solid state or high-concentration solutions, leading to non-radiative decay pathways that drastically diminish fluorescence efficiency. This phenomenon renders many potential fluorescent materials useless for practical applications such as thin-film coatings or solid-state sensors where high concentration is inevitable. Furthermore, achieving multi-color emission typically requires synthesizing entirely new chemical scaffolds with different conjugated systems, a process that is both time-consuming and economically inefficient for industrial scaling. The inability to modulate emission color through physical processing rather than chemical synthesis represents a significant bottleneck in the development of versatile optoelectronic materials.
The Novel Approach
The methodology disclosed in the patent data presents a paradigm shift by engineering a 1,4-dihydropyridine core with bulky substituents that enforce a twisted molecular conformation. This structural distortion effectively prevents close π-π stacking, thereby activating the aggregation-induced emission (AIE) mechanism which enhances fluorescence intensity in the aggregated state. Uniquely, this approach allows for the generation of multiple crystalline polymorphs from a single chemical entity simply by varying the recrystallization solvent system. This means that yellow, orange, and red emitting materials can be produced from the same batch of intermediate, offering unprecedented flexibility in manufacturing. The synthetic route avoids the use of scarce transition metal catalysts, relying instead on abundant organic reagents and standard condensation reactions, which significantly simplifies the purification process and reduces the environmental footprint associated with heavy metal removal.
Mechanistic Insights into the Three-Step Condensation and Cyclization Strategy
The synthesis begins with an addition-elimination reaction between 2,6-dimethyl-4-pyrone and Meldrum's acid in acetic anhydride, generating a highly reactive intermediate that serves as the electron-deficient core for subsequent transformations. This step is critical as it establishes the conjugated framework necessary for fluorescence, while the specific reaction conditions ensure high conversion rates without the formation of complex byproducts. Following this, a Knoevenagel condensation with 4-dimethylaminobenzaldehyde extends the conjugation length and introduces electron-donating groups, creating a push-pull electronic system essential for strong intramolecular charge transfer (ICT). The final cyclization involves a nucleophilic substitution with ethylamine, which closes the dihydropyridine ring and locks the molecular geometry into the desired twisted conformation. This precise control over molecular architecture is what enables the material to exhibit distinct piezochromic behavior, where mechanical stress alters the molecular packing and consequently the emission wavelength.
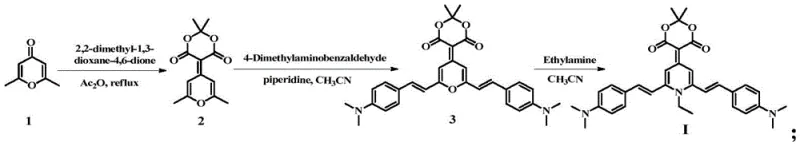
Impurity control is inherently managed through the choice of reagents and the crystallization-driven purification strategy employed in the final steps. The use of acetic anhydride in the initial step acts both as a solvent and a dehydrating agent, driving the equilibrium towards the desired intermediate while minimizing hydrolysis side reactions. In the condensation phase, the use of piperidine as a base catalyst in acetonitrile provides a homogeneous reaction environment that promotes selective bond formation. The final product isolation relies on temperature-controlled precipitation and washing with methanol, which effectively removes unreacted amines and soluble organic impurities. Most importantly, the recrystallization step not only defines the polymorphic form but also serves as a final purification stage, ensuring that the resulting crystalline compounds meet the stringent purity specifications required for high-performance electronic applications.
How to Synthesize 1,4-Dihydropyridine Derivative Efficiently
The preparation of this advanced functional material follows a logical progression of organic transformations that are well-suited for batch processing in standard chemical reactors. The process initiates with the thermal reaction of pyrone derivatives, followed by a base-catalyzed condensation, and concludes with an amine-mediated cyclization. Each step has been optimized to balance reaction kinetics with product stability, ensuring that the sensitive fluorescent core is not degraded during synthesis. Operators should pay close attention to the stoichiometry of Meldrum's acid and the temperature profiles during the initial heating phase, as these parameters directly influence the yield of the key intermediate. For detailed operational parameters including specific molar ratios, temperature ranges, and solvent volumes, please refer to the standardized synthesis guide below.
- React 2,6-dimethyl-4-pyrone with Meldrum's acid in acetic anhydride at elevated temperatures to form the key intermediate.
- Perform a Knoevenagel condensation between the intermediate and 4-dimethylaminobenzaldehyde using piperidine in acetonitrile.
- Execute nucleophilic substitution with ethylamine followed by specific recrystallization protocols to obtain distinct crystalline forms.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages by utilizing commodity chemicals that are readily available in the global market, thereby mitigating supply chain risks associated with specialized reagents. The elimination of precious metal catalysts not only reduces the raw material cost but also simplifies the regulatory compliance landscape, as there is no need for extensive testing and validation for residual heavy metals in the final product. This translates to a significantly streamlined quality assurance process and faster time-to-market for downstream applications. Furthermore, the ability to produce multiple product variants (different emission colors) from a single intermediate inventory allows for greater agility in responding to market demands without maintaining separate stockpiles of diverse chemical entities.
- Cost Reduction in Manufacturing: The process achieves cost efficiency through the use of inexpensive starting materials like 2,6-dimethyl-4-pyrone and acetic anhydride, which are produced at massive scales globally. By avoiding complex catalytic systems and expensive ligands, the overall bill of materials is drastically reduced compared to transition-metal mediated cross-coupling routes. Additionally, the workup procedures involve simple filtration and washing steps rather than energy-intensive chromatography, leading to lower utility consumption and labor costs per kilogram of product. The high atom economy of the condensation reactions further contributes to waste minimization, aligning with green chemistry principles that often correlate with long-term operational savings.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents ensures that production schedules are not disrupted by the degradation of sensitive catalysts or the scarcity of exotic precursors. Since the synthesis does not require inert atmosphere conditions for every step (some steps can be performed in air), the barrier to entry for contract manufacturing organizations is lower, expanding the pool of potential suppliers. This redundancy in the supply base enhances resilience against geopolitical disruptions or logistical bottlenecks. The robust nature of the intermediates also allows for safer transportation and storage, reducing the risk of spoilage during transit and ensuring consistent quality upon arrival at the formulation site.
- Scalability and Environmental Compliance: The reaction conditions described, such as reflux in acetic anhydride or heating in acetonitrile, are easily transferable from laboratory glassware to industrial-scale reactors without significant re-engineering. The solvents used are common organic liquids with established recovery and recycling infrastructure, facilitating effective waste management and solvent reuse programs. The absence of toxic heavy metals simplifies wastewater treatment protocols and reduces the burden on environmental health and safety departments. This ease of scale-up ensures that production volumes can be increased rapidly to meet surging demand for smart materials in the electronics and security sectors without compromising on product consistency or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, application, and handling of this polymorphic fluorescent material. Understanding these aspects is crucial for integrating the compound into existing manufacturing workflows and product designs. The answers are derived directly from the experimental data and characterization results provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What distinguishes this 1,4-dihydropyridine derivative from traditional fluorescent materials?
A: Unlike conventional fluorophores that suffer from aggregation-caused quenching (ACQ), this derivative exhibits Aggregation-Induced Emission (AIE) and unique piezochromic properties, allowing it to emit strong fluorescence in solid states and change color under mechanical pressure.
Q: How does the recrystallization process affect the material's optical properties?
A: The patent demonstrates that utilizing different solvent systems during recrystallization yields three distinct polymorphs (I-y, I-o, I-r) which emit yellow, orange, and red fluorescence respectively, enabling tunable optical output without altering the chemical structure.
Q: Is the synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes readily available starting materials like 2,6-dimethyl-4-pyrone and employs standard organic transformations such as condensation and nucleophilic substitution, avoiding expensive transition metal catalysts and facilitating straightforward scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Dihydropyridine Derivative Supplier
As the demand for intelligent sensing materials continues to grow, having a partner with deep expertise in complex organic synthesis is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed analysis of polymorphic forms and fluorescence quantum yields. We understand the critical nature of batch-to-batch reproducibility in the electronic chemical sector and employ advanced process analytical technology to monitor every stage of the synthesis.
We invite you to collaborate with our technical team to explore how this novel 1,4-dihydropyridine derivative can enhance your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your R&D and sourcing strategies, ensuring a seamless transition from concept to commercial reality.
