Scalable Synthesis of High-Purity Anticoagulant Intermediates via Novel Amidoxime Reduction
Scalable Synthesis of High-Purity Anticoagulant Intermediates via Novel Amidoxime Reduction
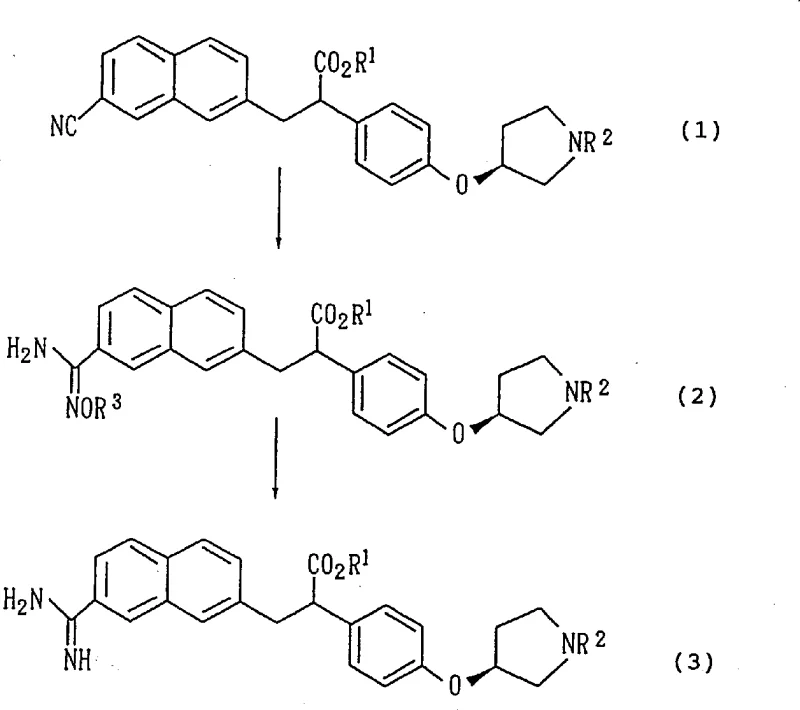
The pharmaceutical industry continuously seeks robust synthetic routes for complex active pharmaceutical ingredient (API) intermediates, particularly those possessing chiral centers critical for biological activity. Patent CN1258275A discloses a groundbreaking process for preparing 3-(7-amidino-2-naphthyl)-2-phenylpropionic acid derivatives, which serve as vital intermediates for aromatic amidine derivatives with potent anticoagulant activity. This technology represents a significant paradigm shift from traditional Pinner reaction methodologies, addressing long-standing challenges regarding optical purity retention and process safety. By leveraging a hydroxylamine-mediated conversion followed by catalytic reduction, this method ensures the structural integrity of sensitive chiral moieties while facilitating industrial scalability. For procurement specialists and R&D leaders, understanding this mechanistic advantage is crucial for securing a reliable API intermediate supplier capable of delivering high-purity materials consistently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic amidines has relied heavily on the Pinner reaction, a classical transformation involving the reaction of a nitrile with an alcohol in the presence of an acid catalyst, typically anhydrous hydrogen chloride, followed by treatment with ammonia. While chemically sound in theory, this approach presents severe drawbacks when applied to complex, chiral molecules like those described in Japanese Patent Laid-Open No. 5-208946. The requirement for strongly acidic conditions and the generation of imino ether intermediates create a hostile environment for stereocenters, leading to partial epimerization. As documented in comparative studies, this degradation of stereochemical integrity results in a noticeable drop in optical purity, falling from an initial 99.7% de to approximately 94.8% de in the final product. Furthermore, the operational hazards associated with saturating large reactors with hydrogen chloride gas and subsequently handling ammonia gas make this method unsuitable for modern, safety-conscious manufacturing environments. The reaction kinetics are also sluggish, often requiring reaction times extending up to a week to reach completion at low temperatures, which drastically reduces throughput and increases energy consumption.
The Novel Approach
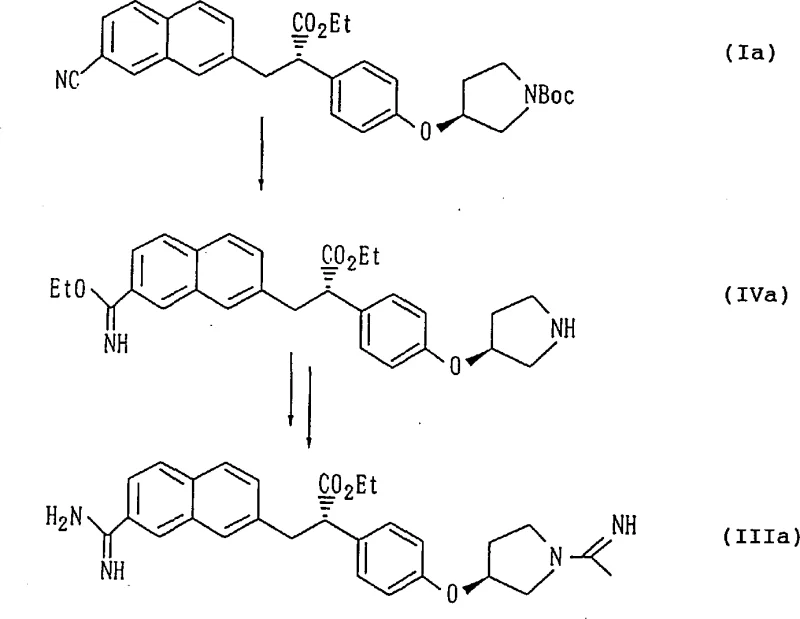
In stark contrast, the methodology outlined in CN1258275A introduces a温和 (mild) yet highly efficient two-step sequence that circumvents the thermodynamic pitfalls of the Pinner reaction. The process initiates with the nucleophilic addition of hydroxylamine to the nitrile group, forming an amidoxime intermediate under neutral to slightly basic conditions. This step is kinetically favorable and proceeds rapidly, often completing within hours rather than days. The subsequent reduction of the amidoxime to the desired amidine can be achieved through catalytic hydrogenation using palladium on carbon with formic acid as a hydrogen donor, or via metal reduction using zinc or iron in acidic media. Crucially, these conditions are sufficiently mild to prevent the racemization of the adjacent chiral center. Experimental data confirms that starting materials with 99.7% optical purity yield final products retaining 99.1% optical purity, demonstrating exceptional stereochemical fidelity. This route not only enhances product quality but also simplifies the workflow by eliminating the need for hazardous gaseous reagents, making it an ideal candidate for cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Amidoxime Formation and Reduction
The core innovation of this process lies in the stability of the amidoxime intermediate and the selectivity of the reduction step. In the first stage, the nitrile carbon, which is electrophilic, undergoes attack by the nucleophilic nitrogen of the hydroxylamine species. This reaction is typically conducted in alcoholic solvents such as ethanol or methanol, which solubilize both the organic nitrile substrate and the inorganic hydroxylamine salts. The use of hydroxylamine sulfate or hydrochloride, neutralized in situ with sodium hydroxide, provides a controlled source of free hydroxylamine without generating excessive heat. The resulting amidoxime possesses a unique electronic structure where the electron density is delocalized across the N-O and C-N bonds, rendering it stable enough for isolation yet reactive enough for subsequent transformation. This stability is key to preventing side reactions that often plague direct nitrile reductions.
The second stage involves the cleavage of the N-O bond to generate the primary amine functionality of the amidine. When utilizing catalytic hydrogenation, the palladium surface facilitates the adsorption of hydrogen (generated in situ from formic acid decomposition) and the amidoxime substrate. The mechanism likely proceeds via the formation of a transient hydroxylamine species which is rapidly reduced further to the amine. Alternatively, metal-mediated reduction in acidic conditions protonates the oxime oxygen, making it a better leaving group, followed by electron transfer from the metal surface. Both pathways operate under conditions that do not promote the enolization of the adjacent alpha-carbon, which is the primary driver of epimerization in the older acid-catalyzed routes. By avoiding strong Lewis acids and high thermal stress, the process effectively locks the stereochemistry in place, ensuring that the final API intermediate meets the stringent purity specifications required for anticoagulant drugs.
How to Synthesize 3-(7-amidino-2-naphthyl) Derivatives Efficiently
The synthesis protocol described in the patent offers a clear, reproducible pathway for laboratory and pilot-scale production. The procedure begins with the suspension of the nitrile precursor in ethanol, followed by the addition of an aqueous hydroxylamine solution. The mixture is heated to reflux, driving the equilibrium towards the amidoxime product, which can often be isolated directly by cooling and crystallization. The crude amidoxime is then subjected to reduction, preferably using 10% palladium-carbon and formic acid in ethanol at ambient temperature. This specific combination minimizes the risk of over-reduction or degradation of other sensitive functional groups, such as the ester moiety present on the propionic acid side chain. Following the reaction, the catalyst is removed by filtration, and the product is converted into a stable salt, such as the maleate or hydrochloride, to facilitate purification and storage.
- React the nitrile precursor with hydroxylamine or its salt in an alcoholic solvent (e.g., ethanol) under reflux to form the amidoxime intermediate.
- Perform catalytic hydrogenation using a palladium-carbon catalyst with formic acid as the hydrogen source, or use metal reduction (Zn/Fe) in acidic media.
- Isolate the final amidine product via crystallization, optionally forming stable salts such as maleate or hydrochloride for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain managers and procurement officers, the adoption of this synthetic route offers tangible benefits beyond mere chemical elegance. The elimination of hazardous gaseous reagents like anhydrous hydrogen chloride translates directly into reduced capital expenditure on specialized corrosion-resistant equipment and gas scrubbing systems. This simplification of the engineering controls allows for faster batch turnover and lower maintenance costs, contributing to substantial cost savings in the overall manufacturing budget. Furthermore, the shortened reaction time—from over a week in conventional methods to merely a few hours—dramatically increases asset utilization rates, enabling manufacturers to respond more agilely to market demand fluctuations without the need for excessive inventory buffering.
- Cost Reduction in Manufacturing: The process utilizes commodity chemicals such as hydroxylamine sulfate, formic acid, and ethanol, which are readily available in the global market at stable prices. By avoiding the need for cryogenic conditions or high-pressure ammonia reactors, the operational expenditure (OPEX) is significantly lowered. The ability to isolate intermediates via simple crystallization rather than complex chromatographic separations further drives down the cost of goods sold (COGS), making the final API intermediate more price-competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Dependence on specialized reagents often creates bottlenecks in the supply chain. This method relies on robust, widely sourced raw materials, mitigating the risk of supply disruptions. The high yield and purity profile reduce the likelihood of batch failures, ensuring a consistent flow of material to downstream API synthesis sites. This reliability is critical for maintaining uninterrupted production schedules for life-saving anticoagulant medications, where supply continuity is a regulatory and ethical imperative.
- Scalability and Environmental Compliance: The reaction conditions are inherently safer and generate less hazardous waste compared to the Pinner reaction. The absence of large volumes of acidic wastewater containing ammonium salts simplifies effluent treatment processes. This aligns with increasingly strict environmental regulations and sustainability goals, reducing the environmental footprint of the manufacturing site. The process is demonstrably scalable, as evidenced by the patent examples, supporting seamless transition from clinical trial material to commercial tonnage production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing production portfolios.
Q: How does this process improve optical purity compared to conventional methods?
A: Conventional Pinner reactions often cause epimerization due to harsh acidic conditions and prolonged reaction times. This novel amidoxime route operates under milder conditions, maintaining optical purity above 99% de compared to the 94.8% de observed in older methods.
Q: What are the key safety advantages of this manufacturing process?
A: The process eliminates the need for handling large volumes of hazardous anhydrous hydrogen chloride gas and ammonia gas, which are required in traditional Pinner synthesis, thereby significantly reducing operational risks and containment costs.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights industrial applicability. The use of standard catalytic hydrogenation equipment and common solvents like ethanol allows for straightforward scale-up from kilogram to multi-ton production without complex engineering controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(7-amidino-2-naphthyl) Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation anticoagulants. Our technical team has extensively analyzed the process disclosed in CN1258275A and possesses the expertise to implement this superior amidoxime reduction pathway effectively. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in practical, large-volume manufacturing. Our facilities are equipped with state-of-the-art hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee optical purity levels exceeding 99% de.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and robust synthetic route. By optimizing the supply chain for these complex intermediates, we can help you accelerate your drug development timelines while managing costs efficiently. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals.
